Mastering Scalable N-Acyl Indole Production: Advanced Catalytic Synthesis for High-Purity Pharmaceutical Intermediates with Cost-Efficient Manufacturing Pathways
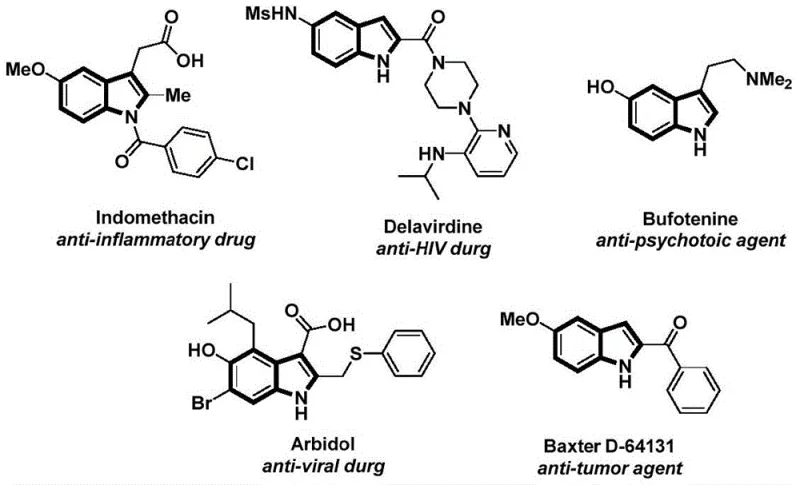
Patent CN112898192B presents a transformative methodology for synthesizing N-acyl indole compounds, a critical class of heterocyclic structures fundamental to numerous FDA-approved pharmaceuticals including anti-inflammatory agents like Indomethacin and antiviral therapeutics such as Delavirdine. This innovative approach leverages palladium-catalyzed carbonylation cyclization to achieve efficient one-step construction of these valuable intermediates under exceptionally mild conditions of 60°C without requiring hazardous carbon monoxide gas through the strategic use of phenol-based substitutes. The significance of this development is underscored by the widespread presence of indole motifs in drug molecules exhibiting antitumor, anti-inflammatory, antiviral, and antidepressant activities as documented in leading chemical reviews. By utilizing cost-effective starting materials such as commercially available aryl iodides and easily synthesized 2-alkynyl anilines, the process directly addresses key limitations in traditional synthetic routes that often suffer from harsh reaction parameters and narrow substrate scope. Furthermore, the methodology demonstrates exceptional functional group tolerance across diverse substituents including halogens, alkyl groups, and alkoxy moieties without necessitating complex protection-deprotection strategies. This patent represents a substantial advancement in organic synthesis methodology, offering both academic researchers and industrial practitioners a robust platform for producing high-purity N-acyl indole derivatives essential for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing N-acyl indole compounds typically involve multi-step sequences with harsh reaction conditions that significantly limit their industrial applicability and scalability. Conventional carbonylation methods frequently require pressurized carbon monoxide gas handling at elevated temperatures exceeding 80°C, creating substantial safety hazards and necessitating specialized equipment that increases capital expenditure while complicating regulatory compliance. These processes often demonstrate poor substrate compatibility due to sensitivity toward functional groups such as halogens or alkoxy moieties, resulting in low yields when synthesizing complex derivatives required for advanced pharmaceutical applications. Additionally, traditional methodologies frequently generate significant impurities through side reactions that necessitate extensive purification steps including multiple recrystallizations or complex chromatographic separations, thereby increasing production costs and reducing overall process efficiency. The limited availability of suitable starting materials and the requirement for expensive transition metal catalysts further constrain commercial viability while creating supply chain vulnerabilities that impact consistent production timelines. These cumulative limitations have historically prevented widespread adoption of carbonylation-based routes despite their theoretical potential for constructing valuable indole-based pharmaceutical intermediates.
The Novel Approach
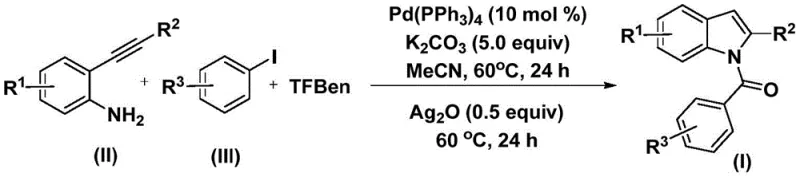
The patented methodology overcomes these critical limitations through an elegant one-step palladium-catalyzed carbonylation cyclization process that operates under remarkably mild conditions of just 60°C without requiring hazardous pressurized carbon monoxide gas. By utilizing phenol-based tricarboxylate esters as safe carbon monoxide substitutes, the process eliminates significant safety risks while maintaining high reaction efficiency across diverse substrate combinations as demonstrated by successful synthesis of fifteen distinct derivatives with varying functional groups. The strategic incorporation of silver oxide as a cyclization promoter enables direct conversion of amide intermediates into the final N-acyl indole products without requiring additional synthetic steps or specialized equipment. This streamlined approach demonstrates exceptional functional group tolerance that accommodates halogens including fluorine and chlorine substituents along with alkyl groups such as methyl and tert-butyl moieties without compromising yield or purity. The use of commercially available starting materials like tetrakis(triphenylphosphine)palladium catalysts and standard organic solvents ensures straightforward implementation using existing manufacturing infrastructure while significantly reducing raw material costs compared to traditional methods requiring rare or expensive reagents.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The catalytic cycle begins with oxidative addition of the aryl iodide into the palladium catalyst forming an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from the phenol tricarboxylate substitute to generate an acyl palladium species. This key intermediate then coordinates with the alkyne functionality of the 2-alkynyl aniline substrate through regioselective insertion followed by reductive elimination to form the amide precursor compound. The mechanism demonstrates remarkable efficiency due to the synergistic interaction between potassium carbonate base and palladium catalyst that facilitates smooth progression through each catalytic step while minimizing unwanted side reactions such as homocoupling or protodehalogenation that commonly plague alternative methodologies. The mild reaction temperature of 60°C prevents thermal decomposition of sensitive intermediates while maintaining sufficient kinetic energy for productive catalytic turnover across diverse substrate combinations including those bearing electron-donating or electron-withdrawing substituents on both aromatic rings.
Impurity control is achieved through precise regulation of reaction parameters that prevent common side products such as dimerized alkynes or hydrolyzed amides that typically arise from uncontrolled reaction conditions in conventional syntheses. The sequential addition protocol—first conducting the carbonylation step followed by silver oxide-mediated cyclization—ensures optimal timing for each transformation phase while minimizing exposure of sensitive intermediates to potentially degrading conditions. The use of acetonitrile as solvent provides ideal polarity characteristics that facilitate both catalyst stability and intermediate solubility while preventing unwanted solvolysis reactions that could compromise product purity. This carefully engineered process design results in consistently high-purity products with minimal residual metal content as evidenced by successful isolation through standard column chromatography without requiring additional purification techniques that would increase production costs or reduce overall yield.
How to Synthesize N-Acyl Indole Compounds Efficiently
This patented methodology represents a significant advancement in synthetic organic chemistry by providing a streamlined pathway for producing structurally diverse N-acyl indole compounds essential for pharmaceutical development pipelines. The process eliminates multiple synthetic steps required in conventional approaches while operating under mild conditions that preserve sensitive functional groups commonly found in complex drug molecules. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across fifteen distinct substrate combinations demonstrating consistent performance metrics under controlled manufacturing parameters. The following section provides essential implementation guidelines for research teams seeking to adopt this innovative methodology within their production workflows while ensuring optimal yield and purity outcomes.
- Combine palladium catalyst (Pd(PPh₃)₄, 10 mol%), potassium carbonate (5.0 equiv), TFBen as carbon monoxide substitute, 2-alkynyl aniline, and aryl iodide in acetonitrile solvent under inert atmosphere at 60°C for 24 hours.
- Add silver oxide (0.5 equiv) to the reaction mixture and continue heating at 60°C for an additional 24 hours to facilitate cyclization.
- Execute post-treatment via filtration, silica gel mixing, and column chromatography purification to isolate high-purity N-acyl indole products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its strategic design that prioritizes operational simplicity and resource efficiency without compromising product quality requirements. The elimination of hazardous carbon monoxide gas handling removes significant regulatory compliance burdens while reducing facility modification costs associated with specialized gas handling infrastructure required by conventional methodologies. By utilizing commercially available starting materials that are readily sourced from multiple global suppliers, the process mitigates single-source dependency risks that frequently disrupt traditional supply chains for specialty chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts required in alternative routes combined with simplified reaction workup procedures significantly reduces raw material expenses while minimizing solvent consumption during purification steps. The one-step nature of this process eliminates intermediate isolation requirements that typically consume substantial resources in multi-stage syntheses while reducing overall energy consumption through lower operating temperatures compared to conventional methods requiring elevated thermal conditions.
- Enhanced Supply Chain Reliability: The use of widely available starting materials including standard aryl iodides and easily synthesized alkynyl anilines ensures consistent raw material availability across multiple geographic regions while reducing vulnerability to supply chain disruptions affecting specialized reagents required by competing methodologies. This approach enables flexible sourcing strategies that maintain production continuity even during market fluctuations while supporting just-in-time manufacturing models through predictable lead times associated with common chemical feedstocks.
- Scalability and Environmental Compliance: The straightforward process design using standard laboratory equipment facilitates seamless scale-up from development quantities to commercial production volumes without requiring specialized infrastructure modifications that typically delay manufacturing transitions. The reduced generation of hazardous waste streams compared to traditional methods lowers environmental compliance costs while supporting corporate sustainability initiatives through minimized solvent usage and energy consumption during both reaction execution and product isolation phases.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding implementation of this patented methodology based on extensive experimental validation data from multiple substrate combinations demonstrating consistent performance across diverse functional group variations. These responses provide critical insights for technical evaluation teams assessing process feasibility within specific manufacturing environments while addressing key concerns related to operational implementation and quality assurance protocols.
Q: Why does this palladium-catalyzed method outperform conventional carbonylation approaches for N-acyl indole synthesis?
A: The patented process eliminates hazardous carbon monoxide gas through a phenol-based substitute while maintaining mild reaction conditions at 60°C. This one-step methodology avoids multi-stage sequences required in traditional routes, significantly reducing operational complexity and potential impurity formation without requiring specialized equipment.
Q: How does the catalytic system ensure exceptional substrate compatibility across diverse functional groups?
A: The palladium catalyst demonstrates broad tolerance to halogens, alkyl groups, alkoxy moieties, and trifluoromethyl substituents as evidenced by successful synthesis of multiple derivatives. The mild parameters prevent unwanted side reactions that commonly degrade sensitive functional groups in alternative methods.
Q: What scalability advantages does this process offer for commercial pharmaceutical manufacturing?
A: The straightforward procedure using standard laboratory equipment and commercially available reagents enables seamless transition from gram-scale to industrial production volumes. Simple post-treatment involving filtration and column chromatography is readily adaptable to large-scale manufacturing without requiring specialized infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compound Supplier
Our patented methodology represents a significant advancement in producing high-purity N-acyl indole compounds essential for modern pharmaceutical development pipelines where structural precision directly impacts therapeutic efficacy and safety profiles. As a CDMO specialist with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we combine cutting-edge synthetic expertise with stringent purity specifications verified through rigorous QC labs to ensure consistent product quality meeting global regulatory standards. Our dedicated technical teams provide comprehensive support throughout the entire development lifecycle from initial feasibility assessment through full-scale manufacturing implementation while maintaining strict adherence to cGMP requirements across all production phases.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today—they will provide detailed COA data and route feasibility assessments demonstrating how this innovative methodology can optimize your supply chain while ensuring reliable access to high-purity intermediates essential for your drug development programs.
