Breakthrough in Metal-Free Oxazolidinone Synthesis for Commercial Pharmaceutical Manufacturing
Breakthrough in Metal-Free Oxazolidinone Synthesis for Commercial Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the demand for greener, safer, and more efficient synthetic routes. A pivotal development in this arena is detailed in patent CN113200932B, which discloses a novel method for synthesizing oxazolidinones using a bifunctional organocatalytic system. This technology leverages tri(dialkylamino)cyclopropenium halides (TDAC·X) to facilitate the [3+2] cycloaddition between glycidol and isocyanates. Unlike traditional methods that rely on heavy metals or harsh bases, this approach utilizes a Hydrogen Bond Donor (HBD) and Hydrogen Bond Acceptor (HBA) dual-activation mechanism. For R&D directors and procurement specialists seeking a reliable oxazolidinone supplier, this patent represents a paradigm shift towards metal-free purity and enhanced process safety, addressing critical pain points in the production of high-value fine chemicals.
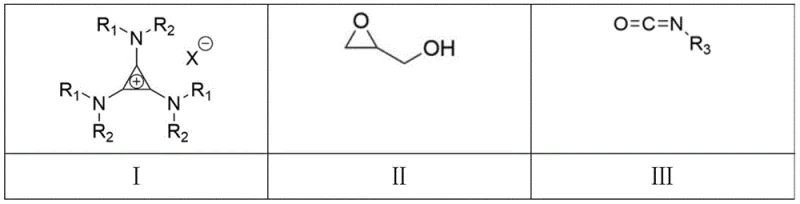
The core innovation lies in the catalyst design, which is derived from the reaction of tetrachlorocyclopropene with commercially available amines. This simplicity in catalyst preparation contrasts sharply with the complexity of ligand synthesis often required for transition metal catalysis. The resulting catalytic system operates effectively under mild thermal conditions, typically between 60°C and 100°C, and demonstrates broad substrate scope across various substituted isocyanates. This versatility makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a robust pathway to diverse oxazolidinone derivatives without the regulatory burden associated with metal contamination.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxazolidinones has relied heavily on metal catalysts or strong base promoters, which introduce significant drawbacks for industrial application. Traditional metal-catalyzed routes often suffer from the leaching of toxic metal residues into the final product, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical standards. Furthermore, basic conditions frequently induce competitive polymerization of glycidol or attack the distal carbon-oxygen bond of the epoxide, leading to the formation of unwanted six-membered ring byproducts. Perhaps most critically for chiral drug synthesis, conventional alkaline catalysts often cause inversion of configuration at the chiral center, resulting in racemization and a loss of optical purity that is unacceptable for enantiomerically pure active pharmaceutical ingredients.
The Novel Approach
The methodology outlined in CN113200932B overcomes these deficiencies through the deployment of neutral organic catalysts that function via a cooperative hydrogen bonding network. By avoiding metals entirely, the process eliminates the risk of biological toxicity and removes the need for specialized metal-scavenging resins during workup. The mild reaction environment preserves the stereochemical integrity of chiral glycidol, ensuring that the resulting oxazolidinone retains the original configuration. This is a decisive advantage for cost reduction in pharmaceutical intermediates manufacturing, as it simplifies the downstream processing and significantly improves the overall yield of the desired enantiomer. The ability to use crude catalyst preparations directly further streamlines the workflow, reducing both material costs and operational time.
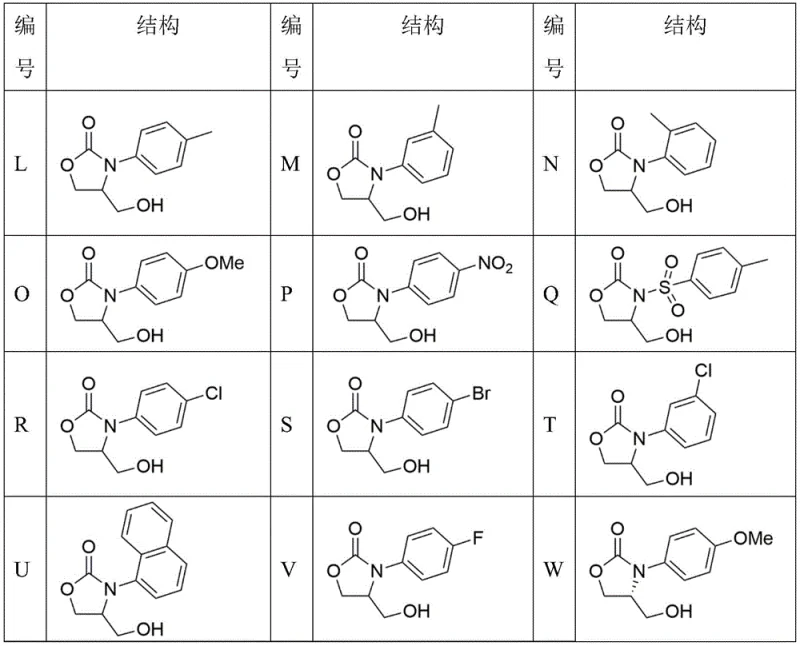
Mechanistic Insights into TDAC-Catalyzed Cycloaddition
The efficacy of this synthetic route is rooted in the unique electronic properties of the tri(dialkylamino)cyclopropenium cation paired with a halide anion. The cation acts as a potent Lewis acid equivalent, activating the epoxide ring of glycidol through electrostatic interactions, while the halide anion serves as a nucleophile or hydrogen bond acceptor to stabilize the transition state. This bifunctional activation lowers the energy barrier for the ring-opening of the epoxide by the isocyanate, facilitating the [3+2] cycloaddition without the need for aggressive reagents. The spatial arrangement of the catalyst likely creates a chiral environment or at least a sterically defined pocket that discourages the inversion of configuration, thereby maintaining the enantiomeric excess of the starting material throughout the transformation.
From an impurity control perspective, the mechanism inherently suppresses side reactions such as homopolymerization of the epoxide. In base-catalyzed systems, the alkoxide intermediate generated after ring opening is highly reactive and prone to attacking other epoxide molecules. In contrast, the TDAC system moderates the nucleophilicity of the intermediate through hydrogen bonding, directing the reaction selectively towards the isocyanate. This selectivity results in a cleaner crude reaction profile, minimizing the formation of oligomeric impurities and simplifying the final isolation of the target oxazolidinone. For quality control teams, this means a more consistent impurity profile and reduced variability between batches, which is essential for regulatory compliance.
How to Synthesize Oxazolidinone Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process begins with the in situ or ex situ preparation of the TDAC catalyst, followed by the combination of reactants in a non-polar solvent like chlorobenzene. The reaction proceeds under an inert atmosphere to prevent moisture interference, which could hydrolyze the isocyanate. Detailed procedural specifics regarding stoichiometry, temperature ramping, and workup procedures are critical for maximizing yield and are summarized in the technical guide below.
- Prepare the TDAC catalyst by reacting tetrachlorocyclopropene with the corresponding amine in an organic solvent like dichloromethane at 0°C, followed by room temperature stirring.
- In a reactor under inert gas, mix the catalyst with chlorobenzene solvent, then add glycidol and the specific isocyanate derivative.
- Heat the reaction mixture to 60°C-100°C (optimally 80°C) for 6-12 hours, then cool and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free catalytic technology offers tangible strategic benefits beyond mere chemical efficiency. The elimination of transition metals from the process map fundamentally alters the cost structure of production by removing the need for expensive catalysts and the associated validation of cleaning procedures to ensure no metal carryover. This translates directly into substantial cost savings and a more resilient supply chain, as the reliance on scarce or price-volatile precious metals is completely eradicated. Furthermore, the mild operating conditions reduce energy consumption and equipment stress, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of heavy metal catalysts and the simplification of purification. Traditional methods often require chromatography or specialized scavengers to remove ppm levels of metals, which adds significant operational expense. By using an organic salt catalyst that can be easily separated or tolerated, the process drastically reduces downstream processing costs. Additionally, the high conversion rates achieved under mild conditions minimize raw material waste, ensuring that expensive isocyanate and chiral glycidol inputs are utilized with maximum efficiency.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically the amines and tetrachlorocyclopropene required for the catalyst, are commercially available and stable. This contrasts with complex chiral ligands that may have long lead times or single-source dependencies. By simplifying the catalyst supply chain, manufacturers can reduce lead time for high-purity oxazolidinones and mitigate the risk of production stoppages due to material shortages. The robustness of the reaction also allows for flexible scheduling, as the mild conditions do not require specialized high-pressure or cryogenic infrastructure.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is facilitated by the absence of exothermic hazards often associated with strong bases or reactive metals. The use of chlorobenzene, while requiring proper handling, is a standard industrial solvent with established recovery protocols. The metal-free nature of the waste stream simplifies environmental compliance and wastewater treatment, reducing the regulatory burden on the manufacturing site. This ease of scale-up ensures that the transition from R&D to commercial production is smooth, supporting the continuous supply of critical pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazolidinone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the advantages of using TDAC catalysts over traditional metal catalysts for oxazolidinone synthesis?
A: TDAC catalysts eliminate toxic metal residues, which is critical for pharmaceutical applications. They also operate under milder conditions and prevent the racemization of chiral substrates, ensuring high optical purity.
Q: Can this synthesis method retain the chirality of the starting glycidol?
A: Yes, the bifunctional HBD/HBA mechanism allows for the [3+2] cycloaddition to proceed with retention of configuration, making it suitable for synthesizing chiral auxiliaries and bioactive molecules.
Q: Is the catalyst preparation complex or costly?
A: No, the catalyst is prepared in a single step from commercially available amines and tetrachlorocyclopropene. The crude product can often be used directly after simple extraction and drying, reducing preparation costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free organocatalysis in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the TDAC-catalyzed synthesis are successfully translated into robust industrial processes. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the absence of metal residues and the maintenance of chiral integrity, which are paramount for your regulatory filings.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific product portfolio. By leveraging our expertise, you can achieve significant process optimizations and secure a stable supply of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data for relevant oxazolidinone derivatives, and comprehensive route feasibility assessments tailored to your project requirements.
