Advanced Synthesis of Proton Pump Inhibitor Intermediates via N-Oxide Activation for Commercial Scale-Up
Advanced Synthesis of Proton Pump Inhibitor Intermediates via N-Oxide Activation for Commercial Scale-Up
The pharmaceutical industry's relentless demand for high-quality proton pump inhibitors (PPIs) such as omeprazole, lansoprazole, and pantoprazole necessitates robust and scalable synthetic routes for their key intermediates. Patent CN1214027C introduces a transformative methodology for obtaining derivatives of [[(pyridyl substituted)methyl]thio]benzimidazole, designated as Formula (I), which serve as critical precursors in the synthesis of these life-saving anti-ulcer agents. This technology addresses long-standing challenges in the production of high-purity pharmaceutical intermediates by replacing hazardous halogenation steps with a safer, more efficient N-oxide activation strategy. By leveraging active carboxylic or sulfonic acid anhydrides, manufacturers can achieve superior impurity profiles and streamlined processing, positioning this method as a cornerstone for reliable pharmaceutical intermediate supplier operations aiming for excellence in quality and efficiency.
![General Formula (I) representing the target [[(substituted pyridyl)methyl]thio]benzimidazole derivatives used in PPI synthesis](/insights/img/benzimidazole-synthesis-n-oxide-pharma-supplier-20260309103721-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Formula (I) compounds has relied heavily on the coupling of 2-halomethylpyridine derivatives (Formula II) with mercaptobenzimidazoles. While widely used, this conventional approach suffers from severe drawbacks that impact both operational safety and product quality. The preparation of Formula (II) typically involves radical halogenation using aggressive reagents like trichloroisocyanuric acid or N-chlorosuccinimide, or the substitution of hydroxymethyl groups with thionyl chloride. These processes often generate highly irritating intermediates, such as 2-chloromethyl-3-methylpyridine, which pose significant handling risks in large-scale manufacturing environments. Furthermore, a critical technical failure of this route is the formation of 4-chloro by-products, particularly when the starting material contains a nitro group at the 4-position. These halogenated impurities are chemically inert to nucleophilic substitution under standard conditions, making them exceptionally difficult to remove and resulting in final drug substances that fail stringent purity specifications required by global regulatory bodies.
The Novel Approach
In stark contrast to the hazardous halogenation pathways, the novel approach disclosed in the patent utilizes the N-oxide of the corresponding methylpyridine (Formula IV) as the starting material. This method fundamentally shifts the activation mechanism from radical halogenation to electrophilic activation using anhydrides. By reacting the N-oxide with reagents such as trifluoroacetic anhydride or methanesulfonic anhydride, the process generates a highly reactive intermediate (Formula V) bearing a superior leaving group (OCOR or OSO2R). This strategic change eliminates the need for handling corrosive halogenating agents and avoids the formation of the problematic 4-chloro impurities entirely. The resulting intermediate is not only less irritating but also allows for a telescoped process where the activation and coupling steps can potentially occur in the same reaction vessel, thereby reducing solvent consumption and waste generation while enhancing the overall economic viability of cost reduction in API manufacturing.
Mechanistic Insights into N-Oxide Activation and Coupling
The core of this innovative synthesis lies in the unique reactivity of the pyridine N-oxide functionality when exposed to strong electrophiles like acid anhydrides. In the first mechanistic step, the oxygen atom of the N-oxide attacks the carbonyl or sulfur center of the anhydride, leading to the formation of an activated species, specifically the Formula (V) intermediate. This intermediate features a leaving group at the 2-methyl position that is far more labile than a simple hydroxyl group but more controlled than a reactive halide. The electronic influence of the N-oxide moiety activates the adjacent methyl group towards nucleophilic attack, facilitating the subsequent displacement reaction. This activation strategy ensures that the reaction proceeds under milder thermal conditions, typically between 20°C and 90°C, preserving the integrity of sensitive functional groups on the pyridine ring that might otherwise degrade under harsh halogenation conditions.
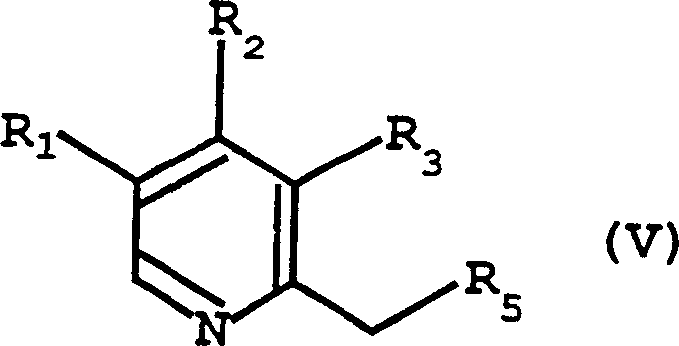
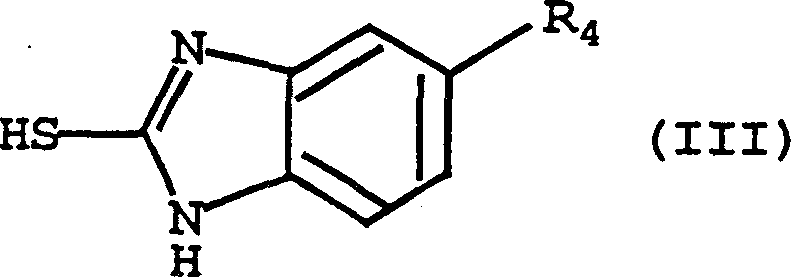
Following the formation of the activated intermediate, the second phase of the mechanism involves a nucleophilic substitution by the mercaptobenzimidazole derivative (Formula III). The sulfur atom of the benzimidazole thiol, enhanced by the presence of a base such as triethylamine or sodium methoxide, attacks the activated methyl carbon of the pyridine ring. This displaces the anhydride-derived leaving group (e.g., trifluoroacetate or mesylate) to form the stable thioether linkage characteristic of Formula (I). Crucially, this pathway avoids the competitive side reactions associated with halogenated intermediates. For instance, in the synthesis of omeprazole precursors, the absence of chlorine atoms prevents the formation of recalcitrant 4-chloro impurities. The use of mild bases and moderate temperatures (10-40°C) during this coupling step further ensures that the benzimidazole ring remains intact, preventing degradation and ensuring the high-purity pharmaceutical intermediates required for downstream oxidation to the active sulfoxide drugs.
How to Synthesize [[(Substituted Pyridyl)methyl]thio]benzimidazole Efficiently
The practical implementation of this synthesis route offers a straightforward protocol adaptable to various scales of production. The process begins with the dissolution of the methylpyridine N-oxide in a suitable organic solvent such as dichloromethane or 1,2-dichloroethane, followed by the controlled addition of the activating anhydride. Detailed standardized synthesis steps see the guide below.
- React the corresponding methylpyridine N-oxide (Formula IV) with an active carboxylic acid anhydride (e.g., trifluoroacetic anhydride) or sulfonic acid anhydride (e.g., methanesulfonic anhydride) in an organic solvent like dichloromethane at 20-90°C to form the activated intermediate (Formula V).
- Without isolating the intermediate, or after isolation, react the Formula (V) compound with the corresponding 2-mercaptobenzimidazole derivative (Formula III) in the presence of a base such as triethylamine or sodium methoxide.
- Maintain the reaction mixture at 10-40°C until completion, then isolate the final Formula (I) product by precipitation in water/alcohol mixtures or extraction, achieving high purity suitable for subsequent oxidation to sulfoxides.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this N-oxide activation technology presents compelling economic and logistical benefits that extend beyond mere chemical elegance. By eliminating the need for specialized halogenating equipment and the rigorous safety protocols associated with handling lachrymatory halomethyl compounds, facilities can significantly lower their operational overheads. The ability to telescope the reaction steps reduces the total cycle time and minimizes the volume of solvents required for intermediate isolations, directly contributing to substantial cost savings in raw material and waste disposal budgets. Furthermore, the simplified impurity profile reduces the burden on quality control laboratories, allowing for faster release times and more predictable batch schedules, which is essential for maintaining continuity in the supply of critical medical therapies.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous chlorinating agents, combined with the potential for one-pot processing, drastically simplifies the production workflow. This reduction in unit operations translates to lower energy consumption and reduced labor costs per kilogram of product. Additionally, the higher selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and yield efficiency without the need for complex and costly purification chromatography steps often required to remove halogenated impurities.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically pyridine N-oxides and common acid anhydrides like methanesulfonic anhydride, are commodity chemicals with robust global supply chains. Unlike specialized halogenated intermediates which may have limited suppliers and long lead times, these reagents are readily available from multiple sources. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring a steady and reliable flow of intermediates to downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by avoiding the generation of stoichiometric amounts of halogenated waste streams. The by-products formed are typically carboxylic or sulfonic acids which are easier to treat and neutralize compared to halogenated organic waste. This ease of waste management facilitates smoother regulatory approvals for plant expansions and supports the commercial scale-up of complex heterocyclic compounds to multi-tonne capacities without exceeding environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology resolves specific bottlenecks in PPI intermediate production.
Q: What are the primary advantages of this N-oxide activation method over traditional halogenation?
A: The primary advantage is the avoidance of highly irritating and hazardous 2-halomethylpyridine intermediates (Formula II). Traditional methods often generate difficult-to-remove 4-chloro impurities, whereas this anhydride activation route produces cleaner intermediates (Formula V) that react efficiently without forming these persistent halogenated by-products, significantly simplifying purification.
Q: Can this process be performed as a one-pot reaction to reduce costs?
A: Yes, the patent explicitly describes that the two steps can be carried out consecutively and optionally in the same reaction medium. The intermediate (Formula V) can be reacted in situ with the mercaptobenzimidazole without isolation, which drastically reduces solvent usage, processing time, and overall manufacturing costs compared to multi-step isolation processes.
Q: What specific anhydrides are recommended for activating the N-oxide?
A: The patent identifies trifluoroacetic anhydride (TFAA) and methanesulfonic anhydride (Ms2O) as particularly effective reagents. TFAA is preferred for generating trifluoroacetoxy intermediates, while methanesulfonic anhydride generates mesylate intermediates, both of which serve as excellent leaving groups for the subsequent nucleophilic substitution by the benzimidazole thiol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [[(Substituted Pyridyl)methyl]thio]benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of critical impurities such as the 4-chloro derivatives discussed in legacy methods. Our facility is equipped to handle the specific solvent systems and temperature controls required for the N-oxide activation process, guaranteeing consistency and reliability for our partners.
We invite you to collaborate with us to optimize your supply chain for proton pump inhibitor intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of Patent CN1214027C can enhance your manufacturing efficiency and reduce your total cost of ownership.
