Advanced Green Catalytic Oxidation for Commercial Lansoprazole Manufacturing
Advanced Green Catalytic Oxidation for Commercial Lansoprazole Manufacturing
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of critical proton pump inhibitors, and the synthesis method disclosed in patent CN102180865B represents a significant technological leap forward in this domain. This patent details a novel approach to synthesizing Lansoprazole, a widely prescribed medication for acid-related disorders, by utilizing a composite metal oxide catalyst that fundamentally alters the oxidation landscape of the precursor thioether. Unlike traditional methods that rely on homogeneous catalysts often associated with heavy metal toxicity and complex downstream processing, this innovation employs a heterogeneous system comprising metal oxides and specific aids like molecular sieves. The core breakthrough lies in the catalyst's preparation, which involves high-temperature calcination between 450°C and 800°C, creating a robust structural framework that facilitates selective oxidation using hydrogen peroxide. This method not only addresses the pressing environmental concerns associated with vanadium and cesium-based catalysts but also offers a streamlined operational protocol that is highly amenable to industrial scale-up. By shifting the paradigm towards solid-state catalysis, manufacturers can achieve higher purity profiles while drastically simplifying the isolation of the final active pharmaceutical ingredient.
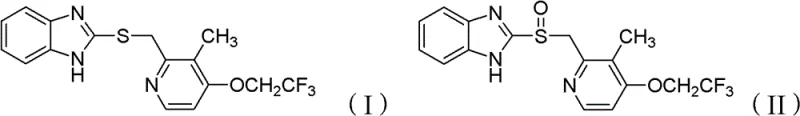
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzimidazole sulfoxides like Lansoprazole has been plagued by significant technical and regulatory hurdles associated with the choice of oxidants and catalysts. Prior art, such as that described in patent CN1381443, frequently relied on organic compounds of vanadium, titanium, or copper, which, while effective in driving the reaction, introduced severe toxicity risks to both operators and the environment. The presence of vanadium residues in the final product necessitates rigorous and costly purification steps, often involving column chromatography or complex chelation processes, which are impractical for multi-ton commercial production. Furthermore, other methods utilizing cesium-based catalysts, as seen in WO02/074766, although improving yield, still depended on halogenated hydrocarbon solvents that pose disposal challenges and regulatory scrutiny under modern green chemistry guidelines. The use of high-concentration hydrogen peroxide in earlier molybdate-catalyzed systems also presented safety hazards regarding thermal runaway and stability, often resulting in suboptimal yields and purity that required extensive recrystallization efforts. These cumulative inefficiencies create a bottleneck for supply chain reliability, as the dependency on hazardous reagents increases lead times and complicates waste management protocols.
The Novel Approach
In stark contrast to these legacy methodologies, the technique outlined in CN102180865B introduces a composite metal oxide catalyst system that effectively decouples high catalytic activity from environmental toxicity. By employing a mixture of metal oxides—such as tungsten, molybdenum, or iron—combined with structural aids like 4A molecular sieves or silica, the process creates a highly active solid surface that promotes the selective transfer of oxygen from hydrogen peroxide to the sulfur atom. This heterogeneous nature allows for the catalyst to be easily separated from the reaction mixture via simple filtration, eliminating the need for energy-intensive distillation or complex extraction procedures typically required to remove soluble metal salts. The operational window is remarkably flexible, functioning efficiently at temperatures ranging from -30°C to 30°C, with optimal results observed between -10°C and 10°C, thereby reducing the energy load associated with cryogenic cooling. Moreover, the ability to recycle the catalyst after washing and drying introduces a circular economy aspect to the manufacturing process, significantly lowering the raw material cost per kilogram of the final API. This approach not only enhances the safety profile of the plant operations by avoiding toxic heavy metals but also ensures a cleaner impurity profile, which is critical for meeting stringent pharmacopeial standards.
Mechanistic Insights into Composite Metal Oxide Catalyzed Oxidation
The efficacy of this synthesis route is rooted in the unique physicochemical properties of the calcined composite metal oxide matrix, which acts as a solid base catalyst with tailored surface acidity and porosity. During the high-temperature calcination step at 450°C to 800°C, the metal oxides and aids undergo a structural reorganization that generates active sites capable of activating the hydrogen peroxide molecule without decomposing it prematurely into water and oxygen. The molecular sieve component, particularly the preferred 4A variety, plays a crucial role in stabilizing the transition state and potentially acting as a shape-selective barrier that prevents the over-oxidation of the sulfoxide to the corresponding sulfone, a common side reaction that plagues proton pump inhibitor synthesis. The mechanism likely involves the adsorption of the thioether substrate onto the catalyst surface, where the activated peroxide species attacks the sulfur center with high regioselectivity. This surface-mediated reaction pathway minimizes the formation of chlorinated by-products that are often observed when halogenated solvents are used in conjunction with acidic catalysts. Furthermore, the basic nature of the composite oxide helps to neutralize any acidic by-products generated during the oxidation, maintaining a stable pH environment that protects the acid-sensitive benzimidazole ring from degradation. This delicate balance of oxidative power and structural stability is what allows the process to achieve yields exceeding 77% in optimized examples, demonstrating a level of control that homogeneous systems struggle to match.
From an impurity control perspective, the use of this solid catalyst system offers distinct advantages in managing the genetic toxicology profile of the drug substance. Traditional methods often leave behind trace amounts of mutagenic metal residues that require specialized scavenging resins to reduce to ppm levels, adding significant cost and complexity to the downstream processing. In this novel method, the physical separation of the catalyst ensures that metal leaching is minimized, and any trace metals present are typically in an oxide form that is less bioavailable and easier to control. The reaction conditions, specifically the use of ethyl acetate as a preferred solvent, further contribute to a cleaner impurity spectrum by avoiding the formation of alkyl halides or other solvent-derived adducts. The selectivity of the catalyst also suppresses the formation of N-oxide impurities, which are structurally similar to the target molecule and difficult to separate by crystallization. By optimizing the mass ratio of the metal oxide to the aid between 1:1 and 1:10, manufacturers can fine-tune the pore size and surface area to maximize the diffusion of reactants while excluding larger impurity precursors. This mechanistic precision translates directly into a more robust manufacturing process that requires fewer purification cycles, thereby improving the overall throughput and consistency of the final Lansoprazole product.
How to Synthesize Lansoprazole Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, reaction, and isolation steps that are designed for scalability and operator safety. The process begins with the preparation of the catalyst, where precise stoichiometric amounts of metal oxides and molecular sieves are ground together and subjected to controlled thermal treatment to activate the catalytic sites. Once the catalyst is ready, it is suspended in a reaction vessel along with the Lansoprazole precursor thioether and a suitable organic solvent, with ethyl acetate being the preferred choice due to its favorable solubility profile and low toxicity. The mixture is then cooled to the specified temperature range, typically around 0°C, to manage the exothermic nature of the oxidation reaction before the controlled addition of 30% hydrogen peroxide. Detailed standardized synthetic steps and specific parameter optimizations for this guide are provided in the section below.
- Prepare the composite metal oxide catalyst by grinding metal oxides (e.g., Tungsten, Molybdenum) with aids (e.g., 4A Molecular Sieve) and calcining at 450-800°C.
- Suspend the precursor thioether and the prepared catalyst in a solvent such as ethyl acetate and cool the mixture to a temperature range of -30°C to 30°C.
- Slowly add 30% hydrogen peroxide solution under stirring, maintain the temperature until reaction completion, then filter and recrystallize to obtain high-purity Lansoprazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers a compelling value proposition centered on cost stability and regulatory compliance. The elimination of toxic vanadium and cesium reagents removes a significant layer of regulatory risk, ensuring that the supply of Lansoprazole intermediates remains uninterrupted by changing environmental laws or workplace safety mandates. The ability to recycle the solid catalyst multiple times without significant loss of activity means that the effective cost of the catalyst per batch is drastically reduced compared to single-use homogeneous systems. Furthermore, the use of common, non-halogenated solvents like ethyl acetate simplifies solvent recovery and recycling infrastructure, leading to substantial operational expenditure savings over the lifecycle of the product. The robustness of the reaction conditions, which tolerate a wide temperature range and do not require inert atmospheres as strictly as some organometallic catalyses, reduces the engineering controls needed in the production facility, thereby lowering capital investment requirements for new capacity. These factors combine to create a supply chain that is not only more cost-effective but also more resilient to external shocks such as raw material shortages or regulatory crackdowns on hazardous chemicals.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the transition from expensive, single-use homogeneous catalysts to a reusable heterogeneous system that eliminates the need for complex metal scavenging steps. By removing the requirement for specialized resins or extensive aqueous workups to strip out heavy metals, the manufacturing process becomes significantly shorter and less resource-intensive. The high selectivity of the catalyst also minimizes the loss of valuable starting material to over-oxidized by-products, directly improving the mass balance and yield of the process. Additionally, the lower energy requirements for temperature control and solvent recovery contribute to a reduced utility bill, further enhancing the margin profile of the manufactured API. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the final pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the use of readily available and non-restricted raw materials such as iron, tungsten, and molecular sieves, which are not subject to the same geopolitical supply constraints as rare earth metals or specialized organometallic complexes. The simplified process flow, characterized by easy filtration and standard crystallization techniques, reduces the dependency on highly specialized equipment or niche contract manufacturing organizations. This accessibility means that production can be scaled up or shifted between facilities with minimal requalification effort, ensuring continuity of supply even in the face of localized disruptions. The reduced hazard profile of the reagents also simplifies logistics and storage requirements, allowing for larger inventory buffers to be held safely on-site to mitigate against demand spikes. Consequently, partners can rely on a more predictable and stable delivery schedule for high-purity Lansoprazole intermediates.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this heterogeneous catalytic system, as the heat transfer and mixing dynamics of solid-liquid suspensions are well-understood and easily modeled for large-scale reactors. The environmental compliance aspect is perhaps the most significant advantage, as the process generates significantly less hazardous waste compared to traditional methods, aligning perfectly with global initiatives to reduce the pharmaceutical industry's carbon footprint. The absence of toxic heavy metals in the effluent stream simplifies wastewater treatment protocols and reduces the cost of environmental remediation. Moreover, the potential for catalyst regeneration means less solid waste is sent to landfills, supporting corporate sustainability goals. This green chemistry profile not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand reputation of the supplier as a responsible partner in the global healthcare value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, derived directly from the experimental data and claims within the patent literature. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating this route for potential adoption or outsourcing. Understanding these nuances is critical for making informed decisions about process validation and technology transfer.
Q: What are the advantages of the composite metal oxide catalyst over traditional vanadium catalysts?
A: Unlike traditional vanadium-based catalysts which are toxic and difficult to remove, the composite metal oxide catalyst described in CN102180865B is non-toxic, easily separated by filtration, and can be recycled, significantly reducing environmental pollution and purification costs.
Q: What represents the optimal reaction temperature for this oxidation process?
A: The patent specifies a broad operating range of -30°C to 30°C, with a preferred embodiment operating between -10°C and 10°C to maximize selectivity for the sulfoxide product while minimizing over-oxidation to the sulfone.
Q: Can the catalyst be reused in subsequent batches?
A: Yes, the solid composite catalyst can be recovered after the reaction by washing with isopropanol, dried, and reused, which enhances the economic feasibility and sustainability of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lansoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is essential for the long-term viability of the pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the composite metal oxide catalysis method described in CN102180865B and possesses the technical capability to implement this route at commercial scale with precision and consistency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in the laboratory are fully realized in the manufacturing plant. Our facilities are equipped with stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to monitor impurity profiles, guaranteeing that every batch of Lansoprazole meets or exceeds international pharmacopeial standards. By leveraging our expertise in heterogeneous catalysis and process optimization, we can deliver a product that offers superior quality while maintaining a competitive cost structure.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this catalytic technology for your supply chain. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your volume requirements and quality targets. Whether you are looking to secure a secondary source for Lansoprazole or completely overhaul your existing manufacturing process, NINGBO INNO PHARMCHEM stands ready to be your strategic partner in innovation. Contact us today to discuss how we can collaborate to bring this advanced synthesis method to life in your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
