Advanced Manufacturing of Imatinib Intermediates: A Technical Breakthrough for Global Supply Chains
The global pharmaceutical landscape is continuously evolving, with a heightened demand for efficient and scalable synthesis routes for critical oncology therapeutics. Patent CN102603710A introduces a transformative preparation method for a key Imatinib intermediate, specifically addressing the longstanding challenges of complex preparation techniques and multiple impurity profiles that have historically plagued the manufacturing of this life-saving tyrosine kinase inhibitor. This technical breakthrough offers a viable pathway for producing high-purity alkylsulfonyl-4-(3-pyridyl)-pyrimidine, a crucial building block in the synthesis of Imatinib mesylate, which is widely recognized for its efficacy in treating chronic myelogenous leukemia and gastrointestinal stromal tumors. By shifting away from hazardous reagents and embracing a more streamlined chemical architecture, this innovation not only enhances product quality but also aligns with modern green chemistry principles essential for sustainable industrial production. The structural integrity of the final API is paramount, and the intermediate produced via this method ensures a robust foundation for the final drug substance, minimizing the risk of genotoxic impurities that often arise from traditional halogenation processes. 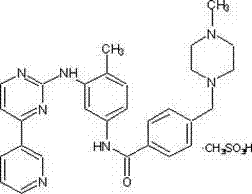
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imatinib intermediates has relied heavily on routes that utilize phosphorus oxychloride (POCl3) for the cyclization of the pyrimidine ring, a process that introduces significant environmental and operational hazards to the manufacturing facility. The use of POCl3 generates substantial amounts of acidic waste streams that require complex neutralization and disposal protocols, thereby inflating the overall operational expenditure and complicating regulatory compliance for environmental safety. Furthermore, conventional pathways often involve multiple protection and deprotection steps, which not only extend the production timeline but also cumulatively reduce the overall yield due to material losses at each stage of the synthesis. The presence of toxic phosphorous residues in the crude product necessitates rigorous purification steps, such as extensive chromatography or recrystallization, which are difficult to scale up efficiently without compromising throughput. Additionally, the harsh reaction conditions associated with traditional methods, including high temperatures and strong acidic environments, can lead to the formation of difficult-to-remove side products that negatively impact the purity profile of the final active pharmaceutical ingredient. 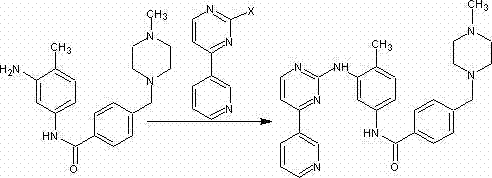
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes 3-acetylpyridine and N,N-dimethylformamidodimethylacetal as initial raw materials, initiating a condensation reaction that is both atom-economical and operationally simple. This strategy bypasses the need for hazardous halogenating agents entirely, instead employing a thiocarbamide cyclization reaction that proceeds under mild alkaline conditions to construct the heterocyclic core with high fidelity. The subsequent substitution reaction with iodomethane or other alkyl halides is highly selective, ensuring that the desired alkyl group is introduced precisely without affecting other sensitive functional groups on the molecule. Finally, the oxidation step utilizes hydrogen peroxide, a green oxidant that produces water as the only byproduct, thereby eliminating the heavy metal waste streams associated with traditional oxidation protocols. This cohesive sequence of reactions results in a significantly shortened process flow, reducing the number of unit operations required and facilitating a more robust and controllable manufacturing process suitable for multi-ton scale production.
Mechanistic Insights into Thiocarbamide Cyclization and Oxidation
The core of this synthetic innovation lies in the mechanistic efficiency of the thiocarbamide cyclization, which facilitates the formation of the pyrimidine ring through a nucleophilic attack that is kinetically favored under the specified alkaline conditions. The reaction mechanism involves the initial formation of an enamine intermediate from the condensation of 3-acetylpyridine, which then undergoes a cyclocondensation with thiocarbamide to yield the sulfhydryl-substituted pyrimidine derivative. This step is critical as it establishes the heterocyclic framework with high regioselectivity, minimizing the formation of isomeric impurities that are common in less controlled cyclization reactions. The use of bases such as sodium hydroxide or potassium hydroxide ensures that the reaction medium remains sufficiently nucleophilic to drive the cyclization to completion without requiring excessive thermal energy. Following the cyclization, the alkylation step introduces the necessary carbon chain, which is subsequently oxidized to the sulfone functionality, a transformation that is crucial for the biological activity of the final Imatinib molecule. 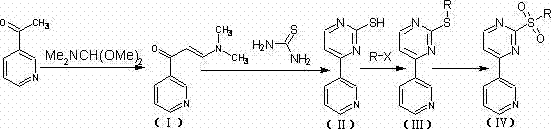
Impurity control is inherently built into this reaction design, as the mild conditions prevent the degradation of the sensitive pyridine and pyrimidine rings that often occurs under harsh acidic or high-temperature regimes. The oxidation of the sulfide to the sulfone using hydrogen peroxide is particularly selective, avoiding over-oxidation to the sulfone or other oxidative byproducts that could complicate downstream purification. The patent data indicates that the crude product purity can reach levels exceeding 99% after simple workup procedures, which is a testament to the cleanliness of the reaction pathway. This high level of chemical purity is essential for meeting the stringent regulatory requirements for pharmaceutical intermediates, where even trace levels of certain impurities can trigger extensive toxicological testing. By controlling the reaction stoichiometry and temperature precisely, manufacturers can ensure a consistent impurity profile batch after batch, thereby enhancing the reliability of the supply chain for downstream API synthesis.
How to Synthesize Alkylsulfonyl-4-(3-pyridyl)-pyrimidine Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and purity, starting with the precise control of the condensation temperature and the molar ratios of the initial reactants. The process begins with the reflux of 3-acetylpyridine with DMF-DMA in a suitable solvent like o-xylene, followed by the addition of thiocarbamide under alkaline conditions to form the cyclic intermediate. Subsequent alkylation with methyl iodide or other haloalkanes must be monitored to ensure complete conversion before proceeding to the final oxidation step with hydrogen peroxide in acetone. While the general chemical transformations are well-defined, the specific operational details regarding mixing rates, addition times, and quenching procedures are critical for industrial success.
- Condense 3-acetylpyridine with N,N-dimethylformamidodimethylacetal to form the enamine intermediate.
- Perform thiocarbamide cyclization under alkaline conditions to construct the pyrimidine ring.
- Execute alkyl halide substitution followed by hydrogen peroxide oxidation to yield the final sulfone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers profound advantages for procurement managers and supply chain directors who are tasked with optimizing costs and ensuring continuity of supply for critical oncology ingredients. The elimination of toxic reagents like POCl3 not only reduces the direct cost of raw materials but also significantly lowers the indirect costs associated with waste disposal, safety equipment, and regulatory compliance monitoring. By simplifying the process flow and reducing the number of synthetic steps, the overall production time is drastically shortened, allowing for faster turnaround times and more responsive inventory management. The use of cheap and accessible starting materials ensures that the supply chain is less vulnerable to fluctuations in the availability of specialized or exotic reagents, thereby enhancing the stability of the manufacturing schedule. Furthermore, the high yield and purity achieved through this method reduce the need for extensive reprocessing, leading to substantial cost savings in terms of solvent usage, energy consumption, and labor hours.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and toxic halogenating agents directly translates to a leaner cost structure, as the procurement of raw materials becomes more economical and the handling requirements are less stringent. The simplified workup procedures reduce the consumption of solvents and purification media, which are often significant cost drivers in fine chemical manufacturing. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold per kilogram. This economic efficiency allows for more competitive pricing strategies without compromising on the quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 3-acetylpyridine and hydrogen peroxide ensures that the supply chain is robust and less susceptible to disruptions caused by the scarcity of specialized reagents. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or equipment corrosion, leading to more predictable production schedules and reliable delivery timelines. This stability is crucial for downstream API manufacturers who depend on a steady flow of high-quality intermediates to maintain their own production schedules and meet market demand. The scalability of the process further ensures that supply can be ramped up quickly in response to increased market needs without the need for significant capital investment in new equipment.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this route, such as the use of hydrogen peroxide and the avoidance of heavy metals, make it highly scalable while maintaining strict adherence to environmental regulations. The reduction in hazardous waste generation simplifies the permitting process for manufacturing facilities and reduces the long-term liability associated with environmental compliance. This alignment with sustainability goals is increasingly important for pharmaceutical companies seeking to minimize their carbon footprint and enhance their corporate social responsibility profiles. The ability to scale this process from laboratory to commercial production without significant modification ensures a smooth technology transfer and rapid market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Imatinib intermediate synthesis pathway. These insights are derived directly from the patent data and are intended to provide clarity on the feasibility and advantages of adopting this technology for large-scale manufacturing. Understanding these details is essential for technical teams evaluating the potential for process optimization and cost reduction in their current supply chains.
Q: How does this new method improve impurity profiles compared to POCl3 routes?
A: The novel route avoids phosphorus oxychloride, eliminating phosphorous-containing impurities and reducing the complexity of downstream purification, resulting in purity levels exceeding 99%.
Q: Is the hydrogen peroxide oxidation step safe for large-scale production?
A: Yes, the reaction is conducted at mild temperatures (25-40°C) in common solvents like acetone, significantly reducing thermal runaway risks associated with traditional strong oxidants.
Q: What are the primary cost drivers reduced in this synthesis?
A: Costs are reduced by utilizing cheap, accessible raw materials like 3-acetylpyridine and eliminating expensive catalyst removal steps and toxic waste disposal fees.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our technical team is adept at adapting complex synthetic routes like the one described in CN102603710A to fit specific client requirements, maintaining stringent purity specifications and rigorous QC labs to guarantee product consistency. We understand the critical nature of oncology intermediates and are committed to delivering high-purity Imatinib intermediates that meet the exacting standards of the global pharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this synthesis, ensuring safety and quality at every stage of the manufacturing process.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain for maximum efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your production volume and quality requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate our capability to support your long-term manufacturing goals. Let us partner with you to optimize your supply chain and ensure the reliable delivery of high-quality pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
