Advanced Manufacturing Strategy for High-Purity Topramezone Intermediates via Optimized Oxidation
Advanced Manufacturing Strategy for High-Purity Topramezone Intermediates via Optimized Oxidation
The global demand for high-efficiency herbicides continues to drive innovation in the agrochemical intermediate sector, specifically for compounds targeting p-hydroxyphenylpyruvate dioxygenase (4-HPPD). A pivotal development in this domain is documented in patent CN108218851B, which discloses an improved preparation method for Topramezone, a broad-spectrum post-emergence herbicide widely used in corn cultivation. This technical breakthrough addresses long-standing inefficiencies in synthetic routes by utilizing 3-nitro-o-xylene as a foundational raw material, guiding it through a sophisticated sequence of oximation, ethylene ring closure, palladium-carbon reduction, diazotization, hydroformylation, and a novel oxidation-condensation rearrangement. The significance of this patent lies not merely in the chemical transformation but in its ability to achieve a total yield of 33.7 percent under optimized conditions, a substantial metric that directly impacts the economic viability of large-scale manufacturing. For research and development directors and procurement strategists, understanding the nuances of this pathway is critical for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality.
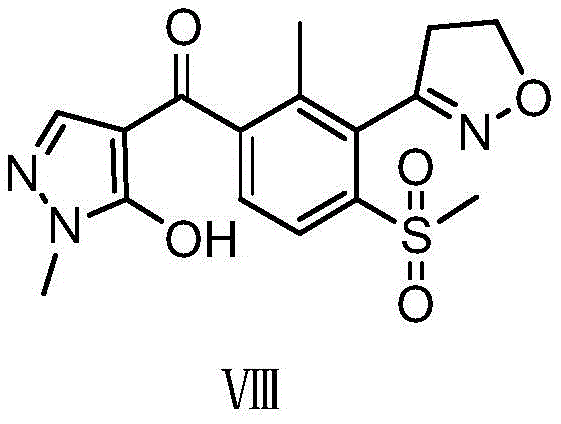
Topramezone, chemically defined as [3-(4,5-dihydro-1,2-oxazol-3-yl)-4-methanesulfonyl-2-methylphenyl](5-hydroxy-1-methylpyrazol-4-yl)methanone, represents a complex molecular architecture requiring precise construction of both pyrazole and isoxazole heterocycles. The structural integrity of the final molecule is paramount for its biological activity against annual gramineous and broadleaf weeds. As illustrated in the structural diagram, the presence of the methylsulfonyl group and the specific orientation of the ketone bridge are critical features that traditional synthesis methods have struggled to assemble efficiently. The patent CN108218851B provides a roadmap that navigates these structural challenges by optimizing reaction conditions at every stage, ensuring that the final product meets the stringent purity specifications required by regulatory bodies and end-users in the agricultural sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial synthesis of Topramezone was plagued by significant bottlenecks that hindered cost reduction in herbicide manufacturing. Existing literature, including patents from major entities like BASF and Japanese corporations, reported routes characterized by low overall yields and reliance on purification techniques that are impractical for ton-scale production, such as column chromatography. For instance, the synthesis route disclosed in Chinese patent CN103788083A, while utilizing similar starting materials, achieved a total yield of only 30.2 percent. This seemingly small difference in percentage translates to massive losses in material throughput and increased waste generation when projected over annual production volumes. Furthermore, conventional methods often involve harsh reaction conditions or expensive reagents that complicate the supply chain and increase the risk of batch-to-batch variability. The reliance on difficult-to-implement steps creates a fragile production environment where minor deviations can lead to significant impurity profiles, necessitating costly rework or disposal.
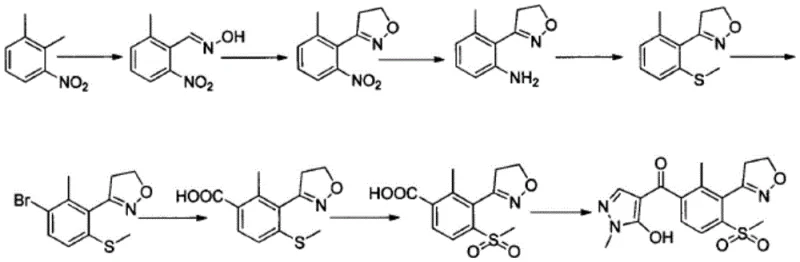
The Novel Approach
The methodology presented in CN108218851B offers a transformative solution by re-engineering the synthetic pathway to enhance both efficiency and scalability. The core innovation lies in the strategic modification of the oxidation and formylation steps, which are traditionally the most problematic stages in constructing the benzoyl-isoxazole scaffold. By replacing inefficient oxidation protocols with a controlled hydrogen peroxide system in glacial acetic acid, the new method achieves a cleaner conversion of the methylthio intermediate to the requisite methylsulfonyl derivative. This approach not only improves the yield of this specific step but also simplifies the downstream workup, eliminating the need for complex purification columns. The result is a streamlined process that maintains high fidelity to the target molecular structure while significantly reducing the operational burden on manufacturing teams. This novel approach demonstrates that through careful optimization of reaction parameters, it is possible to overcome the historical limitations of Topramezone synthesis.
Mechanistic Insights into Optimized Oxidation and Formylation
A deep dive into the reaction mechanism reveals why this specific pathway offers superior control over impurity formation. The critical transformation involves the conversion of compound VI to compound VII, where the aldehyde-functionalized methylthio-isoxazole is oxidized to the corresponding sulfone. In this patent, the reaction is conducted by mixing the substrate with glacial acetic acid at room temperature, followed by the slow addition of a hydrogen peroxide solution. The reaction temperature is carefully maintained between 10 and 50 degrees Celsius for a duration of 5 to 30 hours. This mild thermal profile is crucial; it prevents the degradation of the sensitive isoxazole ring and the aldehyde group, which are prone to side reactions under harsher oxidative conditions. The use of glacial acetic acid as a solvent facilitates the solubility of the organic substrate while stabilizing the peroxide species, ensuring a homogeneous reaction environment that promotes uniform conversion. This mechanistic precision is what allows the process to achieve the reported high yields without generating excessive tarry byproducts.
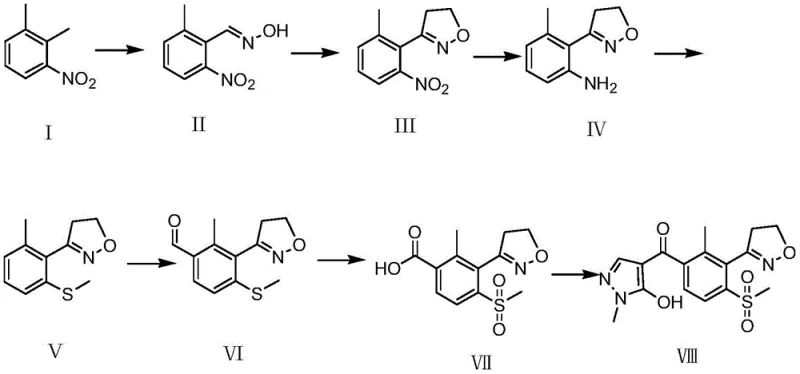
Furthermore, the preceding formylation step, which converts compound V to compound VI, employs a lithiation strategy using n-butyllithium and TMEDA (tetramethylethylenediamine) at low temperatures ranging from -78 to 0 degrees Celsius. The addition of DMF or similar aldehyde sources subsequently installs the formyl group with high regioselectivity. The patent specifies a molar ratio of compound V to TMEDA of 1:2.0-3.0 and a specific concentration of n-butyllithium (2.5 mol/L in n-hexane) to ensure complete deprotonation of the aromatic ring. This level of detail in the mechanistic description underscores the importance of stoichiometry and temperature control in preventing over-lithiation or nucleophilic attack on the isoxazole nitrogen. By rigorously controlling these variables, the process minimizes the formation of regio-isomers and other structural impurities, resulting in a crude product that requires minimal purification before proceeding to the final condensation with 1-methyl-5-hydroxypyrazole.
How to Synthesize Topramezone Efficiently
Implementing this improved synthesis route requires strict adherence to the operational parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the nitro-oxime intermediate and proceeds through high-pressure cyclization and catalytic reduction, each step building the complexity of the molecule. The critical phase involves the lithiation and subsequent oxidation, where precise temperature monitoring and reagent addition rates are essential to maintain the integrity of the intermediates. Operators must be trained to handle reactive species like n-butyllithium and hydrogen peroxide with appropriate safety protocols, particularly during the exothermic oxidation phase. The detailed standard operating procedures derived from this patent provide a robust framework for scaling the reaction from laboratory benchtops to pilot plants, ensuring that the chemical potential of the route is fully realized in a production setting.
- Perform oximation of 3-nitro-o-xylene followed by high-pressure ethylene ring closure to form the dihydroisoxazole core.
- Execute palladium-carbon catalytic reduction and subsequent diazotization to introduce the methylthio group.
- Conduct lithiation and formylation to install the aldehyde group, followed by oxidation with hydrogen peroxide to generate the sulfone.
- Finalize the synthesis through condensation and rearrangement with 1-methyl-5-hydroxypyrazole to yield Topramezone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN108218851B process offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage is the substantial cost savings achieved through the elimination of expensive and time-consuming purification steps. By avoiding column chromatography, which is a major bottleneck in fine chemical manufacturing, producers can drastically reduce solvent consumption and labor costs associated with fraction collection and analysis. This simplification of the workflow translates directly into a more competitive pricing structure for the final active ingredient, allowing agrochemical companies to maintain healthy margins even in volatile market conditions. Additionally, the use of readily available raw materials like 3-nitro-o-xylene ensures that the supply chain remains resilient against shortages, as these commodities are produced by multiple vendors globally.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods sold by minimizing waste and reducing the number of unit operations. The replacement of complex purification methods with straightforward extraction and crystallization steps reduces the consumption of high-purity solvents and silica gel, which are significant cost drivers in intermediate production. Furthermore, the improved yield means that less raw material is required to produce the same amount of final product, effectively amplifying the purchasing power of the procurement budget. This efficiency gain is compounded by the reduced energy consumption associated with shorter reaction times and milder heating requirements compared to legacy routes.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures. The tolerance of the reaction conditions to minor variations in temperature and stirring rates makes the process more forgiving in a multi-purpose plant environment. This reliability is crucial for meeting just-in-time delivery schedules demanded by downstream formulators. Moreover, the availability of key reagents such as hydrogen peroxide and glacial acetic acid ensures that production is not held hostage by the supply constraints of exotic catalysts or specialized ligands. This stability allows supply chain planners to forecast inventory levels with greater confidence and reduce the need for safety stock buffers.
- Scalability and Environmental Compliance: From an environmental perspective, the new method aligns well with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of heavy metal catalysts in the oxidation step and the use of aqueous workups minimize the load on wastewater treatment facilities. This compliance with environmental regulations reduces the risk of production shutdowns due to permit violations and lowers the cost of waste disposal. The scalability of the process is further evidenced by the successful demonstration of the route in multi-gram to kilogram scales within the patent examples, indicating a clear path to commercial scale-up of complex agrochemical intermediates without the need for specialized equipment beyond standard stainless steel reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved Topramezone synthesis. These answers are derived directly from the technical disclosures in patent CN108218851B and are intended to clarify the feasibility and advantages of adopting this route for industrial production. Understanding these details is essential for stakeholders evaluating the transition from legacy processes to this optimized methodology.
Q: What is the primary advantage of the CN108218851B synthesis route compared to prior art?
A: The primary advantage is the significant improvement in total yield, reaching 33.7% under optimized conditions, compared to approximately 30.2% in previous methods. Additionally, this route eliminates the need for complex column chromatography purification, making it far more suitable for industrial scale-up.
Q: How does the new oxidation method improve process safety and cost?
A: The method utilizes hydrogen peroxide in glacial acetic acid for the oxidation of the methylthio group to the sulfone. This replaces harsher or more expensive oxidants, operates at mild temperatures (10-50°C), and simplifies the workup procedure, thereby reducing waste treatment costs and enhancing operational safety.
Q: Is this synthesis route scalable for commercial production of herbicide intermediates?
A: Yes, the route is designed with industrial applicability in mind. It uses cheap and easily obtainable raw materials like 3-nitro-o-xylene and avoids difficult-to-implement steps such as low-temperature column chromatography, ensuring robust supply chain continuity and easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topramezone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive agrochemical market. Our team of expert chemists has thoroughly analyzed the CN108218851B patent and possesses the technical capability to implement this optimized oxidation strategy immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Topramezone intermediate meets the highest industry standards. We understand that in the herbicide sector, purity is not just a specification but a prerequisite for biological efficacy and regulatory approval.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of high-quality Topramezone intermediates that will strengthen your market position and drive your business forward.
