Advanced Electrochemical Reduction for High-Purity 3-Aminopyridine Derivatives and Commercial Scale-Up
Advanced Electrochemical Reduction for High-Purity 3-Aminopyridine Derivatives and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize critical heterocyclic intermediates. Patent CN1115755A introduces a groundbreaking methodology for the preparation of 3-aminopyridines from their corresponding 3-nitropyridine precursors through an innovative electrochemical reduction process. Unlike traditional chemical reductions that rely on stoichiometric amounts of heavy metal reagents, this technique utilizes electrical energy to drive the transformation in an acidic medium. This dual-function reaction not only reduces the nitro group to an amino group but also simultaneously introduces a substituent at the 2-position of the pyridine ring, creating valuable 2-substituted-3-aminopyridine derivatives in a single operational step. This technological leap represents a significant advancement for manufacturers aiming to streamline their supply chains for anticonvulsant and antiviral drug intermediates.
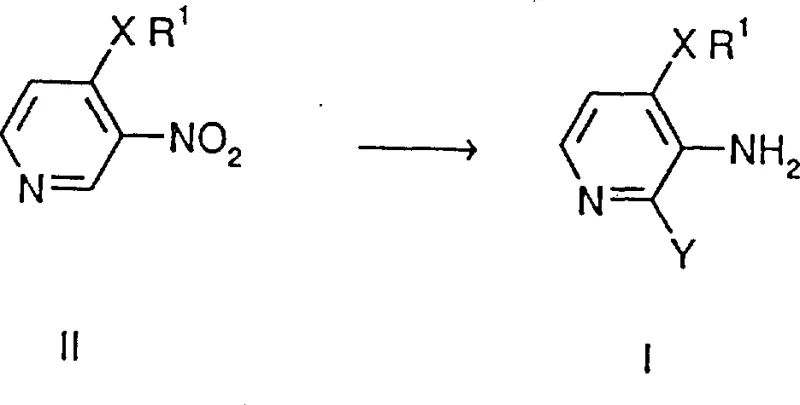
The core innovation lies in the ability to perform this transformation under mild conditions while achieving high regioselectivity. The process operates effectively in acidic solutions containing hydrochloric acid, sulfuric acid, or methyl sulfuric acid, often supplemented with electrolytes or co-solvents like water or dioxane. By controlling parameters such as current density, temperature, and electrode material, manufacturers can precisely dictate the outcome of the reaction. This level of control is essential for producing high-purity pharmaceutical intermediates where impurity profiles must be strictly managed to meet global regulatory standards. The versatility of this method allows for the synthesis of various derivatives, including chloro, alkoxy, and thio-substituted aminopyridines, which serve as key building blocks for complex medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitropyridines to aminopyridines has relied heavily on chemical reducing agents, most notably tin(II) chloride. While chemically effective, this traditional approach suffers from severe drawbacks that impact both economic viability and environmental sustainability. The reduction process generates substantial quantities of tin(IV) salts as by-products, which are not only difficult to separate from the desired organic product but also pose significant disposal challenges. The handling of these heavy metal wastes requires specialized treatment facilities and incurs high operational costs related to environmental compliance and waste management. Furthermore, the presence of residual tin in the final product can be detrimental to downstream catalytic processes or final drug formulation, necessitating rigorous and costly purification steps to ensure the material meets stringent quality specifications required by health authorities.
The Novel Approach
In stark contrast, the electrochemical method described in the patent offers a clean, sustainable alternative that fundamentally changes the economics of aminopyridine production. By replacing chemical reductants with electrons, the process eliminates the generation of heavy metal waste streams entirely. The reaction takes place in an electrolytic cell where the cathode facilitates the reduction of the nitro group, while the acidic medium provides the necessary protons and nucleophiles for the simultaneous substitution at the 2-position. This integration of reduction and functionalization into a single unit operation significantly simplifies the workflow. The use of standard electrode materials like graphite, copper, or platinum, combined with commercially available ion-exchange membranes, ensures that the technology can be implemented using existing infrastructure without the need for exotic or prohibitively expensive equipment, thereby lowering the barrier to entry for adoption.
Mechanistic Insights into Electrochemical Reductive Functionalization
The mechanistic pathway of this electrochemical transformation is driven by the transfer of electrons at the cathode surface, which initiates the reduction of the nitro group. In the acidic environment, the nitro group undergoes a multi-electron reduction process to form the corresponding amine. Crucially, the presence of specific anions or nucleophiles in the catholyte—derived from the acid or added salts—allows for a concurrent nucleophilic attack at the 2-position of the pyridine ring. This regioselective substitution is facilitated by the activation of the ring system during the reduction phase. The use of a diaphragm or cation exchange membrane, such as those based on Nafion fluorocarbon matrices, is critical to separate the anode and cathode compartments. This separation prevents the oxidation of the newly formed amine or the nucleophile at the anode, ensuring high current efficiency and product yield. The careful selection of cathode material, ranging from lead and zinc to graphite and precious metals, further optimizes the overpotential and selectivity of the reaction.
Impurity control in this electrochemical system is inherently superior to chemical reduction methods due to the absence of metal salt contaminants. The primary by-products are typically limited to over-reduced species or minor isomeric variations, which can be easily managed through crystallization or standard extraction techniques. For instance, the patent highlights that products like 2-chloro-3,4-diaminopyridine can be isolated directly from the catholyte by cooling and crystallization, achieving yields as high as 92% of theory. The ability to tune the reaction conditions, such as maintaining temperatures between 50°C and 60°C and adjusting current densities between 1 and 6 A/dm², allows process chemists to minimize side reactions. This precise control over the reaction environment ensures a consistent and clean impurity profile, which is a critical factor for R&D directors evaluating the feasibility of scaling up complex heterocyclic syntheses for clinical and commercial applications.
How to Synthesize 2-Substituted 3-Aminopyridines Efficiently
The synthesis of these valuable intermediates follows a streamlined protocol that leverages the principles of organic electrochemistry. The process begins with the preparation of the catholyte, where the 3-nitropyridine starting material is dissolved in an acidic medium tailored to introduce the desired functional group. For chloro-substitution, hydrochloric acid is used, whereas methoxy-substitution requires the presence of methanol and sulfuric acid. The electrolytic cell is then assembled with appropriate electrodes and a separating membrane to maintain compartment integrity. Detailed standardized synthesis steps see the guide below.
- Prepare the catholyte by dissolving the 3-nitropyridine substrate (1-20% w/w) in an acidic solution containing hydrochloric or sulfuric acid, optionally with alcohols or salts depending on the desired 2-position substituent.
- Assemble the electrolytic cell using a cation exchange membrane (e.g., Nafion) to separate the anode and cathode compartments, utilizing graphite or metal plates as electrodes.
- Conduct electrolysis at a controlled current density of 1-6 A/dm² and temperature between 50-60°C until the theoretical charge load is reached, followed by crystallization or extraction to isolate the pure amino product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology presents a compelling value proposition centered on cost optimization and risk mitigation. The elimination of tin-based reagents removes a significant variable cost associated with raw material procurement and waste disposal. Traditional methods incur hidden costs related to the storage, handling, and treatment of hazardous heavy metal sludge, which can fluctuate with changing environmental regulations. By switching to an electron-driven process, companies can achieve substantial cost savings in waste management and reduce their overall environmental footprint. Furthermore, the simplified workup procedure, often involving direct crystallization from the reaction mixture, reduces solvent consumption and processing time, leading to a more efficient manufacturing cycle that enhances overall throughput.
- Cost Reduction in Manufacturing: The transition from stoichiometric chemical reduction to electrocatalysis fundamentally alters the cost structure of aminopyridine production. By removing the need for expensive and hazardous reducing agents like tin(II) chloride, the direct material costs are significantly lowered. Additionally, the avoidance of heavy metal waste generation eliminates the substantial expenses associated with hazardous waste disposal and regulatory compliance reporting. The process efficiency is further enhanced by the high yields reported in the patent examples, which maximize the utilization of the starting nitropyridine feedstock. This combination of lower input costs and higher output efficiency results in a markedly improved margin profile for the final active pharmaceutical ingredient or intermediate.
- Enhanced Supply Chain Reliability: Relying on electrochemical synthesis diversifies the supply chain by reducing dependence on specific chemical reagents that may be subject to market volatility or supply disruptions. Electricity is a universally available utility, and the electrode materials required, such as graphite or stainless steel, are commodity items with stable supply lines. This robustness ensures continuous production capability even when traditional chemical supply chains face constraints. Moreover, the scalability of electrolytic cells allows for flexible production volumes, enabling manufacturers to respond quickly to fluctuations in demand without the long lead times associated with sourcing specialized chemical reagents. This agility is crucial for maintaining reliable delivery schedules to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: From a sustainability perspective, this technology aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. The absence of heavy metal effluents simplifies the permitting process for new manufacturing facilities and reduces the liability associated with long-term environmental monitoring. The process is inherently scalable, as demonstrated by the use of filter-press type electrolyzers and flow cells which are standard in the fine chemical industry. Scaling from laboratory benchtop units to multi-ton industrial reactors involves linear engineering adjustments rather than fundamental chemical changes, de-risking the commercialization pathway. This ease of scale-up ensures that supply can be ramped up rapidly to meet commercial demands while maintaining full compliance with global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 3-aminopyridines. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details is vital for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary environmental advantages of this electrochemical method over traditional reduction?
A: The primary advantage is the complete elimination of stoichiometric heavy metal reducing agents like tin(II) chloride. Traditional methods generate massive quantities of toxic tin(IV) salt waste that requires complex and costly disposal procedures. This electrochemical approach uses electrons as the reagent, resulting in a significantly cleaner process with minimal inorganic waste streams.
Q: Can this process introduce different functional groups at the 2-position of the pyridine ring?
A: Yes, the process is highly versatile regarding regioselectivity. By modifying the composition of the acidic catholyte—such as using hydrochloric acid for chloro-groups, sulfuric acid/alcohol mixtures for alkoxy-groups, or specific salts for other nucleophiles—the Y group can be selectively introduced at the 2-position while simultaneously reducing the 3-nitro group to an amine.
Q: Is this electrochemical synthesis suitable for large-scale industrial production?
A: Absolutely. The patent specifies the use of conventional electrolytic cells with diaphragms or ion-exchange membranes, which are standard equipment in fine chemical manufacturing. The ability to operate at moderate temperatures (50-60°C) and ambient pressure, combined with high current efficiencies, makes the technology readily scalable from pilot plants to multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering high-purity 3-aminopyridine derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in managing complex heterocyclic chemistry allows us to optimize reaction parameters for maximum yield and minimal impurity formation, providing our partners with a competitive edge in the global market.
We invite pharmaceutical and agrochemical companies to explore the benefits of this advanced manufacturing route for their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic and environmental benefits of switching to electrochemical methods. We encourage you to contact our technical procurement team to request specific COA data for our electrochemically synthesized intermediates and to discuss route feasibility assessments tailored to your specific project requirements. Let us help you build a more sustainable and cost-effective supply chain for your critical drug candidates.
