Scalable Organocatalytic Synthesis of Bioactive Chiral Chroman Compounds for Oncology Applications
Scalable Organocatalytic Synthesis of Bioactive Chiral Chroman Compounds for Oncology Applications
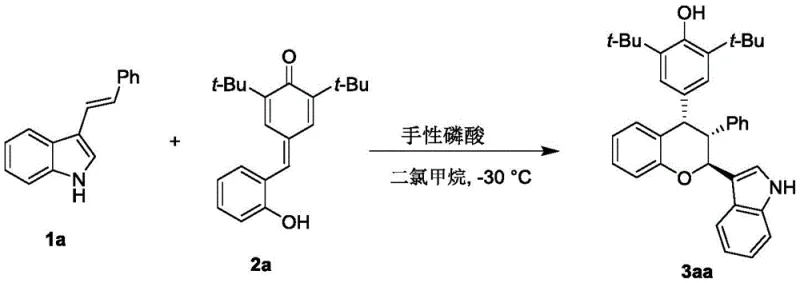
The pharmaceutical industry is constantly seeking robust and scalable methodologies for constructing complex chiral scaffolds, particularly those exhibiting potent biological activity. Patent CN112920173A introduces a significant advancement in this domain by disclosing a novel synthesis method for chiral chroman compounds, specifically designed to address the limitations of prior art regarding cytotoxicity and stereoselectivity. This technology leverages asymmetric organocatalysis to produce structurally diverse chroman derivatives that demonstrate strong cytotoxic activity against critical adenocarcinoma cell lines such as HepG2, MDA-MB-231, and SGC-7901. For R&D directors and procurement specialists, this represents a pivotal opportunity to access high-value intermediates through a greener, more efficient synthetic route that eliminates the reliance on expensive and toxic transition metal catalysts. The core innovation lies in the utilization of chiral phosphoric acid catalysts which facilitate the reaction between p-methylene benzoquinones and 3-indolenes with exceptional stereocontrol.
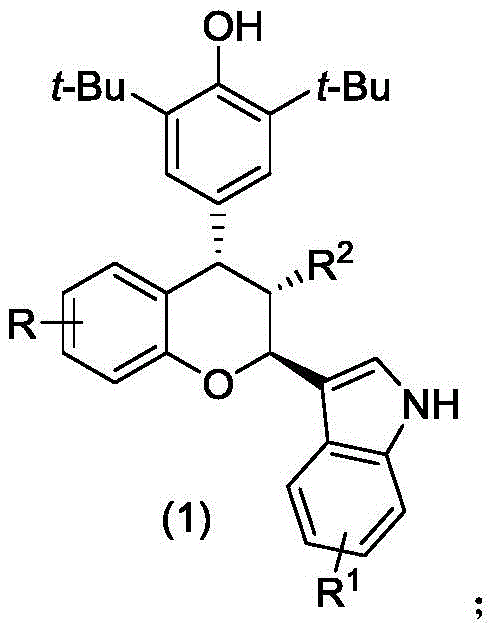
The structural versatility of the resulting chiral chroman compounds, as depicted in the general formula, allows for extensive structure-activity relationship (SAR) studies essential for drug discovery programs. The ability to tune substituents at the R, R1, and R2 positions enables medicinal chemists to optimize pharmacokinetic properties and potency without altering the core synthetic strategy. This flexibility is crucial for developing a reliable pipeline of pharmaceutical intermediates, ensuring that supply chains remain resilient even as molecular requirements evolve during clinical trials. Furthermore, the patent highlights that these compounds are not merely theoretical constructs but possess verified bioactivity, positioning them as high-priority targets for oncology therapeutic development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral chroman frameworks has been plagued by significant technical hurdles that impede efficient commercial manufacturing. Traditional approaches often rely on transition metal catalysis, which introduces severe complications regarding residual metal contamination, a critical quality attribute for any potential API or advanced intermediate. The removal of trace metals like palladium or rhodium requires additional downstream processing steps, such as scavenging or recrystallization, which drastically reduce overall yield and increase production costs. Moreover, many conventional methods suffer from poor enantioselectivity, necessitating costly chiral resolution steps that inherently waste up to 50% of the material. Safety is another major concern; older protocols frequently involve violent reaction conditions or unstable intermediates that pose risks of safety accidents in large-scale reactors, thereby increasing insurance and operational overheads for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology described in CN112920173A offers a paradigm shift by employing chiral phosphoric acid organocatalysts to drive the cycloaddition reaction. This approach operates under remarkably mild conditions, typically utilizing dichloromethane as a solvent at a controlled temperature of -30°C, which significantly enhances process safety and operational simplicity. The reaction proceeds via a concerted mechanism that inherently favors the formation of a single enantiomer, achieving extremely high enantiomeric excess (ee) values without the need for post-reaction resolution. This direct asymmetric synthesis not only improves atom economy but also simplifies the purification workflow to basic filtration and concentration followed by standard chromatography. The use of molecular sieves as additives further drives the equilibrium forward by sequestering water, ensuring high conversion rates and consistent product quality across different batches.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
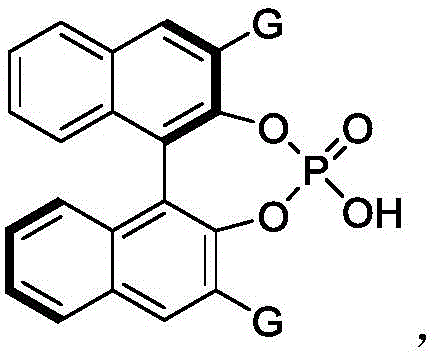
The success of this synthetic route hinges on the unique dual-activation capability of the chiral phosphoric acid catalyst. Mechanistically, the catalyst functions as a Brønsted acid, simultaneously activating both the electrophilic p-methylene benzoquinone and the nucleophilic 3-indolene substrate through a network of hydrogen bonds. This bifunctional activation organizes the transition state within a well-defined chiral pocket created by the bulky substituents on the catalyst backbone, such as the 9-anthracenyl groups found in preferred BINOL-derived catalysts. This precise spatial arrangement ensures that the nucleophilic attack occurs from a specific face of the quinone methide intermediate, thereby dictating the absolute stereochemistry of the newly formed chiral centers in the chroman ring system. Understanding this mechanism is vital for process chemists aiming to replicate these results, as it underscores the importance of maintaining strict anhydrous conditions and precise temperature control to preserve the integrity of the hydrogen-bonding network.
From an impurity control perspective, the organocatalytic nature of this reaction provides a distinct advantage over metal-catalyzed alternatives. Since no heavy metals are introduced into the reaction matrix, the risk of generating metal-associated impurities or catalyzing unwanted side reactions like over-oxidation is virtually eliminated. The primary impurities are likely to be diastereomers or unreacted starting materials, both of which are readily separated via standard silica gel chromatography using petroleum ether and ethyl acetate mixtures. The high diastereoselectivity (dr) observed in the examples, often exceeding 80:20, further simplifies the purification burden. For quality assurance teams, this translates to a cleaner crude profile and a more robust specification sheet, reducing the analytical workload required to release batches for subsequent coupling reactions or biological testing.
How to Synthesize Chiral Chroman Efficiently
To implement this synthesis effectively, operators must adhere to the specific stoichiometric ratios and environmental controls outlined in the patent data. The process begins with the preparation of a dry reaction vessel charged with dichloromethane and activated molecular sieves to ensure anhydrous conditions. The substrates, p-methylene benzoquinone and 3-indolene, are added in a molar ratio of approximately 1:1.2, followed by the introduction of the chiral phosphoric acid catalyst at a loading of 10 mol%. The reaction mixture is then cooled to -30°C and stirred for approximately 12 hours, with progress monitored via thin-layer chromatography (TLC) until the starting materials are fully consumed. Upon completion, the heterogeneous mixture is filtered to remove the molecular sieves, and the filtrate is concentrated under reduced pressure. The resulting residue is purified by flash column chromatography to isolate the target chiral chroman compound in high purity and yield.
- Prepare the reaction mixture by combining p-methylene benzoquinone and 3-indolene substrates in dichloromethane solvent with molecular sieves as an additive.
- Add the chiral phosphoric acid catalyst (such as a BINOL derivative) to the mixture and maintain the reaction temperature at -30°C while stirring.
- Monitor reaction progress via TLC, then filter to remove additives, concentrate the filtrate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of precious metal catalysts represents a direct cost reduction in pharmaceutical intermediate manufacturing, as it removes the need for expensive metal salts and the specialized scavenging resins required to meet regulatory limits on residual metals. This simplification of the bill of materials (BOM) stabilizes raw material costs and reduces dependency on volatile markets for rare earth elements or noble metals. Furthermore, the mild reaction conditions (-30°C) are easily achievable with standard industrial cooling systems, avoiding the extreme cryogenic requirements that can strain facility infrastructure and increase energy consumption. This operational efficiency contributes to a lower cost of goods sold (COGS) and improved margin potential for the final active ingredient.
- Cost Reduction in Manufacturing: The transition to metal-free organocatalysis fundamentally alters the cost structure of production by eliminating expensive catalyst recovery processes and reducing waste disposal costs associated with heavy metal contaminants. By utilizing commercially available chiral phosphoric acids and simple solvents like dichloromethane, the process minimizes the requirement for specialized reagents, leading to significant savings in raw material procurement. Additionally, the high yields and selectivity reported in the patent examples mean that less starting material is wasted, maximizing the throughput of existing reactor capacity and improving overall capital efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain continuity by reducing the risk of batch failures due to sensitive reaction parameters. Unlike processes requiring strict inert atmospheres or ultra-low temperatures, this protocol tolerates standard laboratory and plant conditions, making it easier to transfer technology between different manufacturing sites or contract development and manufacturing organizations (CDMOs). The availability of diverse substrates, as demonstrated by the broad scope in the patent, ensures that supply can be maintained even if specific starting materials face temporary shortages, as alternative analogs can be synthesized using the same platform technology.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles, facilitating smoother regulatory approvals and reducing the environmental footprint of production. The absence of toxic metals simplifies wastewater treatment and solid waste disposal, lowering compliance costs and mitigating the risk of environmental fines. The scalability is further supported by the simple workup procedure involving filtration and concentration, which are unit operations that scale linearly from gram to tonne quantities without significant re-optimization, ensuring a seamless transition from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral chroman synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aimed at clarifying the practical aspects of adopting this route for industrial applications. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this chemistry into their existing portfolios or outsourcing strategies.
Q: What are the key advantages of using chiral phosphoric acid catalysts for chroman synthesis?
A: The use of chiral phosphoric acid catalysts allows for extremely high enantioselectivity under mild reaction conditions (-30°C), avoiding the need for toxic transition metals and complex purification steps associated with traditional metal-catalyzed routes.
Q: What is the biological activity profile of the synthesized chiral chroman compounds?
A: Biological testing indicates that these compounds exhibit strong cytotoxic activity against various adenocarcinoma cell lines, including HepG2 liver cancer, MDA-MB-231 breast cancer, and SGC-7901 gastric cancer cells, making them valuable for oncology research.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method utilizes conventional solvents like dichloromethane and operates at manageable low temperatures, with a simple workup procedure involving filtration and chromatography, which facilitates safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral building blocks for the development of next-generation oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of chiral chroman intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to navigate the complexities of asymmetric synthesis, delivering products with the high enantiomeric excess necessary for biological efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this organocatalytic platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of reliability, quality, and scientific innovation.
