Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
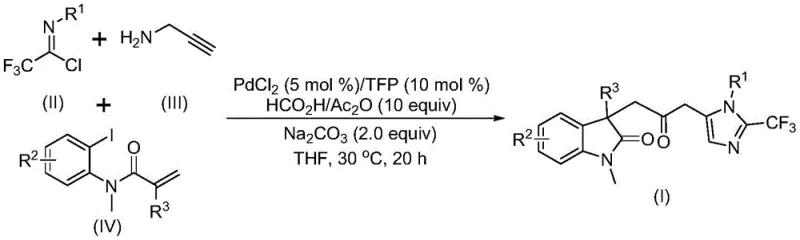
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the synthesis of indolinone-imidazole derivatives that are prevalent in modern drug discovery pipelines. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. By operating under mild conditions at 30°C without the need for toxic carbon monoxide gas, this invention addresses critical safety and efficiency bottlenecks found in conventional synthetic methodologies. The ability to construct multiple chemical bonds in a single step through a multicomponent reaction not only streamlines the synthetic pathway but also enhances the overall atom economy, making it a highly attractive candidate for reliable pharmaceutical intermediate supplier networks looking to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied on strategies that often suffer from significant operational drawbacks and limited substrate scope. Traditional approaches typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires extensive pre-functionalization and results in poor step economy. Alternatively, oxidative cyclization reactions using activated methyl-substituted heterocycles often necessitate stoichiometric amounts of harsh oxidants, generating substantial waste and complicating the purification process. Furthermore, existing carbonylation methods frequently depend on the use of high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that increases capital expenditure. These conventional pathways often struggle with functional group tolerance, limiting the diversity of substituents that can be introduced onto the final molecular scaffold, thereby restricting the chemical space available for medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353511A represents a paradigm shift by employing a palladium-catalyzed multicomponent tandem reaction that efficiently assembles the target architecture in a single pot. This novel approach utilizes a safe in situ generated carbon monoxide surrogate system comprising formic acid and acetic anhydride, effectively eliminating the hazards associated with handling toxic CO gas cylinders. The reaction proceeds at a remarkably low temperature of 30°C, which minimizes energy consumption and prevents the decomposition of thermally sensitive functional groups. By integrating trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the process simultaneously forms carbon-carbon and carbon-nitrogen bonds, drastically reducing the number of isolation steps required. This streamlined workflow not only accelerates the timeline from bench to kilogram scale but also ensures high reaction efficiency and excellent compatibility with diverse electronic and steric environments on the substrate.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. Following this activation, an intramolecular Heck reaction occurs to generate a divalent alkyl-palladium intermediate, which sets the stage for the subsequent carbonylation event. The presence of the formic acid and acetic anhydride mixture facilitates the release of carbon monoxide in situ, which then inserts into the palladium-carbon bond to form a reactive acyl-palladium intermediate. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species that undergoes isomerization. The final cyclization is driven by the activation of this amidine compound by the acyl-palladium intermediate, leading to the formation of the robust carbonyl-bridged biheterocyclic core with high regioselectivity.
Understanding the impurity profile is crucial for maintaining high-purity pharmaceutical intermediate standards, and this catalytic system offers inherent advantages in this regard. The mild reaction temperature of 30°C significantly suppresses side reactions such as polymerization of the acrylamide or decomposition of the imidoyl chloride, which are common issues in high-temperature processes. The use of TFP (trifurylphosphine) as a ligand stabilizes the palladium center, preventing the formation of palladium black and ensuring a consistent catalytic turnover throughout the 12 to 20-hour reaction window. Furthermore, the specific stoichiometry employed, with a slight excess of propargylamine and acrylamide relative to the imidoyl chloride, drives the equilibrium towards the desired product while minimizing the accumulation of unreacted starting materials. This precise control over reaction kinetics results in a cleaner crude reaction mixture, simplifying the downstream purification via column chromatography and ensuring the final product meets stringent quality specifications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding reagent addition and atmospheric control to maximize yield and reproducibility. The process begins with the preparation of a catalyst solution containing palladium chloride and the TFP ligand in an aprotic solvent such as tetrahydrofuran, which has been identified as the optimal medium for solubilizing all components and promoting the catalytic cycle. Detailed standardized synthesis steps see the guide below.
- Combine palladium chloride (5 mol%), TFP ligand (10 mol%), sodium carbonate (2.0 equiv), and a mixture of formic acid/acetic anhydride (10 equiv) in an organic solvent such as THF.
- Add the substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative (IV) to the reaction vessel under inert atmosphere.
- Stir the reaction mixture at 30°C for 12 to 20 hours, then filter, concentrate, and purify the crude product via column chromatography to isolate the target biheterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The reliance on commercially available and inexpensive starting materials, such as propargylamine and acrylamide derivatives, ensures a stable and cost-effective supply chain that is not vulnerable to the volatility of exotic reagent markets. By eliminating the need for high-pressure carbon monoxide infrastructure, facilities can avoid significant capital investments in safety equipment and regulatory compliance measures associated with toxic gas handling. This reduction in operational complexity directly contributes to cost reduction in API manufacturing, allowing for more competitive pricing structures in the final active pharmaceutical ingredient market. Additionally, the robustness of the reaction conditions allows for flexible scheduling and batch processing, enhancing overall supply chain reliability and responsiveness to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant palladium catalysts at low loading levels (5 mol%) combined with inexpensive ligands, which drastically lowers the cost of goods sold compared to precious metal-heavy alternatives. The elimination of toxic gas handling removes the need for specialized scrubbing systems and continuous monitoring equipment, resulting in substantial overhead savings. Furthermore, the high conversion rates achieved under mild conditions minimize solvent usage and energy consumption for heating and cooling, contributing to a leaner and more sustainable manufacturing footprint. These cumulative efficiencies allow manufacturers to offer high-purity intermediates at a price point that supports margin expansion for downstream drug developers.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for long-term production planning, and this method excels by utilizing commodity chemicals that are widely produced and stocked globally. The simplicity of the reaction setup means that production can be easily transferred between different manufacturing sites without the need for highly specialized reactor configurations, thereby mitigating the risk of single-point failures in the supply network. The broad substrate compatibility ensures that variations in raw material grades can be accommodated without compromising product quality, providing a buffer against supply disruptions. This flexibility empowers supply chain heads to maintain continuous production schedules even during periods of market fluctuation, ensuring timely delivery of critical intermediates to clients.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is seamless due to the exothermic nature of the reaction being well-managed at 30°C, removing the thermal runaway risks associated with large-scale batch processing. The absence of toxic carbon monoxide gas aligns perfectly with increasingly stringent environmental, health, and safety (EHS) regulations, simplifying the permitting process for new production lines. Waste generation is minimized through high atom economy and the use of benign solvents like THF, which can be readily recovered and recycled, supporting corporate sustainability goals. This environmentally conscious approach not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this process for integration into their own manufacturing portfolios. Understanding these details is essential for assessing the feasibility of adopting this method for specific project requirements.
Q: What are the key advantages of this Pd-catalyzed method over traditional biheterocycle synthesis?
A: Unlike traditional methods requiring toxic carbon monoxide gas or harsh oxidative conditions, this patent describes a mild, one-pot multicomponent reaction at 30°C using safe CO surrogates (formic acid/acetic anhydride), significantly improving operational safety and substrate compatibility.
Q: What is the substrate scope for the R1, R2, and R3 groups in this synthesis?
A: The method demonstrates broad functional group tolerance. R1 can be alkyl or substituted aryl (e.g., methyl, chloro, trifluoromethyl); R2 accommodates H, alkyl, halogens, or CF3; and R3 allows for alkyl, phenyl, or benzyl groups, enabling diverse library generation for drug discovery.
Q: Is this synthesis protocol suitable for industrial scale-up?
A: Yes, the patent explicitly mentions successful expansion to gram-scale reactions. The use of commercially available starting materials, simple post-treatment (filtration and chromatography), and mild temperature conditions (30°C) make it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to rise in the development of next-generation therapeutics, partnering with an experienced CDMO is essential for navigating the complexities of process chemistry. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from initial discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carbonyl-bridged biheterocyclic compounds delivered meets the highest industry standards for quality and consistency. We understand the critical nature of timeline adherence in drug development and are committed to providing a seamless supply experience.
We invite you to engage with our technical team to explore how this innovative Pd-catalyzed methodology can be tailored to your specific synthetic needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient process. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Contact our technical procurement team today to discuss your requirements and secure a reliable supply of high-quality intermediates for your upcoming projects.
