Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. These complex molecular scaffolds, often featuring indolinone and imidazole motifs linked by a carbonyl bridge, are highly valued in medicinal chemistry for their broad-spectrum biological activities. The patent introduces a transition metal palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions, specifically at 30°C, utilizing inexpensive and readily available starting materials. This technological advancement represents a paradigm shift from traditional high-pressure carbonylation processes, offering a streamlined pathway that eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic frameworks has presented substantial challenges for process chemists and manufacturing teams. Conventional strategies often rely on the direct coupling of two pre-formed heterocyclic substrates, which can suffer from low atom economy and require harsh reaction conditions that degrade sensitive functional groups. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles frequently necessitate stoichiometric amounts of oxidants, generating significant chemical waste and complicating downstream purification. Perhaps the most critical bottleneck in traditional carbonylation chemistry is the reliance on exogenous carbon monoxide gas. Handling pressurized CO cylinders requires specialized infrastructure, rigorous safety protocols, and expensive containment systems to prevent leakage, all of which drastically inflate capital expenditure and operational costs. Furthermore, the use of toxic CO gas poses severe health risks to personnel and creates regulatory hurdles for facility licensing, making these routes less attractive for modern, sustainability-focused manufacturing environments.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN115353511A offers a sophisticated yet operationally simple solution through a multi-component tandem reaction. This innovative approach utilizes a palladium catalyst system, specifically palladium chloride paired with a trifurylphosphine ligand, to orchestrate the simultaneous assembly of three distinct building blocks: trifluoroethylimidoyl chloride, propargylamine, and an acrylamide derivative. 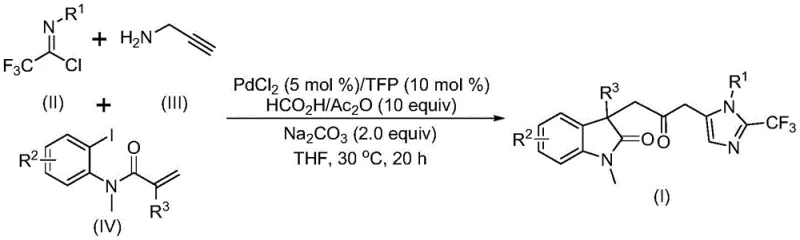 The brilliance of this method lies in its use of a formic acid and acetic anhydride mixture as a safe, liquid carbon monoxide surrogate. This in-situ generation of CO allows the carbonylation step to proceed at atmospheric pressure and a mild temperature of 30°C, completely bypassing the need for high-pressure reactors or toxic gas handling. The reaction demonstrates exceptional versatility, tolerating a wide array of substituents on the aromatic rings, including halogens, alkyls, and electron-withdrawing groups, thereby enabling the rapid synthesis of diverse libraries of biheterocyclic compounds. This not only accelerates the drug discovery process but also provides a robust platform for the commercial production of high-purity pharmaceutical intermediates with improved safety profiles.
The brilliance of this method lies in its use of a formic acid and acetic anhydride mixture as a safe, liquid carbon monoxide surrogate. This in-situ generation of CO allows the carbonylation step to proceed at atmospheric pressure and a mild temperature of 30°C, completely bypassing the need for high-pressure reactors or toxic gas handling. The reaction demonstrates exceptional versatility, tolerating a wide array of substituents on the aromatic rings, including halogens, alkyls, and electron-withdrawing groups, thereby enabling the rapid synthesis of diverse libraries of biheterocyclic compounds. This not only accelerates the drug discovery process but also provides a robust platform for the commercial production of high-purity pharmaceutical intermediates with improved safety profiles.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
For R&D directors focused on process robustness and impurity control, understanding the mechanistic underpinnings of this transformation is crucial. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating an organopalladium intermediate. This is followed by an intramolecular Heck-type reaction, which constructs the indolinone core and forms a divalent alkyl-palladium species. Subsequently, the carbon monoxide released from the decomposition of the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, yielding an acyl-palladium intermediate. This acyl species is the pivotal junction where the carbonyl bridge is established. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine compound, which undergoes isomerization. The final cyclization step involves the activation of this amidine by the acyl-palladium intermediate, leading to the formation of the imidazole ring and the release of the final carbonyl-bridged biheterocyclic product along with the regeneration of the palladium catalyst.
This intricate catalytic cycle offers significant advantages regarding impurity management and product quality. By operating at a low temperature of 30°C, the thermal energy available for side reactions is minimized, effectively suppressing the formation of degradation products or polymerization by-products that often plague high-temperature processes. The use of a specific ligand system, trifurylphosphine, enhances the stability of the palladium center and improves the selectivity of the insertion steps, ensuring that the desired C-C and C-N bonds are formed with high fidelity. Moreover, the in-situ generation of CO ensures a steady, controlled concentration of the carbonylating agent, preventing the accumulation of reactive intermediates that could lead to oligomerization. For procurement and quality assurance teams, this mechanistic precision translates to a cleaner crude reaction profile, which simplifies the purification workflow and reduces the burden on analytical laboratories, ultimately delivering a high-purity API intermediate that meets stringent regulatory specifications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, and the CO source mixture in a suitable organic solvent such as tetrahydrofuran (THF). The three key substrates are then introduced, and the mixture is stirred at 30°C for a duration of 12 to 20 hours. This extended reaction window ensures complete conversion of the starting materials, maximizing yield without the need for excessive catalyst loading. Following the reaction, the workup procedure is straightforward, involving simple filtration to remove inorganic salts and silica gel treatment, followed by standard column chromatography to isolate the pure product.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction mixture at a mild temperature of 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology addresses several critical pain points associated with the sourcing and manufacturing of complex heterocyclic intermediates. The elimination of toxic carbon monoxide gas is perhaps the most significant advantage, as it removes the requirement for specialized high-pressure infrastructure and hazardous gas storage facilities. This fundamentally alters the cost structure of the manufacturing process, allowing production to occur in standard glass-lined or stainless steel reactors without the need for expensive safety interlocks or gas detection systems. Consequently, this leads to a substantial reduction in capital investment and operational overhead, making the final product more cost-competitive in the global market. Furthermore, the use of cheap and commercially available starting materials, such as propargylamine and acrylamide derivatives, ensures a stable and resilient supply chain that is less susceptible to raw material price volatility or shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplification of the reaction engineering requirements. By avoiding high-pressure carbonylation, manufacturers can utilize existing general-purpose equipment, thereby deferring capital expenditures on specialized reactors. Additionally, the mild reaction conditions of 30°C significantly reduce energy consumption compared to processes requiring reflux or heating, contributing to lower utility costs. The high atom economy of the multi-component reaction means that fewer raw materials are wasted, and the high yields reported in the patent examples minimize the loss of valuable intermediates during purification. These factors collectively drive down the cost of goods sold (COGS), providing a competitive edge for companies adopting this technology for large-scale production.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical manufacturers, and this synthesis route enhances reliability through the use of robust, non-hazardous reagents. The reliance on liquid reagents like formic acid and acetic anhydride, rather than compressed gases, simplifies logistics and storage, reducing the risk of supply disruptions caused by transportation regulations or safety incidents. The broad substrate scope demonstrated in the patent indicates that the process is flexible; if a specific substituted acrylamide becomes unavailable, alternative analogs can likely be substituted without re-optimizing the entire process. This flexibility allows procurement managers to diversify their supplier base and mitigate risks associated with single-source dependencies, ensuring a steady flow of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this method has been validated at the gram scale, indicating strong potential for kilogram and tonne-level production. The absence of toxic CO gas greatly simplifies environmental, health, and safety (EHS) compliance, as there is no risk of fugitive emissions of a lethal gas. The waste stream is primarily composed of organic solvents and inorganic salts, which are easier to treat and dispose of compared to heavy metal-contaminated waste or hazardous gas scrubber effluents. This alignment with green chemistry principles not only reduces environmental impact but also streamlines the permitting process for new manufacturing lines, accelerating the time-to-market for new drug candidates utilizing these biheterocyclic scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations.
Q: How does this method improve safety compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in-situ, significantly reducing operational hazards and regulatory burdens.
Q: What is the substrate compatibility of this palladium-catalyzed system?
A: The process demonstrates excellent functional group tolerance, successfully accommodating various substituents on the aryl rings including halogens, alkyl groups, alkoxy groups, and electron-withdrawing groups like nitro and trifluoromethyl.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the patent explicitly validates the method through gram-scale reactions, confirming its robustness and potential for commercial scale-up without the need for specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the pharmaceutical industry continues to demand more complex and potent therapeutic agents, the ability to efficiently synthesize advanced heterocyclic intermediates becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging cutting-edge technologies like the palladium-catalyzed carbonylation cascade to deliver high-value building blocks to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical stage or full-scale commercialization. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of carbonyl-bridged biheterocyclic compounds meets the highest standards of quality and consistency required for GMP manufacturing.
We invite you to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this safer and more efficient methodology. Please contact our technical procurement team today to request specific COA data for our available inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us partner with you to accelerate your drug development timeline and bring life-saving medicines to patients faster.
