Scalable One-Step Synthesis of Bioactive Pyrrol-2-one Scaffolds via Palladium-Catalyzed Bis-Carbonylation
Introduction: A Breakthrough in Heterocyclic Synthesis
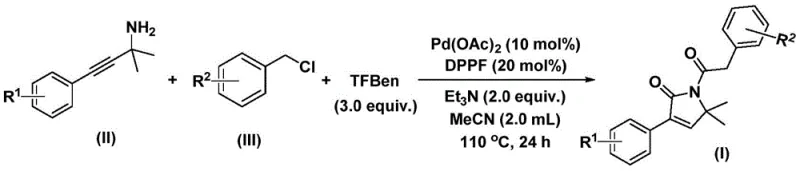
The structural motif of 1,5-dihydro-2H-pyrrol-2-one serves as a critical pharmacophore in numerous bioactive natural products and therapeutic agents, underpinning its immense value in modern drug discovery. As detailed in the recent patent CN112694430B, a novel preparation method has been developed that addresses long-standing challenges in constructing this privileged scaffold efficiently. This technology leverages a palladium-catalyzed bis-carbonylation strategy, transforming simple propargyl amines and benzyl chlorides into complex heterocycles in a single operational step. The significance of this advancement cannot be overstated for the pharmaceutical industry, where the rapid assembly of nitrogen-containing heterocycles is often a bottleneck in lead optimization.
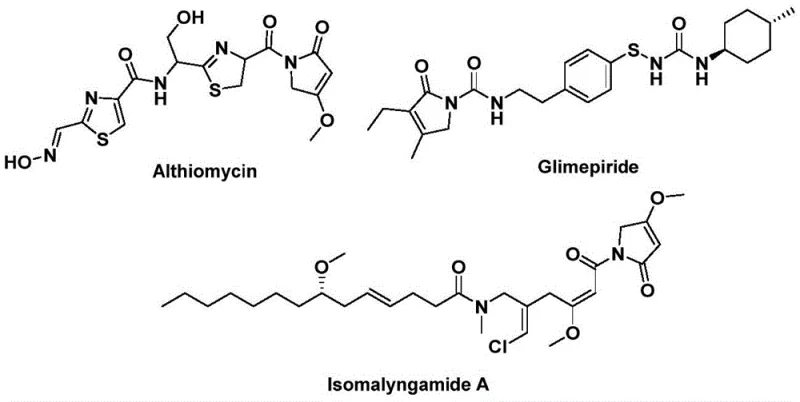
The biological relevance of this core structure is exemplified by potent molecules such as Althiomycin, an antibiotic with significant antibacterial activity, Glimepiride, a widely used hypoglycemic agent for diabetes management, and Isomalyngamide A, which exhibits promising anticancer properties. Accessing these complex architectures traditionally requires multi-step sequences involving hazardous reagents or harsh conditions. The new methodology described in the patent offers a streamlined alternative, utilizing a solid carbon monoxide surrogate to bypass the logistical and safety nightmares associated with gaseous CO. For R&D teams seeking reliable pharmaceutical intermediate suppliers, this route represents a paradigm shift towards safer, more sustainable chemistry.
Furthermore, the versatility of this synthetic approach allows for the introduction of diverse functional groups, enabling medicinal chemists to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. By employing readily available starting materials and a robust catalytic system, this invention not only simplifies the synthetic workflow but also enhances the overall atom economy of the process. As we delve deeper into the technical specifics, it becomes clear that this innovation holds substantial potential for reducing lead time in the development of high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied heavily on classical carbonylation reactions that utilize carbon monoxide gas directly. While effective in principle, the use of gaseous CO presents severe safety hazards, requiring specialized high-pressure autoclaves and rigorous leak detection systems that are often unavailable in standard laboratory settings. Moreover, traditional methods frequently suffer from poor regioselectivity and limited substrate scope, particularly when dealing with sensitive functional groups that may decompose under the requisite high pressures and temperatures. These limitations often force process chemists to resort to lengthy multi-step syntheses involving protection and deprotection strategies, which drastically increase waste generation and reduce overall yield.
Another significant drawback of conventional routes is the reliance on expensive or difficult-to-handle reagents that complicate supply chain logistics. The need for stoichiometric amounts of activating agents or harsh Lewis acids can lead to difficult purification processes, resulting in lower purity profiles that are unacceptable for pharmaceutical applications. Consequently, the commercial scale-up of complex heterocyclic intermediates via these traditional pathways is often plagued by high costs and inconsistent batch-to-batch reproducibility. For procurement managers, these inefficiencies translate into volatile pricing and unreliable delivery schedules, creating friction in the global supply of critical drug ingredients.
The Novel Approach
In stark contrast, the method disclosed in patent CN112694430B introduces a transformative one-pot synthesis that eliminates the need for gaseous carbon monoxide entirely. By utilizing a solid CO surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (referred to as TFBen in the reaction schemes), the reaction can be conducted in standard glassware at atmospheric pressure or mild sealing conditions. This substitution fundamentally alters the risk profile of the synthesis, making it accessible to a broader range of facilities without the need for specialized gas infrastructure. The reaction proceeds via a palladium-catalyzed cascade that seamlessly integrates carbonylation and cyclization steps, delivering the target pyrrol-2-one cores with remarkable efficiency.
The novelty of this approach also lies in its exceptional functional group tolerance, accommodating a wide array of substituents on both the propargyl amine and the benzyl chloride components. Electron-rich and electron-deficient aromatic rings are equally well-tolerated, allowing for the synthesis of diverse derivatives such as those bearing methoxy, fluoro, chloro, bromo, and trifluoromethyl groups. This broad compatibility is crucial for cost reduction in pharmaceutical intermediate manufacturing, as it reduces the need for custom-synthesized, highly specialized starting materials. The ability to achieve high yields (up to 92% in optimized examples) in a single step significantly shortens the production timeline, offering a compelling value proposition for supply chain stakeholders looking to optimize their inventory turnover.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis, orchestrated by a palladium species ligated by 1,1'-bis(diphenylphosphino)ferrocene (DPPF). The cycle initiates with the oxidative addition of the benzyl chloride to the active Pd(0) catalyst, generating a benzyl-palladium(II) intermediate. This step is critical as it activates the electrophilic carbon center for subsequent insertion reactions. Following this activation, the solid CO surrogate releases carbon monoxide in situ, which inserts into the palladium-carbon bond to form an acyl-palladium species. This controlled release of CO is key to maintaining low concentrations of the gas in the solution, thereby minimizing side reactions and enhancing safety.
Subsequently, the nucleophilic propargyl amine attacks the acyl-palladium intermediate, triggering a cyclization event that forms a five-membered ring palladium complex. The elegance of this mechanism is further highlighted by a second carbon monoxide insertion into this cyclic intermediate, expanding the coordination sphere before final reductive elimination occurs. This bis-carbonylation sequence effectively installs two carbonyl groups into the final structure, constructing the characteristic 1,5-dihydro-2H-pyrrol-2-one core with precision. The use of triethylamine as a base facilitates the deprotonation steps necessary for the cyclization and neutralizes the hydrochloric acid byproduct generated from the benzyl chloride, driving the equilibrium towards product formation.
Understanding this mechanism provides valuable insights into impurity control, a primary concern for R&D directors. The specificity of the palladium insertion and the sequential nature of the CO insertions minimize the formation of oligomeric byproducts or alternative cyclization isomers. Furthermore, the choice of acetonitrile as the solvent plays a pivotal role in stabilizing the polar intermediates and ensuring high conversion rates. By strictly controlling the molar ratios of the catalyst, ligand, and base, the process ensures that the catalytic cycle turns over efficiently, leaving minimal residual palladium in the crude product. This inherent cleanliness of the reaction profile simplifies downstream processing, aligning perfectly with the stringent purity specifications required for clinical grade materials.
How to Synthesize 1,5-dihydro-2H-pyrrol-2-one Efficiently
The practical execution of this synthesis is designed for robustness and scalability, making it an ideal candidate for technology transfer from lab to plant. The protocol involves charging a reaction vessel with palladium acetate, the DPPF ligand, triethylamine, the solid CO source (TFBen), the specific propargyl amine derivative, and the corresponding benzyl chloride in acetonitrile. The mixture is then heated to a temperature range of 100-120°C, typically around 110°C, and maintained for a period of 24 to 48 hours. This thermal energy drives the liberation of CO from the surrogate and powers the catalytic cycle to completion. Upon cooling, the reaction mixture is filtered to remove insoluble salts and catalyst residues, followed by adsorption onto silica gel.
- Combine palladium acetate, DPPF ligand, triethylamine, solid CO surrogate (TFBen), propargyl amine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing hazardous gaseous carbon monoxide with a stable, shelf-stable solid surrogate, companies can eliminate the regulatory burdens and insurance costs associated with storing and handling toxic gases. This shift not only enhances workplace safety but also streamlines the procurement process, as solid reagents are easier to transport and store in bulk quantities without specialized containment infrastructure. Consequently, this leads to significant cost reduction in fine chemical manufacturing by lowering overheads related to safety compliance and facility maintenance.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its high atom efficiency and single-step nature. Traditional multi-step routes incur costs at every stage, including solvent usage, workup materials, and labor for intermediate isolations. By consolidating the synthesis into one pot, the consumption of solvents and energy is markedly decreased. Additionally, the use of inexpensive, commodity-grade starting materials like benzyl chlorides and simple amines ensures that the raw material cost base remains low and stable. The high yields reported, often exceeding 80% and reaching up to 92% for optimized substrates, mean that less raw material is wasted, directly improving the gross margin of the final product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the commercial availability of all key reagents. Palladium acetate, DPPF, and the benzyl chloride precursors are standard catalog items available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions—tolerating slight variations in temperature and stoichiometry—ensures consistent batch quality, reducing the incidence of failed batches that can disrupt delivery schedules. This reliability is crucial for maintaining continuous production lines for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturing is never starved of critical inputs due to upstream synthesis failures.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of CO gas with a solid surrogate significantly reduces the carbon footprint and potential emissions of the process. The reaction generates minimal hazardous waste, and the use of acetonitrile, a common solvent with established recycling protocols, further aligns with green chemistry principles. The scalability of the process is evidenced by its successful demonstration across a wide range of substrates without the need for cryogenic conditions or ultra-high pressures. This ease of scale-up facilitates the rapid transition from gram-scale R&D to multi-kilogram or ton-scale commercial production, allowing companies to respond swiftly to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their own portfolios.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This method utilizes a solid carbon monoxide surrogate (TFBen) instead of toxic CO gas, significantly enhancing operational safety and eliminating the need for specialized high-pressure gas equipment.
Q: What is the substrate scope for the benzyl chloride and propargyl amine components?
A: The process demonstrates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl on both aromatic rings.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the reaction uses commercially available reagents, operates at moderate temperatures (100-120°C), and achieves high yields (up to 92%), making it highly viable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-dihydro-2H-pyrrol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN112694430B for the production of advanced heterocyclic building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand that in the competitive landscape of pharmaceutical intermediates, consistency and reliability are paramount, and our dedicated process development team is ready to optimize this route for your specific needs.
We invite you to collaborate with us to leverage this efficient synthesis for your next drug development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can improve your bottom line. We encourage potential partners to contact us to obtain specific COA data for similar compounds and comprehensive route feasibility assessments. Let us help you secure a stable, cost-effective supply of these vital scaffolds, empowering your R&D efforts with the high-quality materials necessary to bring life-saving therapies to market faster.
