Advanced Metal-Free Manufacturing of Pyrano[2,3-b]indol-2-one Scaffolds for Global Supply Chains
Introduction to Next-Generation Heterocyclic Synthesis
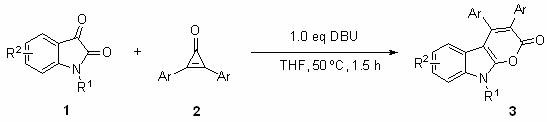
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective synthetic methodologies. Patent CN113651827B introduces a groundbreaking approach to the preparation of pyrano[2,3-b]indol-2-ones, a privileged scaffold found in numerous bioactive molecules and potential drug candidates. This innovation marks a pivotal shift away from traditional transition-metal catalysis towards a streamlined organocatalytic process utilizing 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). For global supply chain leaders and R&D directors, this technology represents not just a chemical curiosity, but a robust solution for producing high-purity heterocyclic intermediates with exceptional atom economy. By leveraging this metal-free protocol, manufacturers can address critical regulatory concerns regarding heavy metal residues while simultaneously optimizing production throughput and reducing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrano[2,3-b]indol-2-one core has relied heavily on the use of silicon-amino rare earth compounds or other transition metal catalysts, often requiring loadings as high as 10 mol%. These conventional pathways present substantial logistical and economic challenges for large-scale operations. The reliance on scarce rare-earth elements introduces volatility into the raw material supply chain, subjecting procurement teams to unpredictable pricing fluctuations and geopolitical supply risks. Furthermore, the presence of metal catalysts necessitates rigorous downstream purification steps, including the use of expensive metal scavengers and extensive chromatographic processing, to meet the stringent ppm-level limits imposed by international pharmacopoeias. This additional processing not only inflates the cost of goods sold (COGS) but also extends the overall manufacturing cycle time, creating bottlenecks that hinder rapid scale-up and market entry for new therapeutic candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113651827B utilizes DBU as a simple, commercially abundant organic base to drive the cyclization between isatin derivatives and cyclopropenone compounds. This novel approach completely obviates the need for any metal catalyst, thereby fundamentally simplifying the reaction workflow and post-reaction workup. The process operates under remarkably mild conditions, typically requiring only 50°C in tetrahydrofuran (THF), which significantly lowers energy consumption compared to high-temperature reflux protocols. As demonstrated in the comparative data, switching from ineffective bases like triethylamine or pyridine to DBU results in a dramatic improvement in conversion efficiency, validating DBU as the superior promoter for this transformation. This shift to organocatalysis aligns perfectly with modern green chemistry principles, offering a sustainable pathway that reduces waste generation and enhances the overall safety profile of the manufacturing process.

Mechanistic Insights into DBU-Mediated Cyclization
The efficacy of this metal-free synthesis lies in the unique ability of DBU to act as a potent non-nucleophilic base that facilitates the activation of the nucleophilic sites on the isatin ring without coordinating to the substrate in a way that deactivates the catalyst. The reaction mechanism likely involves the initial deprotonation or activation of the isatin nitrogen or adjacent carbon, followed by a nucleophilic attack on the strained cyclopropenone ring. The release of ring strain in the cyclopropenone provides a strong thermodynamic driving force for the formation of the new carbon-carbon and carbon-oxygen bonds that construct the fused pyrano-indole system. This cascade proceeds with high regioselectivity and stereocontrol, ensuring that the desired pyrano[2,3-b]indol-2-one isomer is formed exclusively, minimizing the formation of structural isomers that would complicate purification. The robustness of this mechanism is evidenced by its tolerance to a wide variety of electronic environments on the aromatic rings, allowing for the successful incorporation of electron-withdrawing groups like nitro and halogens as well as electron-donating groups like methoxy and methyl.
From an impurity control perspective, the absence of metal species is a transformative advantage for pharmaceutical quality assurance. Traditional metal-catalyzed routes often suffer from the formation of metal-organic complexes that are difficult to remove and can catalyze degradation pathways during long-term storage of the API. By eliminating the metal source entirely, this DBU-mediated process inherently produces a cleaner crude reaction mixture, reducing the burden on analytical laboratories to detect and quantify trace metal impurities. This simplifies the validation of the cleaning process and reduces the risk of batch rejection due to out-of-specification metal content. Furthermore, the mild reaction conditions prevent thermal degradation of sensitive functional groups, preserving the integrity of complex substituents that might be essential for the biological activity of the final drug molecule, thus ensuring a consistent and high-quality impurity profile across different production batches.
How to Synthesize Pyrano[2,3-b]indol-2-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized parameters identified during the patent's development phase to ensure maximum yield and reproducibility. The protocol is designed to be operationally simple, involving the sequential addition of readily available starting materials into a standard reaction vessel under ambient atmosphere, which removes the need for expensive inert gas manifolds or glovebox techniques. The key to success lies in maintaining the specific molar ratios and temperature controls that drive the equilibrium towards the product while minimizing side reactions. For detailed operational guidance, the standardized synthesis steps are outlined below, reflecting the optimal conditions established for the model substrate N-ethylisatin.
- Combine N-substituted isatin (0.24 mmol) and diphenylcyclopropenone (0.48 mmol) in a reaction vessel.
- Add 1.0 equivalent of DBU and 0.5 mL of tetrahydrofuran (THF) as the solvent.
- Stir the mixture at 50°C for 1.5 hours, then concentrate and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DBU-mediated technology offers compelling strategic advantages that extend far beyond simple chemical yield. The transition from a metal-dependent process to an organocatalytic one fundamentally alters the cost structure and risk profile of the supply chain. By removing the dependency on critical raw materials like rare-earth metals, companies can insulate themselves from supply shocks and price volatility, ensuring a more stable and predictable cost base for long-term production contracts. Additionally, the simplification of the downstream processing workflow translates directly into reduced operational expenditures, as fewer unit operations are required to bring the material to specification. This efficiency gain allows for faster turnaround times between batches, enhancing the overall agility of the manufacturing facility to respond to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive rare-earth catalysts and the associated metal scavenging agents leads to a substantial reduction in direct material costs. Without the need for complex metal removal steps, the consumption of solvents and chromatography media is drastically simplified, lowering waste disposal costs and extending the life of purification columns. The use of DBU, a commodity chemical, ensures that catalyst costs remain negligible compared to specialized organometallic complexes, contributing to a leaner and more competitive cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity rare-earth catalysts can often be a bottleneck due to limited global suppliers and stringent export regulations. By switching to DBU and common isatin derivatives, the supply chain becomes significantly more resilient and diversified. The availability of these reagents from multiple global vendors reduces the risk of single-source dependency, ensuring continuous production capability even during periods of regional disruption. This reliability is crucial for maintaining the continuity of supply for critical pharmaceutical ingredients, safeguarding against delays that could impact patient access to medications.
- Scalability and Environmental Compliance: The mild reaction conditions of 50°C and the use of standard solvents like THF make this process highly scalable from gram to multi-ton quantities without requiring specialized high-pressure reactors. The reduced environmental footprint, characterized by lower energy usage and the absence of toxic heavy metal waste, simplifies regulatory compliance and permitting processes. This alignment with green chemistry standards not only reduces the environmental liability of the manufacturing site but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free synthesis route. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this methodology into their existing manufacturing portfolios.
Q: Why is DBU preferred over traditional metal catalysts for this synthesis?
A: DBU eliminates the need for expensive and toxic rare-earth metal catalysts, simplifying purification and ensuring the final product is free from heavy metal residues, which is critical for pharmaceutical applications.
Q: What is the typical yield and purity profile of this method?
A: The process demonstrates excellent efficiency with isolated yields frequently exceeding 90% for various substrates, and the absence of metal catalysts inherently supports a cleaner impurity profile suitable for GMP manufacturing.
Q: Can this process be scaled for industrial production?
A: Yes, the mild reaction conditions (50°C) and short reaction time (1.5 hours) combined with the use of common solvents like THF make this protocol highly amenable to commercial scale-up without specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano[2,3-b]indol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the DBU-mediated pathway described in CN113651827B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyrano[2,3-b]indol-2-one delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced metal-free technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, high-quality supply of these valuable heterocyclic building blocks for your drug development pipeline.
