Advanced Catalyst-Free Synthesis of Pyrano[2,3-b]indol-2-ones for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Heterocyclic Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving towards greener, more efficient, and safer synthetic methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN113651827A, which discloses a novel method for preparing pyrano[2,3-b]indol-2-one derivatives. These fused heterocyclic scaffolds are of immense interest in medicinal chemistry due to their potential biological activities and structural complexity. Traditionally, constructing such intricate frameworks often required harsh conditions or expensive transition metal catalysts that posed purification challenges. However, this new technical disclosure presents a paradigm shift by utilizing a simple organic base, DBU (1,8-diazabicycloundec-7-ene), to drive the cyclization between isatin compounds and cyclopropenone compounds. This approach not only simplifies the operational procedure but also aligns perfectly with the modern industry's demand for metal-free processes, ensuring higher purity profiles essential for downstream drug development.
For R&D directors and process chemists, the implication of this patent is profound. It offers a robust alternative to previous methods that relied on silicon amino rare earth compounds or phosphite esters. By eliminating the need for 10 mol% of specialized catalysts, the new route reduces the chemical footprint and simplifies the post-reaction workup. The versatility of this method is demonstrated through its compatibility with a broad spectrum of substrates, accommodating various electronic and steric environments on both the isatin and cyclopropenone moieties. This flexibility makes it a highly attractive candidate for library synthesis and the rapid generation of analogues for structure-activity relationship (SAR) studies, positioning it as a valuable tool for reliable pharma intermediate supplier networks aiming to diversify their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of pyrano[2,3-b]indol-2-one frameworks often depended heavily on transition metal catalysis or complex Lewis acid systems. For instance, previous iterations of this chemistry utilized silicon amino rare earth compounds as catalysts, typically requiring loadings around 10 mol%. While effective, these metal-based systems introduce significant downstream processing burdens. The removal of trace metal residues to meet stringent pharmaceutical specifications (often in the ppm range) necessitates additional purification steps, such as treatment with scavengers or repeated recrystallizations, which inevitably erode overall yield and increase production costs. Furthermore, the use of phosphite esters and amine additives in conjunction with these metals adds layers of complexity to the reaction mixture, potentially leading to difficult-to-remove impurities that complicate the impurity profile of the final active pharmaceutical ingredient (API).
The Novel Approach
The methodology outlined in CN113651827A circumvents these issues by employing a metal-free, organocatalytic strategy driven by DBU. This base-promoted reaction proceeds efficiently in common organic solvents like tetrahydrofuran (THF) under mild thermal conditions. The elimination of metal catalysts is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it removes the capital expenditure associated with precious metals and the operational expenditure linked to their removal. The reaction demonstrates exceptional atom economy, directly coupling the isatin and cyclopropenone precursors with minimal waste generation. As illustrated in the reaction scheme below, the transformation is direct and high-yielding, converting simple starting materials into complex fused ring systems with remarkable efficiency.
![Reaction scheme showing DBU-promoted synthesis of pyrano[2,3-b]indol-2-one from isatin and cyclopropenone](/insights/img/pyrano-indol-one-synthesis-pharma-supplier-20260303114120-04.png)
Moreover, the operational simplicity cannot be overstated. The reaction typically completes within 1.5 hours at 50°C, a temperature easily maintained in standard glass-lined reactors without the need for cryogenic cooling or high-pressure equipment. This mildness enhances the safety profile of the process, reducing the risk of thermal runaways often associated with more exothermic metal-catalyzed couplings. For supply chain managers, this translates to shorter batch cycles and higher throughput capacity, allowing facilities to respond more agilely to market demands without compromising on quality or safety standards.
Mechanistic Insights into DBU-Promoted Cyclization
The success of this synthesis hinges on the unique reactivity of DBU as a non-nucleophilic, sterically hindered base. In the proposed mechanism, DBU likely facilitates the deprotonation or activation of the nucleophilic sites on the isatin or cyclopropenone species, initiating a cascade of bond-forming events that lead to the fused pyrano-indole core. Unlike Lewis acids which activate electrophiles by coordinating to lone pairs, DBU operates through Brønsted basicity, generating reactive intermediates that undergo rapid cyclization. This mechanistic pathway avoids the formation of stable metal-complexes that can sometimes sequester the product or inhibit turnover. The result is a clean reaction profile where the primary byproduct is simply the protonated form of the base, which is easily removed during aqueous workup or chromatography.
Substrate tolerance is another critical aspect of this mechanism. The reaction accommodates a wide variety of substituents on the isatin ring, including electron-withdrawing groups like nitro, trifluoromethyl, and halogens (fluorine, chlorine, bromine), as well as electron-donating groups like methyl and methoxy. This suggests that the rate-determining step is not overly sensitive to the electronic density of the aromatic system, providing a robust platform for synthesizing diverse derivatives. The general structure of the isatin substrates is shown below, highlighting the variability at the R1 and R2 positions which can be tuned to modulate the physicochemical properties of the final molecule.
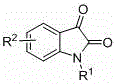
Furthermore, the cyclopropenone component, which serves as the three-carbon synthon for the pyrone ring, also exhibits broad compatibility. Aryl groups with varying electronic properties, such as phenyl, 4-methylphenyl, 4-fluorophenyl, and 4-chlorophenyl, participate effectively in the reaction. This breadth of scope ensures that the process is not limited to a narrow set of idealized substrates but is applicable to real-world drug discovery campaigns where specific substitution patterns are required for potency or metabolic stability. The ability to handle such diversity without modifying the core reaction conditions underscores the reliability of this synthetic route for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Pyrano[2,3-b]indol-2-ones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric and environmental parameters to maximize yield and purity. The protocol involves dissolving the N-substituted isatin and the diarylcyclopropenone in anhydrous THF, followed by the addition of DBU. The mixture is then heated to promote the cyclization. While the patent details extensive optimization studies regarding solvent, temperature, and base loading, the core procedure remains straightforward and scalable. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Combine N-substituted isatin compound and diarylcyclopropenone compound in tetrahydrofuran (THF) solvent.
- Add 1.0 equivalent of DBU (1,8-diazabicycloundec-7-ene) relative to the isatin substrate to the reaction mixture.
- Stir the reaction at 50°C for approximately 1.5 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this DBU-promoted methodology offers substantial strategic advantages for procurement and supply chain operations. The primary driver of value is the drastic simplification of the supply chain for raw materials. By removing the dependency on specialized, often imported, transition metal catalysts, manufacturers can mitigate supply risks associated with geopolitical instability or mining shortages. The reagents used—isatins, cyclopropenones, and DBU—are commodity chemicals available from multiple global vendors, ensuring a competitive pricing environment and reducing the likelihood of single-source bottlenecks. This diversification of the supply base is crucial for maintaining continuity in the production of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts directly impacts the bill of materials (BOM) cost. More importantly, it removes the need for costly metal scavenging resins and the associated validation testing required to prove low metal content in the final API. This simplification of the downstream processing workflow leads to significant operational savings. Additionally, the high yields reported (often exceeding 90%) minimize raw material waste, further enhancing the economic viability of the process. The reduced number of unit operations also lowers energy consumption and labor costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable reagents like DBU and common solvents like THF ensures that production schedules are not disrupted by the degradation of sensitive catalysts. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, which is a common cause of batch failures in complex syntheses. This reliability allows for more accurate forecasting and inventory management, enabling supply chain heads to maintain leaner inventory levels while still meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: Scaling a metal-free process is inherently easier from a regulatory and environmental standpoint. The absence of heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing site. The mild reaction temperature of 50°C is energy-efficient and safe for large-scale reactors, avoiding the hazards associated with high-temperature or high-pressure processes. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: Does this synthesis method require transition metal catalysts?
A: No, the process described in patent CN113651827A explicitly avoids the use of metal catalysts, utilizing DBU as a promoter instead, which eliminates heavy metal contamination risks.
Q: What is the substrate scope for the isatin component?
A: The method tolerates a wide range of N-substituents including allyl, benzyl, ethyl, methyl, and propargyl groups, as well as various aromatic substitutions like halogens, nitro, and trifluoromethyl groups.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal results are achieved using THF as the solvent at 50°C for 1.5 hours with a stoichiometric amount (1.0 eq) of DBU, often yielding over 90% isolated product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano[2,3-b]indol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free synthetic routes like the one described in CN113651827A. As a leading CDMO partner, we possess the technical expertise to translate such innovative academic and patent discoveries into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale optimization to multi-ton supply. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of metal impurities and confirm structural integrity.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. Whether you require custom synthesis of specific isatin derivatives or full-scale production of the final pyrano-indole scaffold, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your timeline and reduce your overall cost of goods sold.
