Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrol-2-One Derivatives for Global Pharma Supply Chains
Introduction to Next-Generation Pyrrol-2-One Manufacturing
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a critical pharmacophore found in a diverse array of bioactive natural products and therapeutic agents. As illustrated in the structural diversity of compounds like Althiomycin, Glimepiride, and Isomalyngamide A, this scaffold is indispensable for developing new antibiotics, hypoglycemic agents, and anticancer drugs. 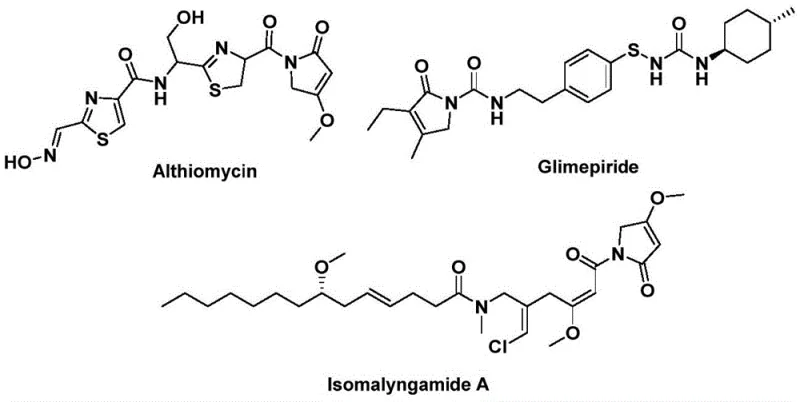 . Recognizing the immense value of this backbone, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as accessible starting materials. By shifting away from traditional, hazardous carbonylation methods, this technology offers a streamlined, one-step pathway to complex heterocycles, positioning it as a cornerstone for modern pharmaceutical intermediate manufacturing.
. Recognizing the immense value of this backbone, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as accessible starting materials. By shifting away from traditional, hazardous carbonylation methods, this technology offers a streamlined, one-step pathway to complex heterocycles, positioning it as a cornerstone for modern pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton via carbonylation has been fraught with significant operational and safety hurdles. Traditional protocols often rely on the direct use of carbon monoxide gas, which poses severe toxicity risks and requires specialized high-pressure equipment to maintain safe reaction conditions. Furthermore, conventional catalytic systems frequently suffer from limited substrate compatibility, struggling to accommodate electron-deficient or sterically hindered substituents without drastic drops in efficiency. These limitations not only inflate the cost of goods due to stringent safety protocols but also restrict the chemical space available for medicinal chemists exploring structure-activity relationships. The reliance on gaseous reagents also complicates the scale-up process, creating bottlenecks in supply chains that demand rapid iteration and large-volume production capabilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN112694430B revolutionizes this synthesis by employing a solid carbon monoxide surrogate, specifically 1,3,5-trimethoxyphenol ester (TFBen), within a palladium-catalyzed system. 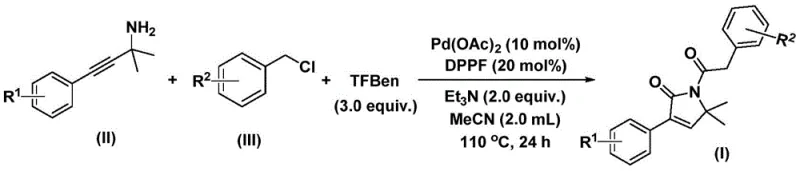 . This innovation allows the reaction to proceed under atmospheric pressure conditions in standard organic solvents like acetonitrile, effectively removing the need for high-pressure autoclaves. The process operates efficiently at moderate temperatures ranging from 100°C to 120°C, typically completing within 24 hours. By integrating the carbonylation and cyclization steps into a single pot, this method drastically reduces processing time and waste generation. The result is a robust, user-friendly protocol that maintains high reaction efficiency while expanding the scope of compatible functional groups, thereby accelerating the discovery and production of valuable pharmaceutical intermediates.
. This innovation allows the reaction to proceed under atmospheric pressure conditions in standard organic solvents like acetonitrile, effectively removing the need for high-pressure autoclaves. The process operates efficiently at moderate temperatures ranging from 100°C to 120°C, typically completing within 24 hours. By integrating the carbonylation and cyclization steps into a single pot, this method drastically reduces processing time and waste generation. The result is a robust, user-friendly protocol that maintains high reaction efficiency while expanding the scope of compatible functional groups, thereby accelerating the discovery and production of valuable pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The elegance of this transformation lies in its intricate catalytic cycle, which orchestrates the assembly of the five-membered lactam ring through precise organometallic steps. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the decomposition of the phenol ester surrogate, inserts into this metal-carbon bond to form an acyl-palladium species. This acyl intermediate then engages with the propargyl amine substrate, triggering a cyclization event that constructs the core five-membered ring structure. Following this, a second molecule of carbon monoxide inserts into the newly formed organopalladium bond, expanding the coordination sphere before final reductive elimination releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. This dual-carbonylation mechanism is highly selective, minimizing the formation of linear byproducts and ensuring that the nitrogen atom is correctly incorporated into the lactam framework.
From an impurity control perspective, the use of a solid CO surrogate provides a steady, controlled release of carbon monoxide, preventing the local concentration spikes that often lead to polymerization or side reactions in gas-phase carbonylations. The choice of the DPPP ligand further stabilizes the palladium center, promoting the specific reductive elimination pathway required for the target scaffold while suppressing competing beta-hydride elimination processes. This mechanistic precision translates directly to cleaner reaction profiles, simplifying downstream purification and reducing the burden on quality control laboratories. The ability to tolerate diverse electronic environments on the aromatic rings of both coupling partners suggests that the electronic properties of the ligand and the palladium center are well-balanced to accommodate both electron-rich and electron-poor substrates without compromising the integrity of the catalytic cycle.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and thermal conditions to maximize yield and purity. The protocol dictates the use of palladium acetate as the precatalyst alongside 1,1'-bis(diphenylphosphino)ferrocene (DPPP) as the supporting ligand, with triethylamine serving as the base to neutralize generated acid. The reaction is typically conducted in acetonitrile, which has been identified as the optimal solvent for solubilizing the diverse range of organic substrates while maintaining catalyst stability. Detailed standardized operating procedures regarding exact molar equivalents, addition sequences, and workup protocols are critical for reproducibility.
- Combine palladium acetate, DPPP ligand, triethylamine, TFBen (CO source), propargyl amine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity pyrrol-2-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from gaseous carbon monoxide to a bench-stable solid surrogate fundamentally alters the risk profile of the manufacturing process, eliminating the need for expensive gas handling infrastructure and specialized safety training associated with toxic gases. This simplification of the operational environment directly correlates to reduced capital expenditure and lower ongoing maintenance costs for production facilities. Furthermore, the high conversion rates and broad substrate tolerance mean that raw material sourcing becomes more flexible, allowing manufacturers to utilize a wider variety of commercially available benzyl chlorides and propargyl amines without fearing batch failures due to subtle electronic differences.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of readily available, inexpensive starting materials significantly lowers the barrier to entry for production. By avoiding the logistical complexities and safety premiums associated with carbon monoxide gas, manufacturers can achieve substantial cost savings in both CAPEX and OPEX. The high efficiency of the catalyst system ensures that precious metal loading remains economical, while the simplified one-pot nature of the reaction reduces solvent consumption and energy usage compared to multi-step alternatives.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like TFBen and common organic halides mitigates the risk of supply disruptions often caused by the regulated transport of hazardous gases. This stability ensures consistent production schedules and reliable delivery timelines for downstream pharmaceutical clients. Additionally, the robustness of the reaction conditions allows for greater flexibility in manufacturing locations, enabling decentralized production strategies that enhance overall supply chain resilience against regional instabilities.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Scaling this process from gram to tonnage quantities does not require exponential increases in safety engineering controls, facilitating a smoother transition from R&D to commercial manufacturing. The reduced waste profile and simpler workup procedures further contribute to a lower environmental footprint, supporting corporate sustainability goals and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production portfolios.
Q: What are the key advantages of using TFBen over carbon monoxide gas?
A: Using 1,3,5-trimethoxyphenol ester (TFBen) as a solid CO surrogate eliminates the need for handling toxic high-pressure carbon monoxide gas, significantly improving operational safety and simplifying reactor requirements for scale-up.
Q: What is the substrate scope for this palladium-catalyzed reaction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as methoxy, fluoro, chloro, bromo, trifluoromethyl, and alkyl groups on both the propargyl amine and benzyl chloride starting materials.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available catalysts and reagents, operates at moderate temperatures (110°C), and avoids hazardous gases, making it highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the drug development lifecycle. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including efficient metal scavenging protocols to ensure residual metal levels comply with the strictest regulatory guidelines.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a partnership built on transparency, quality, and shared success.
