Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing privileged heterocyclic scaffolds, among which the indole nucleus stands out as a cornerstone structure found in numerous bioactive molecules. Patent CN112898192B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for N-acyl indole compounds. This technology leverages a palladium-catalyzed carbonylative cyclization strategy that transforms readily available 2-alkynylanilines and aryl iodides into complex N-acyl indoles in a streamlined process. The significance of this chemical transformation cannot be overstated, as N-acyl indoles serve as critical intermediates for a wide array of therapeutic agents, including anti-inflammatory, anti-tumor, and anti-viral drugs. By utilizing a solid carbon monoxide substitute rather than toxic gas, this invention addresses both safety concerns and operational complexity, marking a substantial leap forward for industrial applicability.
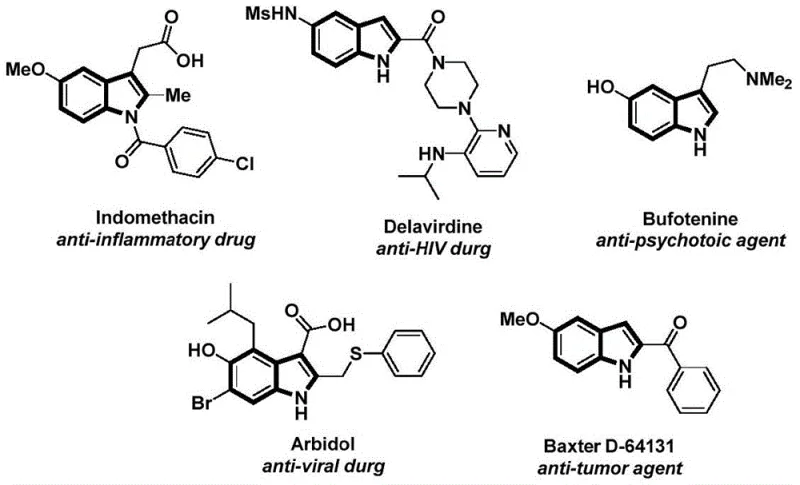
Furthermore, the versatility of this synthetic route allows for the introduction of diverse substituents at multiple positions on the indole ring, enabling medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship studies. The patent explicitly highlights the method's ability to operate under relatively mild conditions while maintaining high reaction efficiency, a crucial factor for scaling up production without compromising product quality. As a reliable pharmaceutical intermediate supplier, understanding such innovative pathways is essential for securing supply chains for next-generation active pharmaceutical ingredients (APIs). The integration of this technology into existing manufacturing frameworks promises to enhance the availability of high-purity indole derivatives, thereby supporting the broader goals of drug discovery and development pipelines globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing N-acyl indole frameworks often suffer from significant drawbacks that hinder their widespread adoption in large-scale manufacturing environments. Many classical methods rely on the direct use of carbon monoxide gas, which poses severe safety hazards due to its high toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that drastically increase capital expenditure. Additionally, conventional approaches frequently involve multi-step sequences where the indole ring is formed first, followed by a separate acylation step, leading to lower overall atom economy and increased waste generation. These fragmented processes often result in cumulative yield losses and necessitate extensive purification steps between stages, which drives up the cost of goods sold and extends the total production timeline. Moreover, older methodologies may exhibit poor functional group tolerance, limiting the structural diversity of the final products and forcing chemists to employ protecting group strategies that further complicate the synthesis.
The Novel Approach
In stark contrast, the methodology described in patent CN112898192B offers a transformative solution by integrating the carbonylation and cyclization events into a cohesive, one-pot sequence that utilizes a solid CO surrogate. This novel approach employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and convenient source of carbon monoxide, effectively eliminating the risks associated with handling gaseous CO while ensuring precise stoichiometric control over the carbonylation step. The reaction proceeds through a tandem mechanism where the initial palladium-catalyzed coupling forms an amide intermediate, which subsequently undergoes silver-mediated oxidative cyclization to yield the target N-acyl indole. This streamlined protocol not only simplifies the operational workflow but also significantly enhances the reaction efficiency, as evidenced by the high yields reported across a broad range of substrates. By reducing the number of unit operations and avoiding hazardous reagents, this method represents a paradigm shift towards greener and more sustainable chemical manufacturing practices.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The mechanistic pathway underpinning this synthesis is a sophisticated orchestration of organometallic steps initiated by the oxidative addition of a palladium(0) species into the carbon-iodine bond of the aryl iodide substrate. This generates a reactive aryl-palladium intermediate that is poised for the subsequent insertion of carbon monoxide released in situ from the thermal decomposition of TFBen. The resulting acyl-palladium species then undergoes nucleophilic attack by the amino group of the 2-alkynylaniline, followed by reductive elimination to furnish an ortho-alkynyl benzamide intermediate. This sequence effectively constructs the amide linkage with high regioselectivity, setting the stage for the final ring-closing event. The elegance of this mechanism lies in its ability to assemble three distinct molecular fragments—the aryl iodide, the CO source, and the alkynyl aniline—into a single complex architecture with minimal byproduct formation.
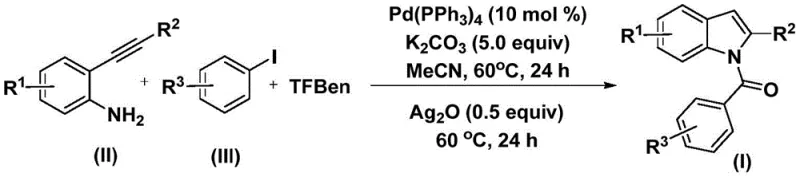
Following the formation of the amide intermediate, the reaction transitions to the cyclization phase driven by the addition of silver oxide (Ag2O). The silver species acts as a mild oxidant and Lewis acid activator, facilitating the intramolecular nucleophilic attack of the indole nitrogen onto the alkyne moiety or promoting an oxidative cyclization pathway depending on the specific electronic nature of the substrates. This step is critical for aromatizing the pyrrole ring and establishing the fully conjugated indole system characteristic of the final product. The use of Ag2O is particularly advantageous as it operates effectively at moderate temperatures (60°C), preventing the degradation of sensitive functional groups that might occur under harsher thermal conditions. Understanding these mechanistic nuances is vital for process chemists aiming to optimize reaction parameters for cost reduction in pharmaceutical intermediate manufacturing, ensuring that the catalytic cycle turns over efficiently to maximize space-time yields.
How to Synthesize N-Acyl Indole Compounds Efficiently
The execution of this synthesis requires careful attention to reagent ratios and temperature control to ensure optimal conversion and selectivity. The standardized protocol involves charging a reaction vessel with the palladium catalyst, base, CO source, and substrates in an appropriate organic solvent such as acetonitrile. The detailed procedural steps, including specific molar equivalents and workup procedures, are critical for reproducing the high yields documented in the patent literature. For process engineers and R&D teams looking to implement this technology, adhering to the specified reaction times and sequential addition of reagents is paramount to success.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynylaniline, and aryl iodide in an organic solvent.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to complete the oxidative cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers compelling advantages that directly address key pain points in the global supply chain for fine chemicals. The reliance on commercially available starting materials, such as simple aryl iodides and substituted anilines, ensures a stable and resilient supply base that is less susceptible to the volatility often seen with exotic or highly specialized reagents. Furthermore, the operational simplicity of the method, which avoids high-pressure reactors and toxic gases, translates into reduced infrastructure requirements and lower barriers to entry for contract manufacturing organizations. This accessibility fosters a more competitive market environment, potentially driving down costs for downstream API manufacturers who rely on these intermediates. The robustness of the chemistry also implies fewer batch failures and more consistent quality, which is essential for maintaining uninterrupted production schedules in a regulated industry.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the elimination of expensive and hazardous gaseous carbon monoxide infrastructure, replacing it with a bench-stable solid surrogate that simplifies logistics and storage. By consolidating what would traditionally be a multi-step synthesis into a streamlined sequence, the method significantly reduces solvent consumption, energy usage, and labor hours associated with intermediate isolations. The high atom economy of the carbonylation step ensures that raw materials are converted into product with minimal waste, further enhancing the overall cost-efficiency of the manufacturing campaign. Additionally, the use of a common solvent like acetonitrile facilitates easier recovery and recycling, contributing to long-term operational savings.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent indicates that this method can accommodate a wide variety of substitution patterns without requiring extensive process re-optimization. This flexibility allows suppliers to respond rapidly to changing demands for different indole derivatives, reducing lead times for high-purity pharmaceutical intermediates. Since the starting materials are commodity chemicals or easily synthesized precursors, the risk of supply disruption due to raw material scarcity is minimized. This reliability is crucial for pharmaceutical companies managing tight development timelines, as it ensures that critical building blocks are available when needed to support clinical trial material production.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process inherently safer and more environmentally friendly than traditional carbonylation methods. Scaling up this reaction from gram to kilogram scales does not require exponential increases in safety measures, allowing for a smoother transition from laboratory discovery to commercial production. The simplified workup procedure, involving filtration and chromatography, aligns well with standard purification capabilities found in most GMP facilities. Consequently, this technology supports sustainable manufacturing goals by reducing the environmental footprint of chemical production while maintaining high standards of product purity and safety.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of this route for specific project requirements.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid carbon monoxide substitute, eliminating the need for handling hazardous gaseous CO while ensuring efficient carbonyl insertion.
Q: What is the role of Silver Oxide (Ag2O) in the reaction?
A: Silver oxide acts as an oxidant in the second stage of the reaction, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure.
Q: Can this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and ensuring the steady supply of high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By leveraging technologies such as the palladium-catalyzed carbonylation described in CN112898192B, we can offer our clients a competitive edge through superior process economics and reliable delivery schedules.
We invite you to collaborate with us to explore how this cutting-edge synthesis can be tailored to your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential benefits of switching to this more efficient route. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and innovation in your supply chain.
