Scalable Synthesis of Topramezone Intermediates: A Technical Breakthrough for Global Agrochemical Supply Chains
Scalable Synthesis of Topramezone Intermediates: A Technical Breakthrough for Global Agrochemical Supply Chains
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key active pharmaceutical ingredients (APIs) and agrochemical intermediates. Patent CN112745270A introduces a transformative methodology for preparing critical intermediates of Topramezone, a potent p-hydroxyphenylpyruvate dioxygenase (4-HPPD) inhibitor. This intellectual property outlines a series of novel compounds represented by General Formula A, specifically designed to streamline the manufacturing of 3-(4,5-dihydroisoxazol-3-yl)-2-methyl-4-methylsulfonyl benzoic acid derivatives. By shifting away from traditional noble metal-catalyzed carbonylation, this technology offers a robust alternative that addresses long-standing challenges in purity, safety, and cost-efficiency. For R&D directors and supply chain leaders, understanding the nuances of this pathway is essential for securing a competitive edge in the herbicide market.
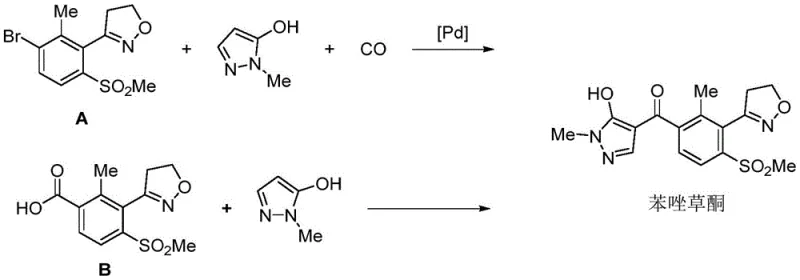
The strategic value of this patent lies in its ability to bypass the complex and hazardous steps associated with legacy synthesis routes. Historically, the construction of the isoxazole ring and the installation of the methylsulfonyl group have been bottlenecks, often requiring harsh reagents or expensive catalysts. The disclosed invention leverages a cyano-containing precursor, 2-methyl-6-methylthiobenzonitrile, as a versatile building block. Through a sequence of chloromethylation or formylation, hydrolysis, hydroxylamination, diazotization halogenation, dipolar cycloaddition, and oxidation, the process achieves the target molecular architecture with remarkable precision. This comprehensive approach not only enhances the chemical integrity of the final product but also aligns with modern green chemistry principles by minimizing toxic waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Topramezone intermediates, such as those reported by BASF and Nippon Kayaku, rely heavily on palladium-catalyzed carbonylation or multi-step sequences involving nitro-reduction. These conventional methods present significant drawbacks for large-scale manufacturing. For instance, the use of palladium and platinum catalysts necessitates stringent removal processes to meet residual metal specifications, adding substantial downstream processing costs. Furthermore, routes involving the reduction of nitro groups pose a risk of N-O bond cleavage within the sensitive isoxazole ring, leading to difficult-to-remove impurities that compromise product quality. The reliance on high-pressure equipment for carbon monoxide insertion also introduces severe safety liabilities and capital expenditure requirements that smaller facilities cannot easily accommodate.
The Novel Approach
In stark contrast, the novel approach detailed in CN112745270A utilizes a metal-free strategy for ring construction and functional group interconversion. By employing a dipolar cycloaddition between a nitrile oxide precursor and ethylene, the method constructs the 4,5-dihydroisoxazole core under mild conditions without the need for transition metals. This shift eliminates the dependency on volatile carbon monoxide gas and expensive noble metal catalysts, fundamentally altering the cost structure of the synthesis. The route is designed to be linear and convergent, avoiding the branching complexities of older methods. The final oxidation step utilizes molecular oxygen or benign oxidants to convert the methylthio group to the required methylsulfonyl moiety, ensuring a cleaner reaction profile that is inherently safer and more scalable for industrial applications.
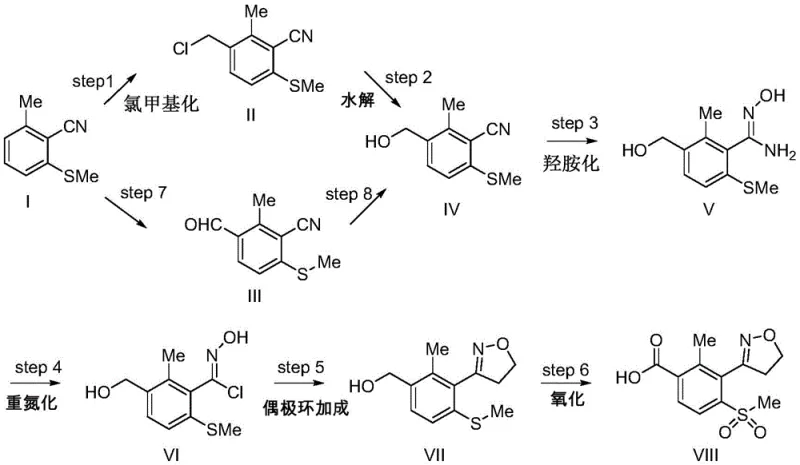
Mechanistic Insights into Dipolar Cycloaddition and Oxidative Functionalization
The cornerstone of this synthetic innovation is the generation of the nitrile oxide species in situ, which serves as the 1,3-dipole for the subsequent cycloaddition. The process begins with the conversion of the amidoxime (Formula V) into the hydroximoyl chloride (Formula VI) using sodium nitrite and hydrochloric acid. This diazotization-halogenation step is critical; unlike traditional aldoxime chlorination which often employs toxic chlorine gas or N-chlorosuccinimide (NCS), this method uses inexpensive and manageable reagents. The resulting hydroximoyl chloride is unstable and spontaneously eliminates hydrogen chloride to form the reactive nitrile oxide. This intermediate immediately undergoes a [3+2] cycloaddition with ethylene gas to form the 4,5-dihydroisoxazole ring (Formula VII). The regioselectivity of this reaction is inherently high due to the electronic nature of the dipole and dipolarophile, ensuring that the isoxazole ring is formed exclusively at the desired position without isomeric byproducts.
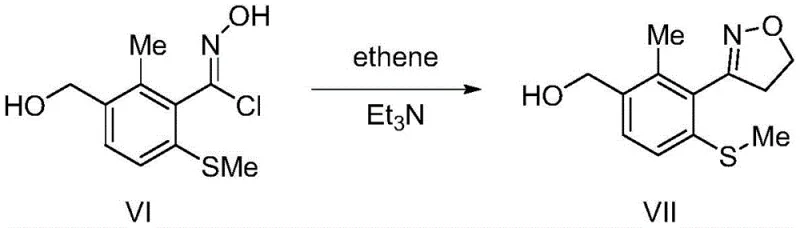
Following the ring closure, the synthesis proceeds to the final oxidation stage, which simultaneously addresses two functional group transformations. The methylthio ether at the para-position must be oxidized to a sulfone, and the benzylic alcohol (or aldehyde precursor) must be converted to a carboxylic acid. The patent discloses the use of molecular oxygen in dioxane or other aprotic solvents as a highly effective oxidant. This aerobic oxidation is mechanistically distinct from stoichiometric oxidations using permanganate or chromium reagents, which generate heavy metal waste. The use of oxygen ensures that the only byproduct is water, drastically simplifying the workup procedure. This dual-oxidation capability demonstrates a sophisticated understanding of chemoselectivity, allowing for the preservation of the sensitive isoxazole ring while aggressively oxidizing the sulfur and carbon centers to their highest oxidation states required for the final herbicide activity.
How to Synthesize Topramezone Intermediate Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and minimize side reactions. The process starts with the functionalization of the benzene ring, where temperature control during chloromethylation is vital to prevent poly-substitution. Subsequent steps involve standard aqueous workups and solvent exchanges that are compatible with standard glass-lined steel reactors. The dipolar cycloaddition step requires the safe handling of ethylene gas, typically managed by maintaining a slight positive pressure in the reactor headspace. For the final oxidation, efficient gas-liquid mass transfer of oxygen is necessary to drive the reaction to completion without over-oxidizing the heterocyclic ring. Detailed standardized operating procedures for each transformation are essential for technology transfer.
- Perform chloromethylation or formylation on 2-methyl-6-methylthiobenzonitrile (Formula I) to introduce a reactive side chain, yielding Formula II or III.
- Convert the side chain to a hydroxymethyl group (Formula IV) via hydrolysis or reduction, followed by reaction with hydroxylamine to form the amidoxime (Formula V).
- Execute diazotization-halogenation to generate the nitrile oxide precursor (Formula VI), followed by dipolar cycloaddition with ethylene and final oxidation to yield the target acid (Formula VIII).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the decoupling of production from the volatile market prices of precious metals. By eliminating palladium and platinum catalysts, the direct material costs are significantly reduced, and the supply chain is no longer exposed to the geopolitical risks associated with PGM (Platinum Group Metals) sourcing. Furthermore, the avoidance of high-pressure carbon monoxide removes the need for specialized autoclaves and rigorous safety monitoring systems, lowering the barrier to entry for contract manufacturing organizations (CMOs) and expanding the pool of potential suppliers. This diversification of the supplier base enhances supply security and reduces the risk of production stoppages due to equipment maintenance or regulatory inspections.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts represents a direct saving on raw material costs, but the financial benefits extend much deeper into the process economics. Without the need for expensive metal scavengers or activated carbon treatments to remove trace palladium, the downstream purification process is drastically simplified. This reduction in unit operations translates to lower energy consumption, reduced solvent usage, and shorter batch cycle times. Additionally, the use of molecular oxygen as an oxidant replaces costly stoichiometric oxidizing agents, further driving down the variable cost per kilogram of the intermediate. These cumulative efficiencies result in a leaner manufacturing process that is highly resilient to margin compression.
- Enhanced Supply Chain Reliability: The starting material, 2-methyl-6-methylthiobenzonitrile, is a commercially available commodity chemical with a stable supply chain, unlike some specialized esters or brominated precursors used in older routes. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—means that production can be sustained across a wider range of manufacturing sites without requiring bespoke infrastructure. This flexibility allows for multi-site production strategies, mitigating the risk of single-point failures. Moreover, the high selectivity of the reactions minimizes the formation of difficult-to-separate impurities, ensuring consistent batch-to-batch quality and reducing the likelihood of rejected shipments that disrupt inventory planning.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior to prior art. The avoidance of toxic chlorine gas for chlorination and flammable dimethyl disulfide for methylation significantly lowers the hazard profile of the facility. The aerobic oxidation step generates minimal waste, aligning with increasingly stringent global environmental regulations regarding heavy metal discharge and solvent emissions. This 'green' profile facilitates easier permitting for capacity expansion and reduces the long-term liability associated with waste disposal. The process is inherently designed for scale-up, with exotherms that are manageable and reaction kinetics that support large batch sizes, making it ideal for meeting the growing global demand for corn herbicides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this new route improve safety compared to traditional Topramezone synthesis?
A: The novel process eliminates the use of highly toxic chlorine gas for aldoxime chlorination and avoids flammable dimethyl disulfide used in traditional diazotization methylation. Additionally, it removes the need for high-pressure carbon monoxide and expensive noble metal catalysts like palladium, significantly reducing operational hazards.
Q: What are the key impurities controlled in this synthesis?
A: The route avoids the reduction of nitro groups which can lead to N-O bond cleavage in the isoxazole ring, a common side reaction in older methods. The high selectivity of the dipolar cycloaddition and the mild oxidation conditions ensure a cleaner impurity profile, minimizing the formation of over-oxidized byproducts or ring-opened species.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the reaction conditions are mild and controllable with good selectivity at each step. The avoidance of column chromatography for purification and the use of readily available reagents like ethylene and oxygen make the entire process route easier to realize industrialization compared to prior art requiring complex separation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topramezone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the synthetic pathway described in CN112745270A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand the critical nuances of handling gaseous reagents like ethylene and managing aerobic oxidations safely at scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, ensuring that every batch of Topramezone intermediate meets stringent purity specifications required by global agrochemical registrars. We are committed to delivering high-quality intermediates that empower your formulation teams to develop next-generation herbicide products.
We invite you to collaborate with us to optimize your supply chain for Topramezone and related 4-HPPD inhibitor herbicides. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By leveraging our optimized version of this metal-free synthesis, we can help you achieve significant reductions in total landed cost while ensuring uninterrupted supply continuity. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us be your strategic partner in navigating the complexities of agrochemical intermediate manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
