Transforming Cyclohexane By-Products into High-Value 2-Aminocyclohexanol for Global Pharmaceutical Supply Chains
The global fine chemical industry is currently witnessing a paradigm shift towards circular economy models, where waste streams from bulk chemical processes are repurposed into high-value specialty intermediates. A prime example of this technological evolution is detailed in Chinese Patent CN111039801A, which discloses a highly efficient utilization method for light oil, a notorious by-product of cyclohexane oxidation. Traditionally regarded as a low-value fuel source due to its complex composition and difficult separation profile, this light oil contains significant amounts of cyclohexene oxide, a reactive epoxide capable of undergoing nucleophilic ring-opening reactions. The patent outlines a robust synthetic pathway that converts this challenging feedstock directly into 2-aminocyclohexanol, a versatile building block extensively used in the synthesis of pharmaceuticals, agrochemicals, and epoxy resin curing agents. By leveraging the inherent reactivity of the epoxide group within the waste matrix, this innovation not only mitigates environmental burdens associated with hazardous waste disposal but also creates a new, cost-effective supply channel for critical nitrogen-containing heterocycles. For R&D directors and procurement strategists, understanding this pathway is essential for securing long-term supply chain resilience and achieving substantial cost reductions in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the management of light oil by-products from cyclohexane oxidation plants has been a significant economic and logistical burden for chemical manufacturers. The conventional approach involved treating this light oil merely as a low-grade fuel for internal combustion or boiler systems, effectively incinerating valuable chemical potential for minimal energy return. Alternative attempts to valorize this stream often focused on separating pure cyclohexene oxide for downstream conversion into adipic acid or other bulk chemicals; however, these methods were plagued by severe technical hurdles. The boiling points of key components within the light oil, such as n-amyl alcohol, cyclohexane, and cyclohexene oxide, are remarkably close, rendering standard fractional distillation inefficient and energy-intensive. Furthermore, the market for traditional derivatives like adipic acid has become saturated, leading to diminished economic margins that fail to justify the high capital expenditure required for complex separation units. Consequently, many facilities were forced to operate with suboptimal resource utilization, relying on external suppliers for high-purity aminocyclohexanol precursors at premium prices, thereby inflating the overall cost structure of their final drug substances.
The Novel Approach
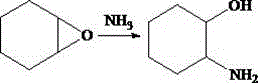
The methodology presented in patent CN111039801A represents a breakthrough in process intensification by bypassing the need for ultra-high purity separation of the starting material. Instead of attempting to isolate pure cyclohexene oxide, the novel approach utilizes the crude light oil directly in an ammonolysis reaction. By reacting the light oil with liquid ammonia or ammonia gas at controlled mass ratios ranging from 100:4 to 100:136, the process selectively targets the reactive epoxide functionality while leaving inert hydrocarbons largely unaffected. This direct conversion strategy operates under relatively mild thermal conditions, specifically between 90°C and 150°C, for short durations of 0.5 to 2 hours. The brilliance of this approach lies in its ability to transform a complex, multi-component mixture into a single, high-value target molecule through selective chemical transformation rather than physical separation. This significantly simplifies the downstream processing train, as the subsequent purification steps only need to separate the desired 2-aminocyclohexanol from unreacted hydrocarbons and minor by-products, rather than resolving azeotropic mixtures of similar boiling points. For a reliable pharmaceutical intermediate supplier, this translates to a streamlined production workflow that enhances throughput and minimizes energy consumption per kilogram of product.
Mechanistic Insights into Ammonolysis of Cyclohexene Oxide
The core chemical transformation driving this process is the nucleophilic ring-opening of the strained three-membered epoxide ring by ammonia. In the reaction medium, ammonia acts as a potent nucleophile, attacking the electrophilic carbon atoms of the cyclohexene oxide ring. Due to the steric environment and the electronic nature of the unsubstituted epoxide, the attack can occur at either carbon, but the reaction conditions favor the formation of the trans-2-aminocyclohexanol isomer through an SN2-like mechanism. The strain energy inherent in the epoxide ring provides the thermodynamic driving force for the reaction, allowing it to proceed efficiently even without the aid of aggressive Lewis acid catalysts which might introduce metallic impurities detrimental to pharmaceutical applications. The absence of transition metal catalysts is a critical design feature, as it eliminates the need for costly and time-consuming heavy metal scavenging steps during purification, thereby ensuring the final product maintains a clean impurity profile suitable for sensitive biological applications.
Furthermore, the process incorporates sophisticated impurity control mechanisms through precise thermal management and pressure regulation. During the reaction phase, maintaining the temperature between 110°C and 150°C optimizes the kinetic balance between conversion rate and selectivity, preventing the formation of polymeric by-products or over-alkylated amines that could complicate purification. Following the reaction, the system is cooled to 60-80°C, a critical step that facilitates the recovery of excess ammonia. This recovery is not merely an economic measure but also a safety and purity protocol, ensuring that no residual basic species remain to catalyze degradation of the product during storage. The subsequent vacuum rectification under a nitrogen protective atmosphere serves as the final barrier against contamination, effectively stripping away low-boiling hydrocarbons inherited from the light oil feedstock while preventing oxidative degradation of the amino-alcohol product. This rigorous control over the reaction environment ensures that the resulting 2-aminocyclohexanol meets the stringent quality standards demanded by global regulatory bodies for use in active pharmaceutical ingredients.
How to Synthesize 2-Aminocyclohexanol Efficiently
The synthesis protocol derived from this patent offers a clear, scalable roadmap for producing high-purity 2-aminocyclohexanol from inexpensive feedstocks. The procedure begins with the charging of cyclohexane oxidation light oil into a pressure-resistant reactor, followed by the introduction of ammonia to achieve the specified stoichiometric excess. The reaction is then driven to completion under autogenous pressure, after which the unreacted ammonia is flashed off and recycled, demonstrating the process's commitment to atom economy and waste minimization. The crude reaction mixture is then subjected to a sequential distillation process, first removing volatile lights and then isolating the target product under reduced pressure to avoid thermal decomposition. This operational simplicity makes the technology highly attractive for commercial scale-up of complex pharmaceutical intermediates, as it relies on standard unit operations found in most fine chemical manufacturing facilities.
- React cyclohexane oxidation light oil with liquid or gaseous ammonia at a mass ratio of 100: 4-136 within a temperature range of 90-150°C for 0.5 to 2 hours.
- Cool the reaction mixture to 60-80°C and depressurize to recover excess ammonia gas for recycling back into the system.
- Separate low-boiling point components from the crude product, followed by vacuum rectification under nitrogen protection to isolate pure 2-aminocyclohexanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers profound strategic advantages that extend beyond simple unit price reductions. By shifting the raw material base from purified cyclohexene oxide to light oil by-products, manufacturers can decouple their production costs from the volatile pricing of refined chemical feedstocks. This feedstock flexibility acts as a natural hedge against market fluctuations, ensuring more stable pricing structures for long-term contracts. Moreover, the elimination of complex pre-separation steps reduces the capital intensity of the production line, lowering the barrier to entry for new capacity and increasing the overall resilience of the supply network against disruptions. The ability to source raw materials from the abundant waste streams of major cyclohexane producers also geographically diversifies the supply base, reducing reliance on single-source vendors and enhancing supply security for critical drug intermediates.
- Cost Reduction in Manufacturing: The most immediate impact of this process is the dramatic lowering of raw material costs, as light oil is essentially a waste product with negligible market value compared to pure chemical reagents. By utilizing this low-cost feedstock, manufacturers eliminate the margin layers associated with intermediate purification and distribution, passing significant savings down the value chain. Additionally, the process operates without expensive transition metal catalysts, removing the financial burden of catalyst procurement, regeneration, and the specialized waste treatment required for heavy metal disposal. The energy efficiency of the process, driven by shorter reaction times and moderate temperatures, further contributes to a leaner cost structure, allowing for competitive pricing in high-volume markets without sacrificing profit margins.
- Enhanced Supply Chain Reliability: Integrating this synthesis route into the supply chain significantly mitigates the risk of raw material shortages that often plague the fine chemical sector. Since light oil is a continuous by-product of the massive scale cyclohexane oxidation industry, its availability is inherently linked to the robust production of nylon precursors, ensuring a steady and predictable flow of feedstock. This abundance allows suppliers to maintain higher inventory levels of finished 2-aminocyclohexanol, drastically reducing lead times for customers facing urgent production needs. Furthermore, the simplified process flow reduces the number of potential failure points in the manufacturing line, resulting in higher plant uptime and more consistent delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this technology aligns perfectly with modern green chemistry principles and increasingly strict emissions standards. By converting a hazardous waste stream into a useful product, the process reduces the overall environmental footprint of the chemical plant, potentially qualifying for green manufacturing incentives and improving the corporate sustainability profile. The closed-loop recovery of ammonia minimizes atmospheric emissions, while the absence of heavy metals simplifies wastewater treatment protocols, ensuring compliance with rigorous international environmental regulations. This ease of compliance facilitates faster regulatory approvals for new manufacturing sites and reduces the administrative burden on EHS teams, making it an ideal candidate for rapid capacity expansion in response to growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-aminocyclohexanol via this novel ammonolysis pathway. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis routes or for procurement specialists negotiating supply agreements.
Q: What is the primary advantage of using light oil by-products for 2-aminocyclohexanol production?
A: The primary advantage lies in the drastic reduction of raw material costs by utilizing a low-value waste stream (light oil) from cyclohexane oxidation instead of purchasing pure cyclohexene oxide, while simultaneously solving an environmental disposal issue.
Q: How does this process ensure high purity suitable for pharmaceutical applications?
A: The process employs a two-stage purification strategy: initial removal of low-boiling impurities followed by rigorous vacuum rectification under a nitrogen protective atmosphere, which prevents oxidation and ensures the final product meets stringent purity specifications required for API intermediates.
Q: Is this ammonolysis method scalable for industrial manufacturing?
A: Yes, the method operates under relatively mild conditions (90-150°C) and short reaction times (0.5-2h) without requiring complex transition metal catalysts, making it highly amenable to large-scale commercial production with simplified equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminocyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of converting waste streams into high-value pharmaceutical building blocks, and we have positioned ourselves at the forefront of this technological advancement. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 2-aminocyclohexanol that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the ammonolysis process described in CN111039801A allows us to offer a sustainable, cost-competitive supply solution that meets the evolving needs of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through innovative chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced production capabilities can support your long-term strategic goals and ensure a uninterrupted supply of critical intermediates.
