Advanced Synthesis of Monohydroxy-Functionalized Dialkylphosphinic Acids for Commercial Polymer Applications
The chemical industry continuously seeks innovative pathways to synthesize high-performance additives that meet stringent safety and environmental standards without compromising on economic feasibility. Patent CN102171228A introduces a groundbreaking method for the preparation of monohydroxy-functionalized dialkylphosphinic acids, dialkylphosphinates, and their salts, utilizing vinyl esters of carboxylic acids as key reagents. This technology addresses the long-standing challenge of producing these valuable intermediates with high space-time yields while avoiding the use of interfering halogen compounds that often complicate downstream purification. By leveraging a sophisticated multi-step catalytic sequence, this process enables the targeted synthesis of specific phosphorus-containing structures that are critical for advanced material applications. The ability to easily isolate the final product under specific reaction conditions represents a significant leap forward in process chemistry, offering manufacturers a robust route to high-purity materials essential for next-generation polymer formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalized phosphinic acids have historically been plagued by significant inefficiencies and environmental drawbacks that hinder their widespread industrial adoption. Many conventional methods rely heavily on halogenated reactants which introduce toxic byproducts and require complex, energy-intensive separation steps to ensure the final product meets purity specifications. The presence of residual halogens can severely degrade the thermal stability of the resulting polymers, limiting their utility in high-performance applications such as automotive components or electronic housings. Furthermore, older processes often suffer from low space-time yields, meaning that reactors must be operated for extended periods to achieve modest conversion rates, thereby driving up capital expenditure and operational costs. The difficulty in selectively functionalizing the phosphorus center without affecting other sensitive groups in the molecule has also been a persistent bottleneck, leading to broad impurity profiles that are difficult to characterize and control.
The Novel Approach
The patented methodology overcomes these historical barriers by employing a clean, three-stage catalytic protocol that utilizes readily available olefins and vinyl esters to construct the desired molecular architecture with precision. This approach eliminates the need for hazardous halogen sources, significantly reducing the environmental footprint and simplifying the waste management protocols required for commercial operation. By using vinyl esters of carboxylic acids in the second step, the process introduces the necessary functional groups through a radical mechanism that is both highly selective and efficient. The final hydrolysis step allows for the gentle unmasking of the hydroxyl functionality under controlled acidic or basic conditions, ensuring that the delicate phosphorus-carbon bonds remain intact. This strategic design not only enhances the overall yield but also facilitates the easy isolation of the final product, making it an economically attractive option for large-scale manufacturing.
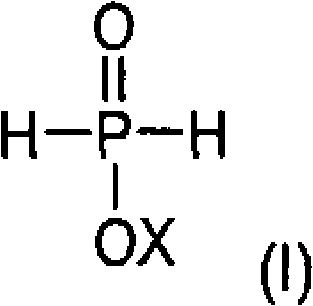
This novel pathway provides a versatile platform for generating a wide array of derivatives by simply varying the olefin or the vinyl ester components, allowing for fine-tuning of the physical and chemical properties of the final additive. The compatibility of this method with various transition metal catalysts ensures that processors can optimize the reaction kinetics to suit their specific equipment configurations and throughput requirements. Moreover, the absence of corrosive halogen byproducts extends the lifespan of reactor vessels and piping, leading to substantial long-term savings in maintenance and replacement costs. For procurement teams, this translates into a more stable supply chain with reduced risk of production shutdowns due to equipment failure or regulatory compliance issues related to hazardous waste disposal.
Mechanistic Insights into Transition Metal Catalyzed Hydrophosphinylation
The core of this synthesis lies in the initial hydrophosphinylation reaction where a phosphinic acid source reacts with an olefin in the presence of a transition metal catalyst to form alkylphosphonous acid derivatives. This step typically employs catalysts from the seventh and eighth subgroups, such as rhodium, nickel, palladium, or platinum, often coordinated with specialized phosphine ligands to enhance activity and selectivity. The mechanism involves the oxidative addition of the phosphorus-hydrogen bond to the metal center, followed by the insertion of the olefin into the metal-phosphorus bond, and finally reductive elimination to release the alkylphosphonous acid product. Understanding this catalytic cycle is crucial for R&D directors aiming to optimize reaction conditions, as factors such as ligand sterics and electronic properties can dramatically influence the regioselectivity of the olefin addition. Careful control of temperature and pressure during this stage ensures that the reaction proceeds with minimal formation of undesired isomers or oligomeric byproducts.
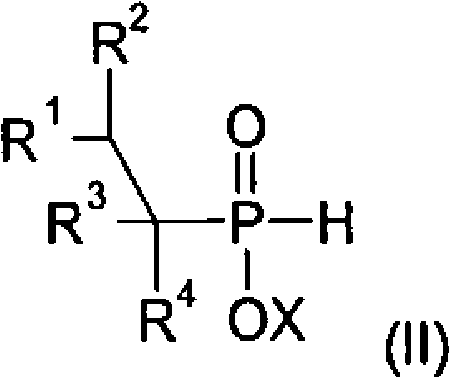
Following the formation of the alkylphosphonous acid intermediate, the process advances to a radical addition step where the intermediate reacts with a vinyl ester of a carboxylic acid. This transformation is mediated by peroxide-forming compounds or azo initiators which generate free radicals capable of abstracting hydrogen atoms from the phosphorus center. The resulting phosphorus-centered radical then adds across the double bond of the vinyl ester, creating a new carbon-phosphorus bond and installing the ester functionality. This radical mechanism is particularly advantageous because it tolerates a wide range of functional groups on both the phosphorus substrate and the vinyl ester, allowing for significant structural diversity in the final product. The subsequent hydrolysis of the ester group under acidic or basic conditions reveals the terminal hydroxyl group, completing the synthesis of the monohydroxy-functionalized dialkylphosphinic acid. Rigorous control over the hydrolysis conditions is essential to prevent degradation of the phosphinic acid backbone while ensuring complete conversion of the ester moiety.
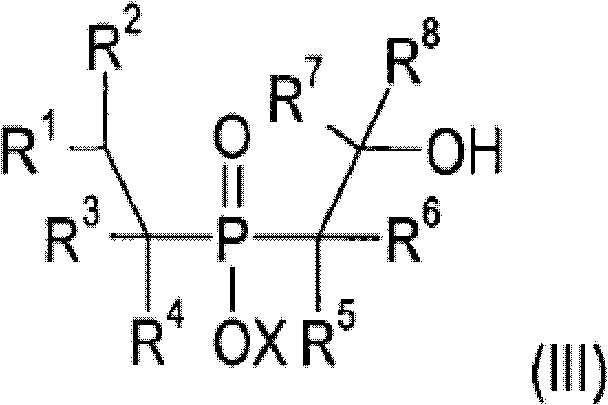
Impurity control is maintained throughout the process through the strategic use of metal scavengers and purification techniques such as distillation or crystallization. Transition metal residues, which can act as pro-degradants in polymer applications, are effectively removed using adsorbents like activated carbon or specialized ion-exchange resins. The process design allows for the recycling of solvents and unreacted starting materials, further enhancing the atom economy and sustainability profile of the manufacturing operation. For quality assurance teams, the well-defined reaction pathway ensures consistent batch-to-batch reproducibility, which is critical for maintaining the mechanical and fire-resistant properties of the downstream polymer compounds. The ability to produce these materials with high purity and low metal content makes them ideal for use in sensitive applications such as electrical connectors and automotive under-the-hood components where reliability is paramount.
How to Synthesize Monohydroxy-Functionalized Dialkylphosphinic Acids Efficiently
The standardized synthesis of these high-value intermediates requires precise adherence to the three-step catalytic protocol outlined in the patent documentation to ensure optimal yield and purity. Operators must carefully monitor reaction parameters such as temperature, pressure, and catalyst loading at each stage to maintain the integrity of the phosphorus-carbon bonds while driving the conversion to completion. The detailed standardized synthesis steps see below guide provides a comprehensive overview of the necessary equipment setup, reagent preparation, and workup procedures required for successful implementation. Adhering to these guidelines ensures that the final product meets the stringent specifications required for use as a flame retardant in engineering thermoplastics.
- React a phosphinic acid source with an olefin in the presence of a transition metal catalyst to form alkylphosphonous acid derivatives.
- React the resulting alkylphosphonous acid with a vinyl ester of a carboxylic acid using a peroxide or azo catalyst to generate monofunctionalized derivatives.
- Hydrolyze the monofunctionalized derivatives using an acid or base catalyst to obtain the final monohydroxy-functionalized dialkylphosphinic acid products.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly address the key pain points faced by procurement managers and supply chain leaders in the specialty chemicals sector. The elimination of halogenated reactants not only reduces the cost associated with hazardous waste disposal but also mitigates the regulatory risks associated with handling toxic materials, leading to a more resilient and compliant supply chain. The use of commodity chemicals such as ethylene and vinyl acetate as starting materials ensures a stable and cost-effective raw material base that is less susceptible to market volatility compared to exotic precursors. Furthermore, the high space-time yields achieved through this catalytic method mean that existing production facilities can be utilized more efficiently, potentially deferring the need for costly capacity expansions. These factors combine to create a compelling economic case for adopting this technology, offering significant cost reduction in polymer additive manufacturing without sacrificing product performance.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating expensive and hazardous halogenated reagents, thereby reducing raw material costs and waste treatment expenses significantly. The high efficiency of the transition metal catalysts allows for lower catalyst loadings and shorter reaction times, which translates directly into reduced energy consumption and increased throughput per reactor volume. Additionally, the simplified purification steps reduce the need for complex separation equipment and solvents, lowering both capital investment and operating expenditures. By streamlining the synthesis pathway, manufacturers can achieve a leaner production model that maximizes resource utilization and minimizes overhead costs associated with environmental compliance and safety monitoring.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the process relies on widely available bulk chemicals like olefins and vinyl esters which are produced on a massive global scale with established logistics networks. This reduces the dependency on niche suppliers and minimizes the risk of supply disruptions caused by geopolitical issues or production outages at single-source vendors. The robustness of the catalytic system also means that production can be easily scaled up or down to match market demand without requiring significant retooling or process requalification. For supply chain heads, this flexibility ensures that customer orders can be fulfilled consistently and on time, strengthening relationships with downstream polymer compounders and end-users who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: The technology is inherently scalable, having been designed for operation in standard industrial reactors such as stirred tanks and loop reactors that are common in fine chemical plants. This compatibility allows for seamless technology transfer from pilot scale to full commercial production, accelerating time-to-market for new product launches. Environmentally, the process aligns with green chemistry principles by avoiding toxic halogens and enabling solvent recycling, which helps companies meet increasingly stringent corporate sustainability goals and regulatory requirements. The reduced environmental footprint enhances the brand reputation of manufacturers and opens up opportunities in markets where eco-friendly credentials are a key purchasing criterion for OEMs and consumers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology for industrial stakeholders. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and application potential. Understanding these details is essential for making informed decisions about integrating these materials into your product portfolio.
Q: How does this process improve upon conventional halogenated flame retardant synthesis?
A: This patented method eliminates the need for interfering halogen compounds as reactants, resulting in a cleaner product profile and simplified purification processes that reduce environmental impact and waste treatment costs.
Q: What are the primary industrial applications for these phosphinic acid derivatives?
A: These compounds serve as highly effective flame retardants for thermoplastic and thermosetting polymers, including polyesters like PBT and polyamides like PA66, offering superior thermal stability and mechanical properties.
Q: Is the catalytic system suitable for large-scale commercial production?
A: Yes, the process utilizes standard transition metal catalysts and common reactor types such as stirred tanks and loop reactors, ensuring high space-time yields and straightforward scalability from pilot to industrial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monohydroxy-Functionalized Dialkylphosphinic Acids Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring this sophisticated synthesis route to life on a commercial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of monohydroxy-functionalized dialkylphosphinic acids meets the highest industry standards for flame retardancy and thermal stability. Our commitment to quality ensures that these critical additives perform reliably in your final polymer formulations, protecting your brand reputation and end-user safety.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to this halogen-free technology. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies. Let us help you optimize your supply chain with high-performance additives that drive innovation and sustainability in your polymer applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
