Advanced Manufacturing of Vitamin A Acetate Intermediate C15 for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Vitamin A Acetate Intermediate C15 for Global Pharmaceutical Supply Chains
The global demand for Vitamin A and its derivatives, particularly Vitamin A acetate (CAS No. 127-47-9), continues to surge across pharmaceutical, nutraceutical, and feed additive sectors due to its critical role in visual health, immune function, and epithelial tissue maintenance. However, the supply chain for this essential nutrient has historically been constrained by complex, hazardous, and costly synthesis routes. A significant technological breakthrough is detailed in patent CN111484524A, which introduces a novel, highly efficient preparation method for the key C15 intermediate. This innovation addresses long-standing industry pain points by replacing dangerous acetylene chemistry and expensive beta-ionone precursors with a safer, more economical pathway involving sequential substitution and Wittig reactions. For procurement leaders and R&D directors seeking a reliable vitamin A acetate intermediate supplier, this patent represents a pivotal shift towards sustainable and scalable manufacturing capabilities.
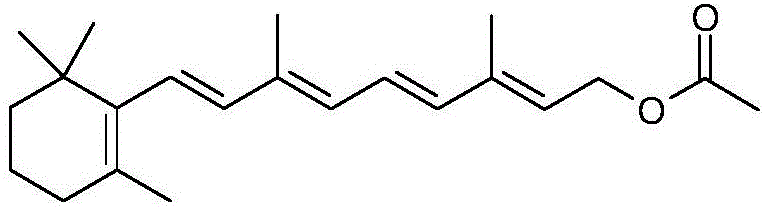
Vitamin A acetate is not merely a commodity chemical but a high-value active ingredient essential for treating conditions ranging from nyctalopia to various epithelial cancers. The structural complexity of the molecule, characterized by a conjugated polyene chain attached to a trimethylcyclohexenyl ring, necessitates precise stereochemical control during synthesis. Traditional methods often struggle with isomer separation and low overall yields, creating bottlenecks in the supply of high-purity pharmaceutical intermediates. The new methodology described in the patent leverages a strategic disconnection of the carbon chain, utilizing 1-halo-2-methyl-4-acetoxy-2-butylene as a foundational building block. This approach not only simplifies the synthetic logic but also aligns with modern green chemistry principles by minimizing waste and avoiding heavy metal catalysts, thereby offering a robust solution for cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Vitamin A acetate has relied on three primary routes, each fraught with significant technical and economic drawbacks that hinder optimal supply chain efficiency. The Roche method (C14 + C6 route) involves a Grignard reaction starting from beta-ionone, requiring six distinct steps including Darzens condensation and dehydrobromination, which results in cumulative yield losses and complex waste streams. Alternatively, the Rhone-Poulenc route depends on harsh high-vacuum molecular distillation to purify intermediates like C15 aldehyde and C18 ketone, a process that is energy-intensive and difficult to scale for large-volume production. Perhaps most critically, the BASF C15 + C5 route, while shorter, traditionally relies on ethynylation reactions using lithium amide and acetylene gas.
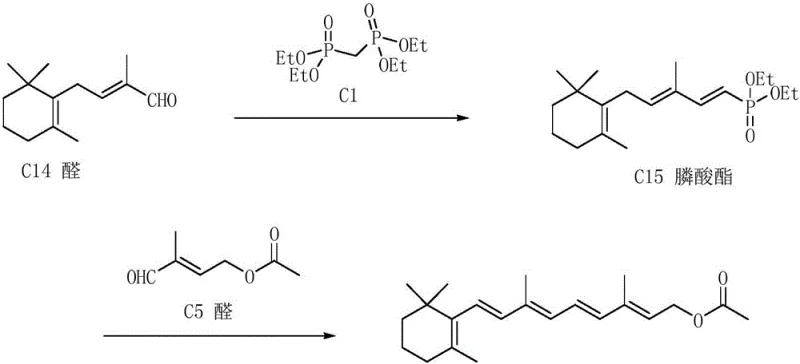
These conventional ethynylation processes demand stringent anhydrous conditions and ultra-low temperatures, posing severe safety risks and operational challenges for commercial scale-up of complex polymer additives and fine chemicals. Furthermore, the reliance on beta-ionone as a starting material ties the production cost to the volatile pricing of this terpene derivative. The generation of malodorous byproducts, such as dimethyl sulfide in thioylide methods, and the production of large volumes of wastewater in chloroacetate methods further exacerbate environmental compliance issues. For supply chain heads, these factors translate into unpredictable lead times, higher regulatory burdens, and increased total cost of ownership, making the search for alternative synthetic routes a strategic imperative.
The Novel Approach
The patented process outlined in CN111484524A fundamentally reengineers the synthesis of the C15 intermediate by bypassing the problematic acetylene and beta-ionone steps entirely. Instead, it employs a modular strategy starting from 1-halo-2-methyl-4-acetoxy-2-butylene, a readily available and stable raw material. The core innovation lies in a sequence of two nucleophilic substitution reactions (SN2) and two Wittig condensations, which are classical, high-yielding transformations well-suited for industrial application. By converting the halo-butene directly into a phosphorus-containing Wittig reagent precursor, the method achieves high atom economy and reaction selectivity without the need for cryogenic conditions or hazardous gases.
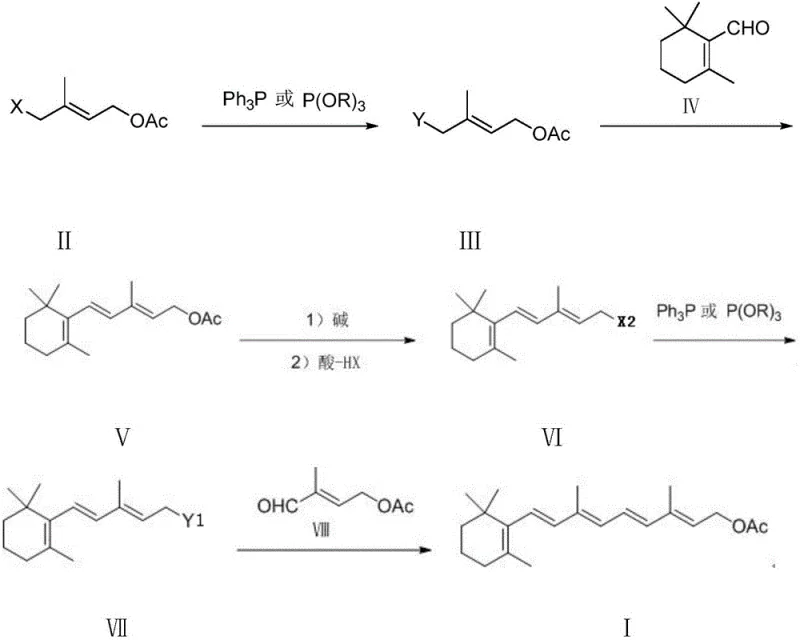
This novel approach offers a streamlined pathway where the C15 phosphorus-containing compound—whether a triphenylphosphonium salt or a phosphonic diester—is generated with exceptional purity. The process eliminates the need for complex post-treatment drying steps often required for phosphonates in older methods, as the reaction conditions are inherently more forgiving. The final coupling with 2-methyl-4-acetoxy-2-butenal (C5 aldehyde) proceeds under mild alkaline conditions to yield Vitamin A acetate directly. For a reliable agrochemical intermediate supplier or pharma partner, this translates to a drastically simplified operational workflow, reduced equipment corrosion, and a significant decrease in the environmental footprint associated with solvent recovery and waste neutralization.
Mechanistic Insights into Sequential Substitution and Wittig Condensation
The chemical elegance of this synthesis lies in its precise control over reaction kinetics and thermodynamics during the formation of the conjugated polyene system. The first critical step involves the nucleophilic substitution of the allylic halide (Formula II) with triphenylphosphine or a phosphite triester. This SN2 reaction is highly favorable due to the activation of the allylic position, proceeding efficiently at moderate temperatures between 20°C and 140°C depending on the phosphine source. The resulting phosphonium salt or phosphonate (Formula III) serves as a stabilized ylide precursor, ready for chain extension. Unlike unstable organometallic reagents, these phosphorus intermediates are solid, stable, and easy to handle, which significantly reduces the risk of decomposition during storage or transport.
Subsequent reaction with beta-cyclocitral (Formula IV) under basic conditions generates the extended carbon skeleton via a Wittig olefination. The patent specifies the use of bases like sodium ethoxide or potassium tert-butoxide in polar aprotic solvents such as DMF or toluene, ensuring rapid deprotonation and ylide formation. The stereoselectivity of this step is crucial for maintaining the all-trans configuration required for biological activity. Following this, the acetate protecting group is hydrolyzed under mild alkaline conditions and subsequently converted back to a halide (Formula VI) through acidification. This 'protect-deprotect-halogenate' sequence might seem counterintuitive but allows for the precise installation of the leaving group necessary for the final Wittig salt formation, ensuring that the final C15 intermediate (Formula VII) is structurally perfect for the final coupling.
How to Synthesize Vitamin A Acetate Intermediate C15 Efficiently
The synthesis protocol described in the patent provides a clear, reproducible roadmap for manufacturing the C15 intermediate with industrial-grade consistency. The process is divided into four distinct operational units: the initial phosphination, the first chain extension via Wittig reaction, the functional group interconversion (hydrolysis and halogenation), and the final phosphination to create the active coupling agent. Each step is optimized for yield and purity, with specific attention paid to temperature control and stoichiometry to minimize side reactions. The detailed experimental examples in the patent demonstrate that liquid phase purities exceeding 99% are achievable without resorting to chromatographic purification, relying instead on crystallization and distillation.
- Perform a substitution reaction on 1-halo-2-methyl-4-acetoxy-2-butene with triphenylphosphine or phosphite triester to form the initial Wittig reagent precursor.
- React the resulting phosphorus compound with beta-cyclocitral under alkaline conditions followed by acidification to generate the acetoxy-pentadiene intermediate.
- Hydrolyze the ester group of the intermediate under basic conditions and acidify to obtain the corresponding allylic halide.
- Execute a final substitution reaction with triphenylphosphine or phosphite triester to yield the target Vitamin A acetate intermediate C15.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The shift away from beta-ionone and acetylene decouples the production cost from volatile natural product markets and hazardous gas supply chains. By utilizing stable, commodity-grade halo-butene starting materials, manufacturers can secure long-term supply contracts at predictable price points, effectively insulating the business from raw material spikes. Furthermore, the elimination of ultra-low temperature requirements (-78°C or lower) reduces energy consumption and allows for the use of standard stainless steel reactors rather than specialized cryogenic vessels, leading to substantial capital expenditure savings.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, driven primarily by the removal of expensive reagents and the simplification of downstream processing. Traditional routes often require high-vacuum molecular distillation to separate closely related isomers, a process that is both energy-intensive and prone to product degradation. In contrast, this new method achieves high selectivity through chemical design, allowing for simpler work-up procedures like extraction and crystallization. The ability to recycle triphenylphosphine oxide byproducts back into triphenylphosphine further enhances the atom economy, reducing the effective cost of the phosphorus reagents. Additionally, the avoidance of lithium amide eliminates the need for specialized handling protocols and expensive quenching procedures, resulting in significantly reduced operational expenditures.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable vitamin A acetate intermediate supplier, and this process strengthens resilience by diversifying the raw material base. The reliance on bulk chemicals like triphenylphosphine and simple halo-alkenes ensures that production is not bottlenecked by the availability of niche terpenes. Moreover, the improved safety profile of the reaction—operating at ambient or moderately elevated temperatures without pyrophoric reagents—minimizes the risk of unplanned shutdowns due to safety incidents. This operational stability translates directly into more reliable delivery schedules for downstream API manufacturers, fostering stronger partnerships and long-term contractual agreements.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, the environmental advantages of this route become a key differentiator. The process generates significantly less wastewater compared to chloroacetate methods and avoids the release of noxious sulfur compounds associated with thioylide routes. The solvents used, such as acetonitrile, toluene, and alcohols, are readily recoverable and recyclable within a closed-loop system, aligning with green chemistry mandates. From a scalability perspective, the exothermic profiles of the reactions are manageable, allowing for safe scale-up from pilot plant to multi-ton commercial production without the need for complex engineering controls. This ease of scale-up ensures that supply can be rapidly ramped up to meet market surges without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel C15 synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN111484524A, offering a transparent view of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What are the primary safety advantages of this new C15 synthesis route compared to traditional methods?
A: Unlike traditional acetylene-based routes that require hazardous lithium amide and low-temperature operations, this patented method utilizes mild substitution and Wittig reactions at moderate temperatures (20-140°C), significantly enhancing operational safety and reducing the risk of thermal runaway.
Q: How does this process impact the overall cost of Vitamin A acetate manufacturing?
A: The process eliminates the need for expensive beta-ionone raw materials and avoids complex purification steps like high-vacuum molecular distillation. By using readily available halo-butene starting materials and achieving high atom economy, it substantially lowers raw material and processing costs.
Q: What purity levels can be expected from the C15 intermediate produced via this method?
A: The method demonstrates exceptional selectivity, yielding intermediates with liquid phase purity exceeding 99% in multiple examples. This high purity reduces the burden on downstream purification, ensuring consistent quality for the final Vitamin A acetate API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin A Acetate Intermediate Supplier
The technological advancements detailed in patent CN111484524A underscore the immense potential for optimizing the global supply of Vitamin A derivatives. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative laboratory chemistry into robust commercial reality requires deep expertise and state-of-the-art facilities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this new synthesis route are fully realized in practice. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize advanced HPLC and GC methodologies to verify every batch against the highest international standards.
We invite forward-thinking pharmaceutical and nutraceutical companies to collaborate with us on leveraging this next-generation synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, and discover how our optimized manufacturing processes can drive efficiency and reliability in your Vitamin A acetate supply chain.
