Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methods to access nitrogen-rich heterocycles that serve as privileged scaffolds in drug discovery. A recent breakthrough detailed in patent CN116253692A introduces a highly efficient preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds. These heterocyclic structures are renowned for their broad spectrum of biological activities, including anticancer, antifungal, anti-inflammatory, and antimalarial properties. The strategic incorporation of a trifluoromethyl group into these scaffolds significantly enhances physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, making them invaluable assets for modern medicinal chemistry programs. This new methodology represents a paradigm shift from traditional synthetic routes, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on classical condensation reactions, such as the interaction between amidrazones and 1,2-diketones or alkynes. Other established protocols involve multicomponent reactions utilizing hydrazides, dicarbonyl compounds, and ammonium acetate, or the cyclization of hydrazine with oxazolone and oxadiazole derivatives. While these methods have served the scientific community for decades, they are increasingly viewed as suboptimal for modern process chemistry. Conventional routes often suffer from harsh reaction conditions, requiring high temperatures or stringent anhydrous environments that complicate operational safety and energy consumption. Furthermore, these traditional substrates can be difficult to synthesize themselves, leading to longer overall synthetic sequences. Perhaps most critically for industrial application, many older methods exhibit poor structural diversity, limiting the ability of chemists to rapidly generate analog libraries for structure-activity relationship (SAR) studies, and often result in lower reaction efficiencies that hinder cost-effective production.
The Novel Approach
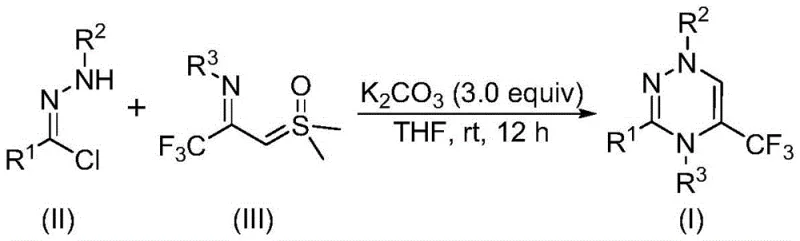
In stark contrast to the limitations of the past, the novel approach disclosed in the patent utilizes a synergistic [3+3] cycloaddition strategy that is both operationally simple and chemically powerful. By employing readily available chlorohydrazones and trifluoroacetyl sulfur ylides as starting materials, this method bypasses the need for complex substrate pre-functionalization. The reaction is promoted by potassium carbonate, an inexpensive and non-toxic inorganic base, and proceeds smoothly in common organic solvents like tetrahydrofuran (THF). Crucially, the process operates under ambient conditions—specifically at temperatures between 20°C and 40°C and in the presence of air—eliminating the need for expensive inert gas setups or cryogenic cooling. This accessibility allows for a broader range of functional group tolerance, enabling the synthesis of diverse derivatives with high yields, thereby accelerating the timeline from bench-scale discovery to pilot plant production.
Mechanistic Insights into Potassium Carbonate Promoted Cycloaddition
The elegance of this synthesis lies in its mechanistic simplicity and efficiency. The reaction initiates with the promotion of the chlorohydrazone by potassium carbonate, which facilitates the elimination of hydrogen chloride to generate a reactive nitrile imine intermediate in situ. This transient species is highly electrophilic and poised for cycloaddition. Subsequently, the nitrile imine undergoes a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide. This step is the cornerstone of the transformation, constructing the six-membered 1,2,4-triazine ring while simultaneously incorporating the vital trifluoromethyl group. The final stage of the mechanism involves the elimination of dimethyl sulfoxide (DMSO), driving the equilibrium toward the formation of the stable aromatic triazine product. Alternatively, the reaction may proceed through a stepwise pathway involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, ultimately converging on the same high-purity product. This dual mechanistic possibility ensures robustness across different substrate electronic profiles.
From an impurity control perspective, the use of mild inorganic bases like potassium carbonate offers distinct advantages over stronger, more nucleophilic bases that might degrade sensitive functional groups or promote polymerization of the reactive intermediates. The absence of transition metal catalysts removes a major source of potential contamination, specifically heavy metal residues that are strictly regulated in pharmaceutical ingredients. By avoiding metals such as palladium or copper, the process inherently reduces the burden on downstream purification steps, such as scavenging or recrystallization, which are often required to meet stringent regulatory limits for residual metals. This clean reaction profile ensures that the resulting trifluoromethyl substituted 1,2,4-triazine compounds possess a superior purity profile right out of the reactor, facilitating faster regulatory approval and reducing the risk of batch failures due to impurity spikes.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators should focus on the precise stoichiometric balance of reagents and the choice of solvent to maximize conversion. The patent specifies a molar ratio preference where the chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate are used in a ratio of approximately 1:2:3, ensuring complete consumption of the limiting reagent. Tetrahydrofuran is identified as the preferred solvent due to its ability to dissolve all reactants effectively while promoting high conversion rates. The reaction mixture is simply stirred at room temperature for a period of 10 to 14 hours, after which standard workup procedures involving filtration and silica gel treatment are employed. For detailed standardized operating procedures and specific safety guidelines regarding the handling of sulfur ylides and chlorohydrazones, please refer to the technical guide below.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this metal-free synthesis protocol offers substantial strategic benefits that extend beyond mere chemical yield. The elimination of precious metal catalysts represents a direct reduction in raw material costs, as palladium and copper salts are subject to significant market volatility and high price points. Furthermore, the removal of these metals from the process flow drastically simplifies the supply chain logistics by removing the need for specialized metal scavenger resins and the associated waste disposal protocols. This streamlining of the manufacturing process translates into a more resilient supply chain that is less susceptible to bottlenecks caused by the scarcity of specialized catalytic reagents. The reliance on commodity chemicals like potassium carbonate and common solvents ensures a stable and predictable cost structure for long-term production contracts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the complete avoidance of expensive transition metal catalysts. In traditional cross-coupling or cyclization reactions, the cost of the catalyst and the subsequent purification steps to remove trace metals can account for a significant portion of the total manufacturing cost. By utilizing potassium carbonate, a ubiquitous and low-cost inorganic salt, the direct material costs are significantly reduced. Additionally, the mild reaction conditions mean that energy consumption for heating or cooling is minimized, further lowering the operational expenditure (OPEX) associated with utility usage in the production facility. The simplified post-treatment process, which avoids complex metal scavenging, also reduces labor hours and consumable costs associated with purification.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials. Chlorohydrazones and trifluoroacetyl sulfur ylides can be synthesized from bulk commodities such as acyl chlorides, hydrazines, and trifluoroacetic acid, all of which are produced on a massive global scale. This reduces the risk of supply disruptions that often plague specialized reagents. Moreover, the reaction's tolerance to air and moisture means that storage and handling requirements for the reaction mixture are less stringent, reducing the risk of batch loss due to environmental exposure during transfer or processing. This robustness ensures consistent delivery schedules and reliable inventory management for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne levels is straightforward due to the absence of exothermic hazards often associated with strong bases or reactive metals. The ability to run the reaction at room temperature in an open vessel (air atmosphere) simplifies reactor design and reduces the capital expenditure required for specialized pressure-rated or inert-gas-blanketed equipment. From an environmental standpoint, the process generates less hazardous waste, particularly heavy metal-contaminated sludge, aligning with increasingly strict global environmental regulations. This green chemistry profile not only reduces waste disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, a key metric for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process chemists aiming to adapt this method for specific target molecules or for quality assurance teams evaluating the robustness of the manufacturing process.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN116253692A utilizes potassium carbonate as a promoter, completely avoiding the need for costly heavy metal catalysts like palladium or copper, which simplifies purification and reduces costs.
Q: What are the typical reaction conditions for this triazine formation?
A: The reaction proceeds efficiently at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours, eliminating the need for inert gas protection or extreme heating.
Q: Can this method accommodate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates high tolerance for various substituents including alkyl, alkoxy, halogens, and nitro groups on the phenyl rings, allowing for significant structural diversity in the final 1,2,4-triazine products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the methodology described in CN116253692A and is fully prepared to execute this synthesis with precision and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to full-scale market supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl 1,2,4-triazine compound delivered meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific drug discovery programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this metal-free approach can optimize your budget. We encourage you to contact us today to discuss your specific needs,索取 specific COA data for our existing inventory, or request route feasibility assessments for novel derivatives. Let us be your trusted partner in bringing innovative pharmaceutical solutions to life through superior chemical manufacturing.
