Advanced Catalytic Synthesis of Mixed-Substituted Dialkylphosphinic Acids for High-Performance Polymer Flame Retardancy
The chemical industry is constantly seeking more efficient and environmentally benign pathways to produce high-performance additives, particularly for the demanding sector of flame-retardant polymers. Patent CN102164930A introduces a groundbreaking methodology for the production of mixed-substituted dialkylphosphinic acids, their esters, and salts, addressing critical limitations in current manufacturing technologies. This invention delineates a sophisticated two-step catalytic process that significantly enhances space-time yields while eliminating the reliance on interfering halogen compounds, which have long been a bottleneck in achieving high purity and regulatory compliance. By leveraging specific transition metal catalysts in the initial stage followed by radical-mediated addition, the process allows for the precise construction of complex organophosphorus architectures. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective supply chains for critical polymer additives. The ability to target specific esterification and salt formation post-synthesis further underscores the versatility of this platform, enabling tailored solutions for diverse thermoplastic matrices ranging from polyesters to polyamides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of dialkylphosphinic acids has been plagued by significant inefficiencies and environmental concerns that hinder large-scale adoption. Traditional routes often rely on harsh reaction conditions that necessitate the use of interfering halogen compounds as reactants, leading to complex purification challenges and the generation of hazardous waste streams. Furthermore, existing methods frequently struggle to achieve high space-time yields, resulting in prolonged batch times and increased capital expenditure for reactor capacity. The difficulty in isolating final products without extensive downstream processing, such as multiple distillation or crystallization steps, adds substantial operational costs. Additionally, conventional techniques often lack the selectivity required to produce mixed-substituted derivatives with the precision needed for high-performance flame retardancy, often yielding mixtures that compromise the thermal stability of the final polymer. These limitations create a fragile supply chain where cost volatility and regulatory pressure regarding halogenated byproducts pose continuous risks to manufacturers of electronic components and automotive parts.
The Novel Approach
The methodology outlined in CN102164930A offers a transformative solution by decoupling the synthesis into two distinct, highly controlled catalytic stages that maximize efficiency and product quality. In the first stage, a phosphinic acid source reacts with olefins under the influence of a specialized transition metal catalyst system, facilitating a clean addition reaction to form alkylphosphonous acid intermediates. This is followed by a second stage where these intermediates undergo further reaction with olefins in the presence of peroxide or azo catalysts, effectively building the desired mixed-substituted dialkylphosphinic acid derivative. This sequential approach allows for exceptional control over the substitution pattern, ensuring high purity without the need for interfering halogens. The process is designed for high space-time yields, meaning that manufacturers can produce significantly more product per unit of time and reactor volume. Moreover, the flexibility to perform targeted esterification or salt formation in subsequent steps ensures that the final output can be perfectly matched to the specific rheological and thermal requirements of the end-user's polymer compounding process.
Mechanistic Insights into Transition Metal and Radical Catalyzed Hydrophosphinylation
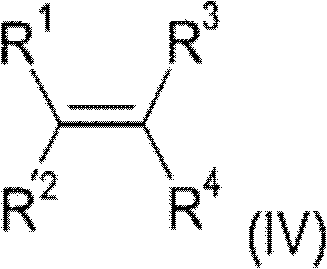
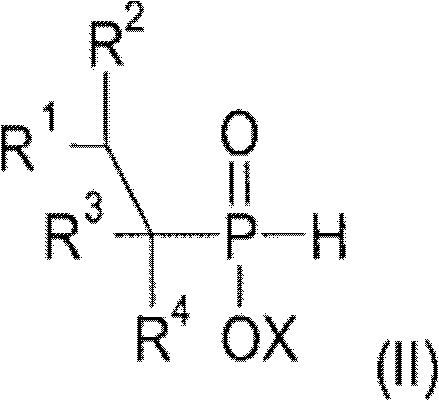
The core innovation of this technology lies in the precise orchestration of two different catalytic mechanisms to achieve selective C-P bond formation. The initial step relies on a transition metal catalyst, typically derived from Group 8 to 10 metals such as Palladium, Nickel, Rhodium, or Platinum, often coordinated with specific phosphine ligands. This catalyst activates the phosphinic acid source, allowing for the regioselective addition across the double bond of the olefin. The choice of ligand is critical, as bidentate phosphines or specific monodentate variants can drastically influence the turnover number and stability of the active catalytic species. This step generates an alkylphosphonous acid intermediate, which serves as the foundational building block for the subsequent modification. The ability to utilize a wide range of olefins, from simple ethylene and propylene to functionalized styrenes and allyl derivatives, demonstrates the robustness of this catalytic system in accommodating diverse steric and electronic environments.
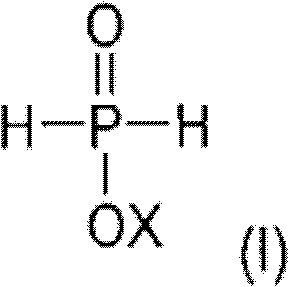
Following the formation of the intermediate, the process transitions to a radical-mediated mechanism for the second alkylation. Here, catalysts such as peroxides (e.g., hydrogen peroxide, persulfates) or azo compounds (e.g., AIBN, Vazo initiators) generate free radicals that initiate the addition of a second olefin molecule to the phosphorus center. This radical pathway complements the transition metal step by allowing for the introduction of different alkyl groups, thereby creating the 'mixed-substituted' nature of the final product. The synergy between these two catalytic regimes ensures that the final dialkylphosphinic acid derivative possesses the exact structural attributes required for optimal flame retardant performance. This dual-catalyst strategy avoids the scrambling of substituents often seen in single-step processes, resulting in a product with superior thermal stability and compatibility within polymer matrices.
Controlling impurities is paramount in the production of flame retardant intermediates, as trace metals or unreacted precursors can degrade the mechanical properties of the host polymer. The patent describes sophisticated workup procedures involving the use of metal scavengers and specific adsorption materials to remove residual transition metals from the first catalytic step. Materials such as functionalized silicas, ion exchange resins, or specific metal oxides are employed to trap catalyst residues, ensuring the final product meets stringent purity specifications. Furthermore, the process allows for the direct conversion of the acid into salts (such as Aluminum, Zinc, or Titanium salts) or esters without isolating the free acid, minimizing exposure to moisture and reducing the potential for hydrolysis-related impurities. This integrated approach to purification and derivatization ensures that the final additive contributes to the polymer's fire resistance without compromising its color, clarity, or mechanical integrity.
How to Synthesize Mixed-Substituted Dialkylphosphinic Acids Efficiently
Implementing this synthesis route requires careful attention to reaction parameters such as temperature, pressure, and catalyst loading to ensure optimal conversion and selectivity. The process is adaptable to various reactor configurations, including stirred tanks and loop reactors, and can be operated in liquid, gas, or supercritical phases depending on the volatility of the olefin feedstock. Solvent selection is also flexible, ranging from water and alcohols to hydrocarbons and ethers, allowing manufacturers to optimize for cost and environmental impact. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for scaling this technology from laboratory benchtop to commercial production volumes.
- React a phosphinic acid source with olefins in the presence of a transition metal catalyst (Catalyst A) to form alkylphosphonous acid intermediates.
- Subject the resulting alkylphosphonous acid to a second addition reaction with olefins using peroxide or azo catalysts (Catalyst B) to yield mixed-substituted derivatives.
- Convert the final acid derivatives into metal salts (e.g., Aluminum, Zinc) or esters through neutralization or esterification for specific polymer applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond mere technical performance. The elimination of halogenated reactants simplifies regulatory compliance, reducing the administrative burden and risk associated with shipping and handling hazardous materials. This inherently safer chemistry translates into lower insurance costs and fewer logistical bottlenecks, ensuring a more resilient supply chain for critical flame retardant intermediates. Furthermore, the high space-time yield of the process means that production capacity can be significantly increased without proportional increases in capital investment, allowing suppliers to respond more agilely to market demand fluctuations. The ability to produce a wide variety of derivatives from a common set of feedstocks also reduces inventory complexity, enabling a more streamlined and cost-efficient operation.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by eliminating expensive and hazardous halogenated reagents, replacing them with readily available olefins and efficient catalyst systems. The high selectivity of the reaction minimizes the formation of byproducts, thereby reducing the load on downstream purification units and lowering waste disposal costs. Additionally, the ability to recycle solvents and catalyst supports further drives down the variable cost per kilogram of the final product. By avoiding complex multi-step protection and deprotection strategies common in older methods, the overall manufacturing footprint is reduced, leading to significant operational expenditure optimization.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is significantly more secure due to the reliance on commodity chemicals like ethylene, propylene, and standard transition metal salts, which are available from a broad global supplier base. This diversification of supply reduces the risk of single-source dependency that often plagues specialty chemical manufacturing. The robustness of the catalyst systems also implies longer campaign lengths and fewer unplanned shutdowns for catalyst regeneration or replacement. Consequently, manufacturers can offer more reliable lead times and consistent product availability, which is critical for just-in-time manufacturing models in the automotive and electronics industries.
- Scalability and Environmental Compliance: The technology is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to full-scale industrial reactors. The absence of halogenated waste streams aligns perfectly with increasingly stringent global environmental regulations, such as REACH and TSCA, future-proofing the production facility against regulatory tightening. The process generates minimal aqueous or organic waste, and the use of water as a potential solvent in certain steps highlights its green chemistry credentials. This environmental profile not only mitigates compliance risks but also enhances the brand value of the end-products by supporting sustainability goals in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on catalyst selection, product scope, and downstream processing. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating these intermediates into their existing formulation portfolios.
Q: How does this patent process improve upon traditional halogenated flame retardant synthesis?
A: The process described in CN102164930A eliminates the need for interfering halogen compounds as reactants, offering a cleaner, more environmentally compliant route to high-purity phosphinic acids compared to traditional halogenated methods.
Q: What types of catalysts are required for the two-step synthesis?
A: The first step utilizes transition metal catalysts (such as Palladium, Nickel, or Rhodium complexes) for hydrophosphinylation, while the second step employs radical initiators like peroxides or azo compounds for the subsequent alkylation.
Q: Can these intermediates be directly used in polyamide and polyester compounding?
A: Yes, the resulting mixed-substituted dialkylphosphinic acid salts (particularly Aluminum and Zinc salts) are specifically designed for compounding into thermoplastics like PBT, PET, and PA66 to achieve UL-94 V-0 ratings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dialkylphosphinic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity organophosphorus intermediates in the development of next-generation flame-retardant materials. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate complex catalytic pathways like CN102164930A into robust commercial realities. Our facilities are equipped to handle diverse synthetic challenges, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of dialkylphosphinic acid or salt meets the exacting standards required for high-performance engineering plastics. Our commitment to quality assurance guarantees that your supply chain remains uninterrupted and compliant with global safety norms.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Whether you require custom aluminum or zinc salts for polyamide compounding or specialized esters for polyester modification, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and innovation in your product portfolio.
