Scalable Production of Bioactive 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds Using Novel Palladium Catalysis
Introduction to Next-Generation Pyrrolinone Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and synthetic pharmaceuticals. As highlighted in recent intellectual property developments, specifically patent CN112694430B, there is a critical industry shift towards more efficient, one-pot methodologies for constructing these complex heterocycles. This patent discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation to assemble the pyrrolinone ring system directly from readily available propargyl amines and benzyl chlorides. The significance of this chemical architecture cannot be overstated, as it underpins the biological activity of potent agents such as Althiomycin, an antibiotic with significant antibacterial properties, and Glimepiride, a widely prescribed hypoglycemic agent for diabetes management.
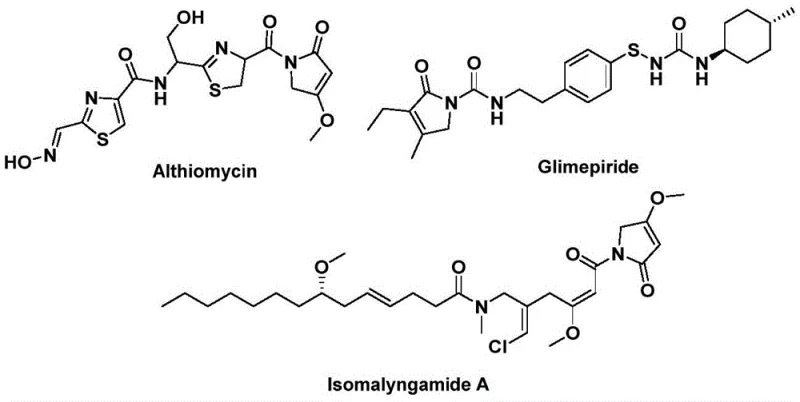
Furthermore, the scaffold is present in Isomalyngamide A, which has demonstrated potential anticancer activity, underscoring the versatility of this heterocyclic core in drug discovery pipelines. Traditional approaches to synthesizing these molecules often involve multi-step sequences with poor atom economy or require hazardous reagents. The novel approach detailed in the patent data offers a streamlined alternative that not only simplifies the synthetic route but also enhances the overall sustainability of the manufacturing process. For R&D directors and process chemists, this represents a tangible opportunity to reduce the complexity of synthesizing high-value intermediates while maintaining rigorous purity standards required for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has been fraught with synthetic challenges that hinder efficient commercial production. Conventional carbonylation strategies frequently rely on the direct use of carbon monoxide gas, which poses severe safety hazards due to its high toxicity and flammability, necessitating specialized high-pressure autoclaves and rigorous safety protocols that drive up capital expenditure. Moreover, traditional routes often suffer from limited substrate compatibility, where sensitive functional groups on the aromatic rings may degrade under harsh reaction conditions or fail to participate in the cyclization process effectively. Many existing methods also require pre-functionalized starting materials that are expensive and difficult to source, leading to inflated raw material costs and extended supply chain lead times. Additionally, multi-step syntheses typically result in cumulative yield losses and generate substantial chemical waste, creating environmental compliance burdens that modern green chemistry initiatives strive to eliminate.
The Novel Approach
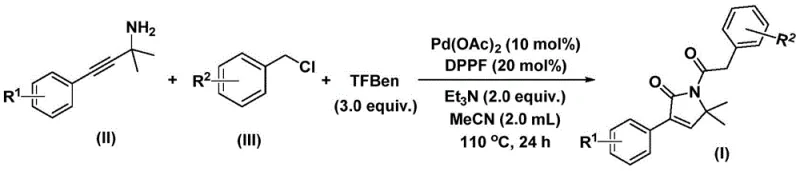
In stark contrast to these legacy methods, the technology described in patent CN112694430B introduces a sophisticated yet operationally simple protocol that utilizes a solid carbon monoxide surrogate, specifically identified as 1,3,5-tricarboxylic acid phenol ester (TFBen), to safely deliver the necessary carbonyl units in situ. This innovation effectively decouples the synthesis from the logistical nightmares associated with handling gaseous CO, allowing the reaction to proceed in standard glassware or reactors at atmospheric pressure equivalents. The process employs a robust palladium catalytic system comprising palladium acetate and the bidentate ligand DPPF (1,1'-bis(diphenylphosphino)ferrocene), which facilitates a highly selective bis-carbonylation cascade. By reacting propargyl amines with benzyl chlorides in the presence of this catalyst system and a base like triethylamine in acetonitrile, the method achieves the formation of the target pyrrolinone ring in a single operational step. This telescoped approach drastically reduces processing time and solvent consumption, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while expanding the accessible chemical space through excellent functional group tolerance.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The mechanistic elegance of this transformation lies in the precise orchestration of organometallic steps that convert simple linear precursors into a complex cyclic structure. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, the solid CO surrogate decomposes under the reaction conditions to release carbon monoxide, which inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack by the amino group of the propargyl amine, followed by coordination of the alkyne moiety to the metal center, setting the stage for the crucial cyclization event.
Following the initial ring closure, a second molecule of carbon monoxide inserts into the newly formed organopalladium bond, expanding the metallacycle and establishing the second carbonyl functionality essential for the lactam structure. The cycle concludes with a reductive elimination step that releases the final 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) catalyst to continue the turnover. This dual-insertion mechanism is critical for controlling the regioselectivity and ensuring the formation of the five-membered lactam ring rather than alternative oligomerization byproducts. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as ligand electronics and temperature, to suppress side reactions and maximize the yield of the desired pharmacological scaffold, ensuring a clean impurity profile that simplifies downstream purification.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
The practical execution of this synthesis is designed for scalability and reproducibility, utilizing standard laboratory and plant equipment without the need for exotic hardware. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, and the solid CO source in an organic solvent, followed by the addition of the amine and halide substrates. The mixture is then heated to a moderate temperature range of 100-120°C for a duration of 24 to 48 hours to ensure complete conversion. Post-reaction workup is straightforward, involving filtration to remove insoluble salts and catalyst residues, followed by standard silica gel column chromatography to isolate the pure product.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, triethylamine base, and the solid carbon monoxide surrogate (TFBen) in acetonitrile solvent.
- Add the propargyl amine substrate and benzyl chloride derivative to the mixture under inert atmosphere conditions.
- Heat the reaction mixture to 110°C for 24 hours, then filter and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this patented methodology offers compelling advantages that directly address the pain points of volatility and cost in the fine chemical sector. The shift from hazardous gaseous reagents to stable, solid surrogates fundamentally alters the risk profile of the manufacturing process, reducing insurance premiums and safety infrastructure costs while enabling production in a wider range of facilities. Furthermore, the reliance on commodity chemicals such as benzyl chlorides and propargyl amines, which are produced on a massive global scale, ensures a resilient supply chain that is less susceptible to the bottlenecks often seen with specialized, custom-synthesized building blocks.
- Cost Reduction in Manufacturing: The implementation of this one-pot bis-carbonylation strategy eliminates multiple isolation and purification steps inherent in traditional multi-step syntheses, leading to significant reductions in labor, solvent usage, and energy consumption. By avoiding the use of high-pressure equipment required for gaseous carbon monoxide, manufacturers can utilize standard reactor vessels, thereby lowering capital depreciation costs and maintenance overheads. The high reaction efficiency and yields reported in the patent data imply less raw material waste per kilogram of product, directly improving the gross margin profile for procurement managers negotiating long-term supply contracts.
- Enhanced Supply Chain Reliability: The use of robust, commercially available starting materials mitigates the risk of supply disruptions caused by the scarcity of exotic reagents. Since the reaction tolerates a wide variety of substituents on the aromatic rings, suppliers can maintain a flexible inventory of intermediates that can be rapidly converted into diverse final products based on fluctuating market demand. This adaptability shortens the effective lead time for high-purity pharmaceutical intermediates, allowing buyers to operate with leaner inventory levels and respond more agilely to changes in their own downstream API production schedules.
- Scalability and Environmental Compliance: The process operates in acetonitrile, a solvent with well-established recovery and recycling protocols in the chemical industry, facilitating adherence to strict environmental regulations regarding volatile organic compound (VOC) emissions. The absence of toxic gas handling simplifies the permitting process for new production lines and reduces the regulatory burden associated with workplace safety monitoring. As the industry moves towards greener manufacturing practices, adopting a methodology that minimizes waste generation and energy intensity positions supply chain leaders to meet corporate sustainability goals while securing a competitive advantage in tenders that prioritize eco-friendly production methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and scope of the method for technical decision-makers evaluating its adoption.
Q: What are the key advantages of using TFBen over gaseous CO in this synthesis?
A: Using TFBen (a solid CO surrogate) eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the logistics of scale-up compared to traditional carbonylation methods requiring toxic carbon monoxide gas.
Q: What is the substrate scope for the benzyl chloride component in this reaction?
A: The process exhibits excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing substituents such as fluoro, chloro, bromo, and trifluoromethyl groups on the aromatic ring.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes commercially available starting materials and standard organic solvents like acetonitrile, with reaction temperatures (110°C) that are easily achievable in standard industrial reactors, making it highly viable for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing reliable supply chains for our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrol-2-one intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against exacting standards.
We invite you to engage with our technical procurement team to discuss how this innovative palladium-catalyzed route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this streamlined process. We encourage potential partners to contact us immediately to obtain specific COA data for our catalog compounds and to schedule a consultation for route feasibility assessments, ensuring your next project starts with a foundation of chemical excellence and supply chain security.
