Advanced Manufacturing of N-Phenyl-2-Pyrimidyl Amine Derivatives for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of critical oncology therapeutics, particularly tyrosine kinase inhibitors like Imatinib. Patent CN1900073A introduces a significant technological advancement in the synthesis of N-(2-methyl-5-nitro)phenyl-4-(3-pyridyl)pyrimidin-2-amine, a pivotal intermediate in the manufacturing of Imatinib mesylate. This innovation addresses longstanding challenges in the supply chain by replacing cumbersome multi-step sequences with a direct, acid-catalyzed condensation strategy. By leveraging readily available 4-aromatic heterocyclic-2-halogenated pyrimidines and substituted anilines, this method offers a streamlined approach that enhances overall process efficiency. For R&D directors and procurement specialists, understanding this shift is crucial as it represents a move towards more sustainable and cost-effective chemical manufacturing. The patent details a versatile protocol that not only improves yield but also simplifies the purification landscape, thereby reducing the burden on quality control laboratories.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
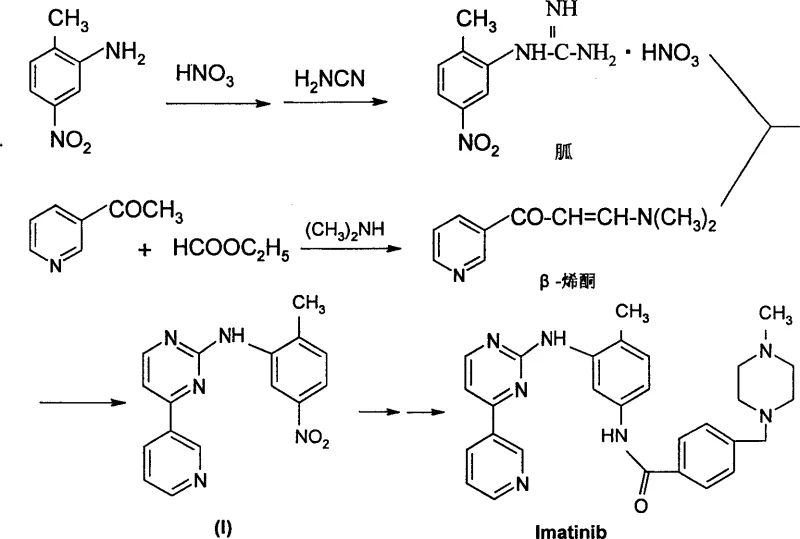
Historically, the synthesis of N-phenyl-2-pyrimidyl amine derivatives has relied on complex and labor-intensive pathways that pose significant hurdles for large-scale production. Traditional methods, as referenced in prior art such as US5521184, typically involve the initial preparation of guanidine nitrates and beta-keto enamine compounds from expensive and sometimes toxic starting materials like 5-nitro-o-toluidine and 3-acetylpyridine. These precursors must then undergo a cyclic condensation reaction to form the pyrimidine ring, a process that is inherently lengthy and prone to generating difficult-to-remove impurities. The reliance on such intricate sequences results in lower overall yields and higher operational costs due to the extensive purification steps required. Furthermore, the handling of specialized reagents increases safety risks and complicates waste management protocols, making these conventional routes less attractive for modern, high-volume pharmaceutical manufacturing environments.
The Novel Approach
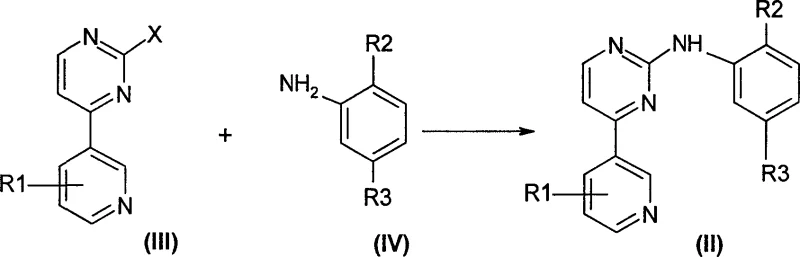
In stark contrast to the legacy methodologies, the process disclosed in CN1900073A utilizes a direct nucleophilic substitution reaction that dramatically simplifies the synthetic architecture. This novel approach employs 4-aromatic heterocyclic-2-halogenated pyrimidines, which can be easily sourced or prepared via established cross-coupling techniques, reacting them directly with substituted anilines in the presence of an acidic catalyst. This single-step condensation bypasses the need for constructing the pyrimidine ring from scratch during the final coupling stage, thereby reducing the total number of unit operations. The versatility of this method allows for the use of various halogens (chlorine, bromine, iodine) and a wide range of aniline derivatives, providing flexibility in process design. By eliminating the intermediate formation of unstable enaminones and guanidine salts, the new route ensures a cleaner reaction profile and facilitates easier isolation of the target product through standard extraction or crystallization techniques.
Mechanistic Insights into Acid-Catalyzed Nucleophilic Substitution
The core of this innovative synthesis lies in the activation of the halogenated pyrimidine ring by an acidic catalyst, which enhances the electrophilicity of the carbon atom at the 2-position. In the presence of strong organic acids like methanesulfonic acid or p-toluenesulfonic acid, or mineral acids such as hydrochloric acid, the nitrogen atom of the pyrimidine ring becomes protonated. This protonation withdraws electron density from the aromatic system, making the adjacent carbon bonded to the halogen leaving group significantly more susceptible to nucleophilic attack by the amino group of the substituted aniline. The reaction proceeds through a tetrahedral intermediate, followed by the elimination of the halide ion and restoration of aromaticity to yield the desired N-phenyl-2-pyrimidyl amine derivative. This mechanism is highly efficient because the acid catalyst not only activates the electrophile but also helps in managing the basicity of the aniline, preventing side reactions that could occur under neutral or basic conditions.
Controlling impurities in this reaction is achieved through careful selection of the reaction medium and temperature parameters. The patent specifies that the reaction can be conducted in a variety of solvents including anhydrous dioxane, ethanol, or acetonitrile, with temperatures ranging from 50°C to 100°C. Operating within this thermal window ensures sufficient energy for the substitution to proceed while minimizing thermal degradation of the sensitive pyrimidine moiety. Furthermore, the use of specific acidic catalysts allows for fine-tuning the reaction rate; for instance, methanesulfonic acid provides a balance of solubility and acidity that promotes high conversion without excessive charring or polymerization. Post-reaction workup involves simple neutralization and extraction, which effectively removes unreacted anilines and acid residues, resulting in a crude product of high purity that often requires only recrystallization to meet stringent pharmaceutical specifications.
How to Synthesize N-(2-methyl-5-nitro)phenyl-4-(3-pyridyl)pyrimidin-2-amine Efficiently
To implement this synthesis effectively, manufacturers should focus on the precise stoichiometry of the halogenated pyrimidine and the substituted aniline, typically employing a slight excess of the aniline to drive the equilibrium towards completion. The choice of solvent is critical; anhydrous conditions are generally preferred to prevent hydrolysis of the halogenated starting material, although the patent notes that aqueous mixtures can be tolerated depending on the catalyst system. The reaction is typically monitored by HPLC or TLC until the starting halogenated pyrimidine is fully consumed, ensuring maximum yield before proceeding to the workup phase. Detailed standardized operating procedures regarding addition rates, temperature ramping, and quenching protocols are essential for reproducibility and safety on a commercial scale.
- Prepare the reaction mixture by combining 4-aromatic heterocyclic-2-halogenated pyrimidine and substituted aniline in a suitable anhydrous organic solvent such as dioxane or ethanol.
- Add an acidic catalyst, such as methanesulfonic acid, p-toluenesulfonic acid, or hydrochloric acid, to facilitate the nucleophilic substitution reaction.
- Heat the reaction mixture to a temperature between 50°C and 100°C, typically under reflux conditions, until the conversion is complete, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial reduction of raw material costs, as the process utilizes commodity chemicals like substituted anilines and halogenated pyrimidines which are widely available in the global market compared to the specialized precursors required by older methods. This shift reduces dependency on niche suppliers and mitigates the risk of supply disruptions caused by the scarcity of complex intermediates. Additionally, the simplification of the synthetic sequence means fewer processing steps, which directly translates to lower labor costs, reduced energy consumption, and decreased solvent usage per kilogram of final product. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of meeting the high-volume demands of the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps required to build the pyrimidine ring in situ significantly lowers the cost of goods sold (COGS). By purchasing pre-formed halogenated pyrimidines, manufacturers avoid the capital expenditure and operational overhead associated with producing guanidine nitrates and beta-keto enamines. Furthermore, the use of inexpensive acid catalysts like hydrochloric acid or methanesulfonic acid replaces more costly reagents, and the simplified workup reduces the volume of waste solvents that require treatment or disposal. This leaner manufacturing model allows for competitive pricing strategies without compromising on profit margins, making it an attractive option for cost-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials enhances the robustness of the supply chain against geopolitical or logistical shocks. Substituted anilines and halogenated pyridines are produced by numerous chemical vendors worldwide, ensuring a diversified sourcing strategy that prevents bottlenecks. The shorter reaction time and simpler processing requirements also mean that production cycles are faster, allowing for quicker turnaround times from order to delivery. This agility is crucial for maintaining inventory levels and responding rapidly to fluctuations in market demand for Imatinib and its analogues, ensuring continuous availability for downstream drug formulation.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is superior due to its reduced waste generation and safer operating conditions. The avoidance of toxic cyanamide derivatives and the reduction in solvent intensity align with green chemistry principles, facilitating easier regulatory approval and compliance with increasingly strict environmental regulations. The reaction conditions are mild enough to be scaled from pilot plants to multi-ton reactors without significant engineering challenges, ensuring that the process remains stable and safe at commercial volumes. This scalability ensures that manufacturers can ramp up production quickly to meet global health needs while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in CN1900073A, providing clarity on the feasibility and advantages of the technology. Understanding these details is essential for technical teams evaluating the transition from legacy processes to this improved methodology.
Q: What are the primary advantages of this new condensation method over traditional routes?
A: The novel method described in CN1900073A eliminates the need for complex precursor synthesis like guanidine nitrates and beta-keto enamines, significantly shortening the reaction steps and utilizing commercially available, lower-cost starting materials.
Q: Which acidic catalysts are suitable for this synthesis?
A: The process is versatile and supports a range of acidic catalysts including mineral acids like hydrochloric acid and sulfuric acid, as well as organic acids such as methanesulfonic acid, p-toluenesulfonic acid, and trifluoroacetic acid.
Q: Is this process scalable for industrial production of Imatinib intermediates?
A: Yes, the method uses common solvents like dioxane and ethanol and operates at moderate temperatures (50-100°C), making it highly suitable for large-scale commercial manufacturing with simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable supply chains for life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition to this advanced synthesis method is seamless and effective. We are committed to delivering high-purity N-phenyl-2-pyrimidyl amine derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with a focus on safety, quality, and environmental stewardship, making us an ideal partner for your long-term manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for Imatinib production.
