Advanced Catalytic Synthesis of Bioactive Chiral Chroman Compounds for Pharmaceutical Applications
Advanced Catalytic Synthesis of Bioactive Chiral Chroman Compounds for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN112920173A, which discloses a novel class of chiral chroman compounds exhibiting potent cytotoxic activity against aggressive adenocarcinoma cell lines. This patent not only introduces a new chemical entity with promising anticancer properties but also details a highly efficient asymmetric organocatalytic synthesis method. The core innovation lies in the utilization of chiral phosphoric acid catalysts to facilitate the reaction between p-methylene benzoquinones and 3-indolenes. This approach addresses long-standing challenges in stereoselective synthesis, offering a pathway to high-purity intermediates essential for drug development. For R&D directors and procurement specialists, understanding the nuances of this technology is vital for securing a reliable supply of high-value pharmaceutical intermediates.
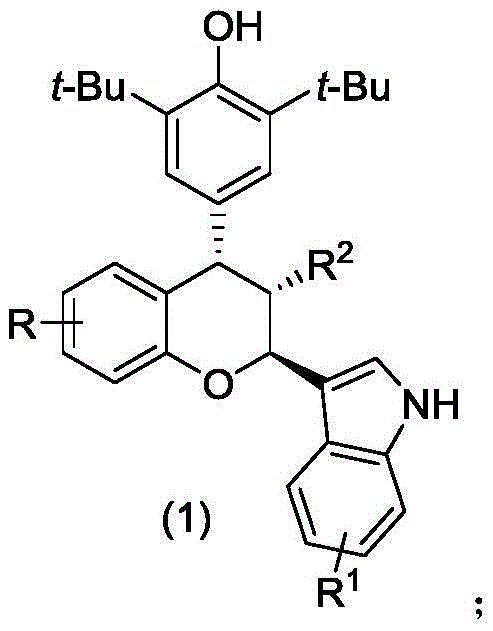
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral chroman derivatives has been plagued by significant technical hurdles that impede efficient manufacturing. Traditional methodologies often rely on harsh reaction conditions, including extreme temperatures and the use of stoichiometric amounts of hazardous reagents, which pose severe safety risks in an industrial setting. Furthermore, conventional routes frequently suffer from poor stereocontrol, resulting in racemic mixtures or products with low diastereomeric ratios that require costly and time-consuming purification steps. The violent nature of some prior art reactions increases the likelihood of operational errors and safety accidents, directly impacting production costs and yield consistency. Additionally, many existing methods lack substrate generality, limiting the structural diversity of the final products and restricting their applicability in medicinal chemistry programs. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inflated costs for high-purity chiral intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112920173A introduces a transformative catalytic strategy that operates under remarkably mild conditions. The novel approach employs a chiral phosphoric acid catalyst to drive the [4+2] cycloaddition or related cascade reactions between p-methylene benzoquinones and 3-indolenes. This organocatalytic system functions effectively at low temperatures, specifically around -30°C, in dichloromethane solvent, ensuring a controlled and safe reaction environment. The use of molecular sieves as additives further enhances the reaction efficiency by managing water content, which is critical for maintaining catalyst activity. This method achieves exceptional enantioselectivity and diastereoselectivity, often yielding products with high optical purity in a single step. The versatility of this protocol is demonstrated by its tolerance to a wide range of substituents on both the quinone and indole scaffolds, allowing for the rapid generation of diverse compound libraries.
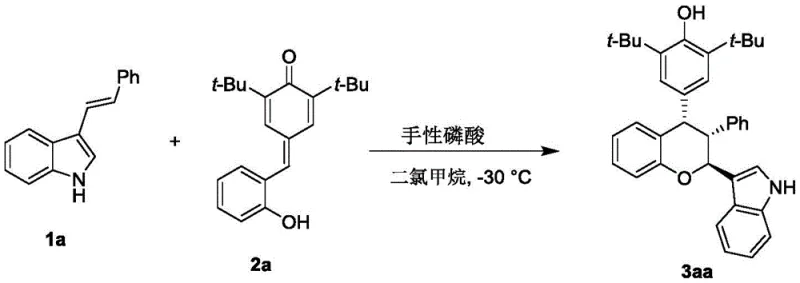
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthesis hinges on the precise mechanistic role played by the chiral phosphoric acid catalyst, which acts as a bifunctional organocatalyst. The catalyst, typically a BINOL-derived phosphoric acid with bulky substituents such as 9-anthracenyl groups, creates a well-defined chiral pocket around the reacting species. Through hydrogen bonding interactions, the acidic proton of the phosphate activates the electrophilic p-methylene benzoquinone, while the basic phosphoryl oxygen may simultaneously interact with the nucleophilic indole nitrogen or other functional groups. This dual activation lowers the energy barrier for the C-C bond formation while rigidly controlling the spatial orientation of the transition state. The steric bulk of the catalyst's 3,3'-substituents (e.g., anthracenyl or naphthyl groups) effectively shields one face of the reactive intermediate, forcing the incoming nucleophile to attack from the less hindered trajectory. This rigorous stereochemical control is what enables the production of chiral chromans with high enantiomeric excess (ee) values, often exceeding 70% and reaching much higher levels depending on the specific substrate combination.
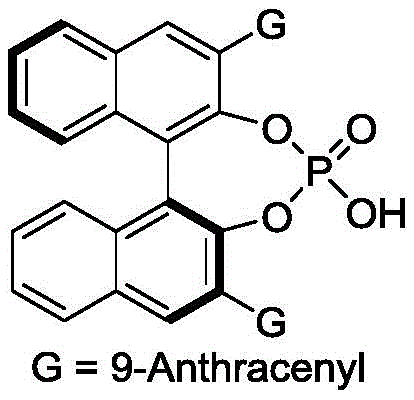
Furthermore, the mechanism ensures excellent impurity control, which is a paramount concern for pharmaceutical manufacturing. By favoring a specific transition state geometry, the catalyst minimizes the formation of unwanted diastereomers and regioisomers that typically complicate downstream processing. The mild reaction conditions prevent thermal degradation of sensitive functional groups, preserving the integrity of the molecular scaffold. The reaction proceeds through a concerted or stepwise ionic pathway that avoids the generation of free radical species, thereby reducing the risk of polymerization or side reactions common in quinone chemistry. This clean reaction profile translates directly into simplified purification protocols, often requiring only standard silica gel column chromatography to achieve analytical purity. For process chemists, this mechanistic clarity provides a solid foundation for scaling the reaction without encountering unexpected exotherms or byproduct accumulation.
How to Synthesize Chiral Chroman Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and stereoselectivity. The process begins with the preparation of dry reaction vessels and solvents, as moisture can deactivate the chiral phosphoric acid catalyst. The reactants, p-methylene benzoquinone and 3-indolene derivatives, are dissolved in anhydrous dichloromethane, and activated molecular sieves are added to the mixture to scavenge any trace water. The chiral catalyst is then introduced, typically at a loading of 10 mol%, and the reaction mixture is cooled to -30°C before stirring commences. Monitoring the reaction progress via thin-layer chromatography (TLC) is essential to determine the optimal endpoint, usually achieved within 12 hours. Upon completion, the mixture is filtered to remove the molecular sieves and catalyst residues, followed by concentration under reduced pressure. The crude product is then purified using silica gel column chromatography with a petroleum ether and ethyl acetate gradient to isolate the target chiral chroman. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Prepare the reaction mixture by combining p-methylene benzoquinone and 3-indolene derivatives in dichloromethane solvent with molecular sieves.
- Add the chiral phosphoric acid catalyst (BINOL derivative) to the mixture and maintain the temperature at -30°C while stirring.
- Monitor reaction progress via TLC, then filter, concentrate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral phosphoric acid catalyzed synthesis offers substantial benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies for pharmaceutical intermediates. The shift from harsh, metal-catalyzed, or stoichiometric methods to this organocatalytic protocol eliminates the need for expensive transition metals and the subsequent rigorous heavy metal removal steps required by regulatory agencies. This simplification of the downstream processing significantly reduces manufacturing costs and shortens the overall production cycle time. Moreover, the mild reaction conditions enhance operational safety, reducing the risk of plant shutdowns due to safety incidents and ensuring a more consistent supply continuity. The high atom economy and excellent yields reported in the patent examples mean that less raw material is wasted, contributing to a more sustainable and cost-effective manufacturing process. These factors collectively position this technology as a superior choice for securing a reliable supply of complex chiral building blocks.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the reduction in purification complexity lead to significant cost savings. By avoiding expensive chromatographic separations required for racemic mixtures and removing the need for metal scavengers, the overall cost of goods sold (COGS) is drastically lowered. The high yield and selectivity ensure that raw materials are converted efficiently into the desired product, minimizing waste disposal costs and maximizing resource utilization.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and a robust catalytic system mitigates the risk of supply disruptions. Unlike processes dependent on scarce or geopolitically sensitive reagents, this method utilizes common organic substrates that can be sourced from multiple suppliers. The simplicity of the reaction setup allows for flexible manufacturing across different facilities, enhancing the resilience of the supply chain against regional disturbances or logistical bottlenecks.
- Scalability and Environmental Compliance: The mild conditions and absence of toxic heavy metals make this process inherently greener and easier to scale. Scaling from gram to kilogram or ton quantities does not require specialized high-pressure or high-temperature equipment, reducing capital expenditure for capacity expansion. Furthermore, the reduced environmental footprint aligns with increasingly stringent global regulations on chemical manufacturing, facilitating smoother regulatory approvals and market access for the final drug products.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the synthesis and properties of these chiral chroman compounds. The following insights are derived directly from the experimental data and technical specifications outlined in the patent literature. Understanding these details is crucial for assessing the feasibility of integrating this synthetic route into your existing development pipelines.
Q: What are the key advantages of this chiral chroman synthesis method?
A: The method utilizes chiral phosphoric acid catalysis to achieve extremely high enantioselectivity under mild conditions (-30°C), avoiding the violent reactions and safety hazards associated with conventional synthesis routes.
Q: What is the biological activity of the synthesized compounds?
A: The resulting chiral chroman compounds exhibit strong cytotoxic activity against various adenocarcinoma cell lines, including HepG2 liver cancer, MDA-MB-231 breast cancer, and SGC-7901 gastric cancer cells.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses conventional reaction conditions, simple post-treatment procedures, and readily available substrates, making it highly suitable for commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing the technical expertise to translate complex academic discoveries like CN112920173A into commercial reality. Our team of experienced chemists is adept at optimizing organocatalytic processes, ensuring that the high enantioselectivity and yields observed in the lab are maintained during commercial scale-up. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by our state-of-the-art facilities and rigorous QC labs. Our commitment to quality ensures that every batch of chiral chroman intermediate meets stringent purity specifications, providing our partners with the confidence needed to advance their drug candidates through clinical trials.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their anticancer drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations for your next project.
