Revolutionizing Caprolactam Production: One-Step Synthesis for Commercial Scale-Up
The chemical industry is constantly seeking methods to optimize the production of high-volume intermediates like caprolactam, a critical precursor for polyamide engineering plastics and pharmaceuticals. Patent CN103524415A introduces a groundbreaking one-step synthesis method that transforms cyclohexanol directly into caprolactam using hydrogen peroxide and hydroxylamine within a unified reaction system. This innovation addresses the longstanding inefficiencies of traditional multi-stage processes by integrating oxidation, oximation, and rearrangement stages into a single vessel, thereby drastically reducing the physical footprint and energy requirements of the manufacturing plant. By utilizing a specialized catalytic system comprising quaternary ammonium salt acidic ionic liquids and sodium tungstate dihydrate, this method achieves high conversion rates while minimizing the generation of hazardous waste streams. For R&D directors and supply chain leaders, this technology represents a pivotal shift towards greener, more cost-effective chemical manufacturing that aligns with modern sustainability goals and regulatory pressures. The ability to produce high-purity caprolactam through such a streamlined pathway offers substantial strategic advantages for companies looking to secure reliable supply chains for polyamide and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
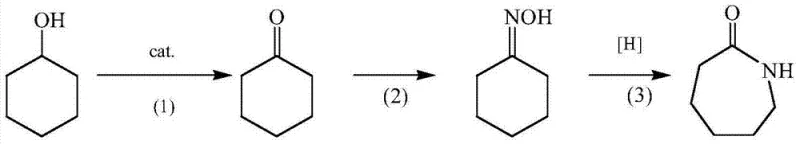
Traditional industrial production of caprolactam typically relies on the cyclohexanone-hydroxylamine method, which involves a fragmented series of discrete reaction steps that inherently increase operational complexity and cost. In the conventional workflow, cyclohexanol must first be oxidized to cyclohexanone in a separate unit, followed by a distinct oximation step to form cyclohexanone oxime, and finally a Beckmann rearrangement using fuming sulfuric acid to yield the final lactam. This multi-step approach necessitates extensive equipment arrays, including multiple reactors, separation columns, and storage tanks, which significantly inflate capital expenditure and maintenance overheads. Furthermore, the reliance on fuming sulfuric acid for the rearrangement stage generates large quantities of low-value ammonium sulfate as a by-product, creating substantial waste disposal challenges and environmental compliance burdens. The corrosive nature of the reagents also accelerates equipment degradation, leading to frequent downtime and increased replacement costs for critical infrastructure. These cumulative inefficiencies result in a long process flow with high energy consumption, making traditional methods increasingly uncompetitive in a market that demands leaner and cleaner production technologies.
The Novel Approach
In stark contrast to the fragmented legacy processes, the novel approach detailed in the patent consolidates the entire synthetic pathway into a single, cohesive operation that eliminates the need for intermediate isolation and transfer. By introducing cyclohexanol, hydrogen peroxide, and hydroxylamine directly into one reactor containing the ionic liquid and tungsten catalyst, the system facilitates a seamless transition from oxidation to oximation and finally to rearrangement without breaking the reaction stream. This integration not only simplifies the physical plant layout but also enhances thermal efficiency by retaining reaction heat within the system, thereby lowering the overall energy input required per unit of product. The use of hydrogen peroxide as a clean oxidant replaces harsher traditional oxidants, while the ionic liquid catalyst system obviates the need for large volumes of corrosive mineral acids, significantly reducing equipment stress and safety risks. This streamlined methodology allows for a more compact manufacturing footprint and a drastic reduction in auxiliary chemical consumption, positioning it as a superior alternative for modern chemical facilities aiming to maximize throughput while minimizing environmental impact.
Mechanistic Insights into Ionic Liquid-Catalyzed Cyclization
The core of this technological advancement lies in the sophisticated interplay between the quaternary ammonium salt acidic ionic liquid and the sodium tungstate dihydrate catalyst, which together create a highly active microenvironment for the triple-stage reaction. The ionic liquid serves a dual function, acting as both a phase transfer catalyst to improve the solubility of reactants and as an acidic promoter to drive the Beckmann rearrangement of the intermediate oxime. This dual functionality ensures that the cyclohexanol is efficiently oxidized to cyclohexanone by the hydrogen peroxide-tungsten complex, which then immediately reacts with the hydroxylamine present in the same medium to form the oxime. The acidic protons provided by the ionic liquid then catalyze the rearrangement of the oxime to caprolactam without requiring the addition of external strong acids like sulfuric acid. This mechanistic synergy prevents the accumulation of unstable intermediates and suppresses side reactions that typically lead to impurity formation, thereby enhancing the overall selectivity of the process. The tunable nature of the ionic liquid allows for precise control over the acidity and hydrophobicity of the reaction medium, enabling optimization for specific substrate concentrations and temperature profiles.
Impurity control is another critical aspect where this catalytic system excels, as the homogeneous nature of the reaction mixture promotes uniform conversion and minimizes localized hot spots that often cause degradation. In traditional processes, the separation of ammonium sulfate and the neutralization of excess acid can lead to product loss and the formation of difficult-to-remove salts, whereas this one-pot method generates significantly less inorganic waste. The catalyst system demonstrates remarkable stability, with experimental data indicating that the ionic liquid and tungsten species can be recovered and reused multiple times without significant loss in catalytic activity or selectivity. This recyclability is crucial for maintaining consistent product quality over long production runs, ensuring that the impurity profile remains within strict specifications required for pharmaceutical and high-grade polymer applications. By minimizing the formation of by-products such as ammonium sulfate and reducing the need for extensive downstream purification, the process inherently delivers a cleaner crude product that requires less energy-intensive refining steps.
How to Synthesize Caprolactam Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the reactants and the specific preparation of the ionic liquid catalyst to ensure optimal performance. The detailed standardized synthesis steps involve precise temperature control during the dropwise addition of hydrogen peroxide and the subsequent introduction of hydroxylamine sulfate to maintain the reaction equilibrium. Operators must adhere to the specified reaction times and extraction protocols using dichloromethane to isolate the final product with high recovery rates. For a comprehensive guide on the exact operational parameters and safety precautions, please refer to the standardized protocol provided below.
- Load cyclohexanol, sodium tungstate dihydrate, and acidic ionic liquid into a reactor equipped with condensation facilities.
- Add hydrogen peroxide dropwise at 80°C under normal pressure and maintain reaction for 30 to 600 minutes.
- Introduce hydroxylamine sulfate directly to the mixture, react at 70-100°C, and extract caprolactam using dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers profound benefits that directly address the pain points of procurement managers and supply chain directors focused on cost efficiency and reliability. The consolidation of three reaction stages into a single unit operation fundamentally alters the cost structure of caprolactam manufacturing by reducing the number of required processing units and associated utility consumption. This simplification translates into lower capital investment for new plants and reduced operational expenditures for existing facilities looking to retrofit their production lines. The elimination of fuming sulfuric acid not only lowers raw material costs but also removes the logistical complexities and safety hazards associated with transporting and storing highly corrosive chemicals. Furthermore, the reduction in waste generation simplifies compliance with environmental regulations, potentially lowering fees related to waste disposal and carbon emissions. These factors combine to create a more resilient supply chain that is less vulnerable to raw material price volatility and regulatory changes.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this manufacturing process is the elimination of expensive and hazardous reagents like fuming sulfuric acid, which traditionally account for a significant portion of raw material expenses. By replacing these with reusable ionic liquids and hydrogen peroxide, the process drastically lowers the recurring cost of goods sold while simultaneously reducing the need for expensive corrosion-resistant equipment. The ability to recycle the catalyst system multiple times further amortizes the cost of the catalytic materials over a larger production volume, enhancing the overall economic viability of the route. Additionally, the reduced energy consumption resulting from the integrated exothermic reactions lowers utility bills, contributing to a leaner cost base that improves margin potential in competitive markets.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the simplified process flow, which reduces the number of potential failure points and maintenance requirements within the production facility. The use of readily available raw materials such as cyclohexanol and hydrogen peroxide ensures that procurement teams can source inputs from a broad supplier base without relying on specialized or restricted chemicals. The robustness of the catalyst system against deactivation means that production campaigns can run for extended periods without interruption for catalyst regeneration or replacement. This continuity is vital for meeting strict delivery schedules and maintaining inventory levels for downstream customers who depend on just-in-time supply models. The reduced risk of equipment corrosion also minimizes unplanned downtime, ensuring a steady and predictable output of high-purity caprolactam.
- Scalability and Environmental Compliance: Scalability is inherently supported by the one-pot design, which allows for easier scale-up from pilot to commercial production without the need for complex re-engineering of multi-stage separation trains. The environmental compliance aspect is strengthened by the significant reduction in ammonium sulfate by-products, which are difficult to dispose of and often subject to strict regulatory limits. The cleaner process profile facilitates easier permitting for new facilities and reduces the risk of fines or shutdowns due to environmental non-compliance. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturer, making the product more attractive to environmentally conscious buyers and investors. The combination of scalability and compliance ensures long-term operational viability in a tightening regulatory landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this one-step caprolactam synthesis technology. These answers are derived directly from the patent data and are intended to provide clarity on the process capabilities and limitations for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new project developments.
Q: What are the primary advantages of this one-step caprolactam synthesis method?
A: The primary advantages include a significantly shortened process flow by integrating three reaction stages into one system, reduced energy consumption due to lower operating temperatures, and environmental benefits from eliminating oleum and reducing ammonium sulfate by-products.
Q: How does the ionic liquid catalyst system improve reaction efficiency?
A: The quaternary ammonium salt acidic ionic liquid works synergistically with Na2WO4·2H2O to provide both acid catalysis and phase transfer capabilities, enhancing cyclohexanol conversion rates up to 100% and stabilizing catalyst performance over multiple reuse cycles.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability by simplifying equipment requirements into a single reactor setup, reducing corrosion risks associated with traditional sulfuric acid methods, and facilitating easier separation and purification steps for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caprolactam Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, leveraging advanced synthetic routes like the one described in Patent CN103524415A to deliver superior value to our global partners. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards. We understand the critical nature of supply chain continuity and are equipped to handle complex synthesis requirements with the flexibility and speed that modern markets demand.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of adopting this technology for your production lines. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance metrics against your internal requirements. Let us partner with you to drive efficiency and sustainability in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
