Advanced Catalytic Route for Pyrano[2,3-b]indol-2-one Production: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112961166A, which discloses a highly efficient catalytic preparation method for pyrano[2,3-b]indol-2-one compounds. This specific fused heterocyclic skeleton is of immense theoretical and practical importance due to its prevalence in natural products and its potent biological activities. The patent introduces a novel one-pot reaction strategy that couples substituted isatin, phosphite esters, and cyclopropenones using a specialized silicon-amino rare earth compound as the catalyst. This technological advancement represents a paradigm shift from traditional multi-step syntheses, offering a streamlined approach that significantly enhances reaction efficiency while maintaining exceptional product quality. For R&D directors and process chemists, this development opens new avenues for accessing high-purity intermediates with reduced operational complexity.
![General reaction scheme for the synthesis of pyrano[2,3-b]indol-2-one compounds using rare-earth catalysis](/insights/img/pyrano-indol-one-catalytic-synthesis-pharma-supplier-20260308130002-05.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrano[2,3-b]indol-2-one framework has been fraught with synthetic challenges that hinder efficient commercial production. Prior art methodologies often rely on cumbersome multi-step sequences that suffer from low overall yields and poor atom economy. For instance, earlier routes involved the reaction of michelia acid derivatives with N-(2-iodophenyl)propynamide, requiring three distinct reaction steps under stringent conditions. Another common approach utilized the Mukaiyama reagent to couple 2-hydroxy-3-acetylindole derivatives with phenylacetic acid, a process that not only generates significant chemical waste but also necessitates the handling of moisture-sensitive reagents. Furthermore, some existing methods require high-temperature conditions that can lead to substrate decomposition or the formation of difficult-to-remove impurities. These conventional pathways often depend on noble metal catalysts or stoichiometric amounts of strong bases, which escalate raw material costs and complicate the purification process due to heavy metal residue concerns.
![Comparison of prior art synthesis routes for pyrano[2,3-b]indol-2-one skeletons](/insights/img/pyrano-indol-one-catalytic-synthesis-pharma-supplier-20260308130002-014.png)
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112961166A introduces a revolutionary one-pot catalytic system that dramatically simplifies the synthetic landscape. By employing a silicon-amino rare earth compound, specifically [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3, the reaction achieves a direct convergence of three simple building blocks: substituted isatin, diethyl phosphite, and cyclopropenone. This novel approach operates under relatively mild thermal conditions, typically around 110°C in toluene, eliminating the need for extreme temperatures or cryogenic environments. The use of a lanthanide-based catalyst, particularly when Ln is Lanthanum, provides superior Lewis acidity that activates the substrates effectively without the toxicity associated with heavy transition metals. This methodology not only boosts the yield of the target product to impressive levels, reaching up to 95%, but also ensures a cleaner reaction profile. The simplicity of adding all components into a single vessel reduces labor costs and minimizes the risk of material loss during intermediate isolation steps.
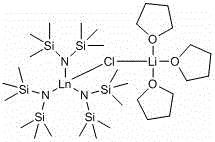
Mechanistic Insights into Rare-Earth Catalyzed Cyclization
The efficacy of this transformation lies in the unique electronic properties of the silicon-amino rare earth catalyst. The central lanthanide ion acts as a potent Lewis acid, coordinating with the carbonyl oxygen of the isatin and the phosphorus center of the phosphite ester. This dual activation lowers the energy barrier for the nucleophilic attack and subsequent cyclization steps. Unlike traditional Brønsted acid or base catalysis, which can lead to side reactions such as polymerization or hydrolysis, the rare earth center provides a controlled coordination environment that directs the reaction pathway selectively towards the desired pyrano-fused product. The bridging chloride and the bulky trimethylsilyl amino ligands create a steric environment that prevents catalyst deactivation and promotes turnover. This mechanistic precision is crucial for maintaining high selectivity, ensuring that the complex polycyclic structure is formed with minimal byproduct generation. The stability of the catalyst in organic solvents like toluene further contributes to the robustness of the process, allowing for consistent performance across different batches.
From an impurity control perspective, this catalytic mechanism offers distinct advantages for pharmaceutical manufacturing. The absence of noble metals means that the final product is free from toxic metal residues that often require expensive scavenging resins or complex extraction protocols to remove. The one-pot nature of the reaction limits the exposure of reactive intermediates to the external environment, thereby reducing the formation of degradation products. Furthermore, the reaction conditions are optimized to favor the thermodynamic product, minimizing the presence of regioisomers or stereoisomers that could complicate downstream crystallization. The use of diethyl phosphite as a reactant also ensures that any unreacted starting materials are relatively polar and easily separated from the less polar product during the aqueous workup. This inherent cleanliness of the reaction profile translates directly into higher purity specifications for the final API intermediate, reducing the burden on quality control laboratories.
How to Synthesize Pyrano[2,3-b]indol-2-one Efficiently
The operational simplicity of this patented process makes it highly attractive for both laboratory scale-up and industrial implementation. The procedure begins with the preparation of the reaction vessel under strictly anhydrous and oxygen-free conditions, typically using an inert gas like argon to protect the sensitive catalyst. The silicon-amino rare earth catalyst is introduced first, followed by the precise addition of diethyl phosphite and the substituted isatin derivative in a toluene solvent system. After a brief period of stirring at room temperature to ensure homogeneity, the cyclopropenone component is added, initiating the cascade reaction. The mixture is then heated to 110°C, where it is maintained for approximately 1.5 hours to drive the conversion to completion. Upon cooling, the reaction is quenched with water, and the organic layer is separated, dried, and concentrated. The crude product is then purified using standard flash column chromatography to yield the high-purity target compound. For a detailed breakdown of the standardized operating procedures and safety parameters, please refer to the technical guide below.
- Under anhydrous and oxygen-free conditions, mix the silicon-amino rare earth catalyst, substituted isatin, and diethyl phosphite in toluene.
- Add 2,3-diphenylcyclopropenone to the mixture and heat the reaction system to 110°C for approximately 1.5 hours.
- Quench the reaction with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The shift away from noble metal catalysts and stoichiometric coupling reagents represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By utilizing earth-abundant rare earth metals instead of precious metals like palladium or rhodium, the raw material cost base is drastically lowered, insulating the supply chain from the volatile price fluctuations often seen in the precious metals market. Additionally, the elimination of expensive reagents like the Mukaiyama reagent further compresses the bill of materials. The simplified one-pot process reduces the number of unit operations required, which in turn lowers utility consumption, labor hours, and equipment occupancy time. These factors combine to create a more economically resilient production model that can withstand market pressures while maintaining healthy margins.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of costly transition metal catalysts and the reduction of waste disposal costs associated with stoichiometric reagents. The high catalytic efficiency means that only small amounts of the rare earth complex are needed to drive the reaction, maximizing the return on investment for every kilogram of catalyst purchased. Furthermore, the use of common solvents like toluene and simple workup procedures minimizes the need for specialized waste treatment facilities. The overall process intensity is lower, meaning less energy is consumed per kilogram of product produced, contributing to a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted isatins and diethyl phosphite ensures a stable and secure supply chain. These commodities are produced by multiple global suppliers, reducing the risk of single-source bottlenecks that can disrupt production schedules. The robustness of the reaction conditions allows for flexible manufacturing planning, as the process is less sensitive to minor variations in environmental conditions compared to air-sensitive organometallic reactions. This reliability translates into shorter lead times for high-purity pharmaceutical intermediates, enabling faster response to customer demand and more agile inventory management strategies for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern green chemistry principles. The absence of heavy metal contamination simplifies the regulatory approval process for new drug applications, as residual metal limits are easier to meet. The process generates less hazardous waste, reducing the environmental burden and associated disposal fees. The scalability of the one-pot reaction is excellent, as heat transfer and mixing requirements are straightforward in standard stainless steel reactors. This ease of scale-up facilitates the commercial production of complex heterocyclic intermediates without the need for specialized high-pressure or cryogenic equipment, ensuring a smooth transition from pilot plant to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare-earth catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: What are the primary advantages of the silicon-amino rare earth catalyst over traditional methods?
A: The silicon-amino rare earth catalyst eliminates the need for expensive noble metals and harsh stoichiometric reagents like the Mukaiyama reagent. It enables a direct one-pot condensation with high atom economy and simplified downstream purification.
Q: Is this catalytic process suitable for large-scale commercial production?
A: Yes, the process utilizes robust reaction conditions (110°C in toluene) and commercially available starting materials. The absence of sensitive transition metals and the high yield (up to 95%) make it highly scalable for industrial manufacturing.
Q: What is the substrate scope for the isatin component in this reaction?
A: The method demonstrates excellent tolerance for various substituents on the isatin ring, including electron-withdrawing groups like fluoro, chloro, and bromo, as well as electron-donating groups like methyl and methoxy, ensuring versatility for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano[2,3-b]indol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN112961166A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped to handle sensitive rare-earth catalysis under strictly controlled anhydrous conditions, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and commitment to process excellence, we are uniquely positioned to deliver high-purity pyrano[2,3-b]indol-2-one derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthetic route for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing quotes. Let us help you optimize your supply chain and accelerate your path to market with our superior manufacturing capabilities.
