Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Pharmaceutical Manufacturing
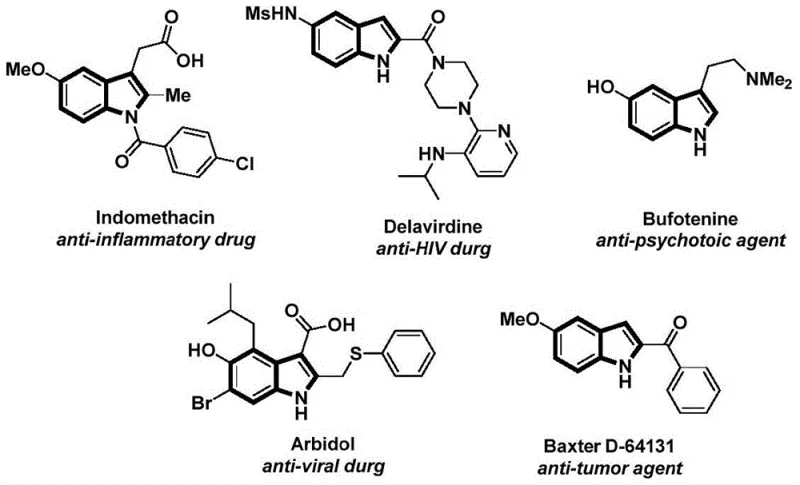
The structural integrity and functional diversity of the indole scaffold make it a cornerstone in modern medicinal chemistry, underpinning a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-HIV drugs such as Delavirdine. As depicted in the structural overview of key pharmaceutical agents, the N-acyl indole motif is particularly prevalent in compounds exhibiting antitumor, antiviral, and psychotropic activities. Recognizing the critical demand for efficient access to these privileged structures, recent intellectual property developments, specifically patent CN112898192B, have introduced a transformative synthetic methodology. This patent discloses a robust preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. By utilizing readily available 2-alkynylanilines and aryl iodides in conjunction with a solid carbon monoxide surrogate, this technology addresses long-standing challenges in heterocyclic synthesis, offering a streamlined pathway that is highly attractive for the scalable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-acyl indole frameworks has often relied on traditional Fischer indole synthesis or multi-step sequences involving pre-functionalized precursors, which frequently suffer from significant operational drawbacks. Conventional carbonylation strategies typically necessitate the use of gaseous carbon monoxide, a toxic and hazardous reagent that requires specialized high-pressure equipment and rigorous safety protocols, thereby complicating the manufacturing process and inflating infrastructure costs. Furthermore, many existing routes exhibit poor atom economy and limited substrate scope, often failing to tolerate sensitive functional groups such as halogens or electron-donating moieties without extensive protecting group manipulation. These inefficiencies result in prolonged reaction times, lower overall yields, and the generation of substantial chemical waste, creating bottlenecks for procurement teams seeking cost-effective and reliable sources of complex heterocyclic building blocks for drug discovery pipelines.
The Novel Approach
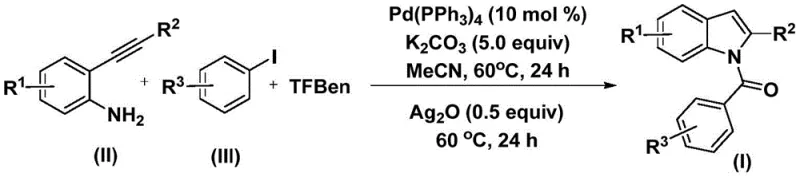
In stark contrast to these legacy methods, the methodology outlined in patent CN112898192B presents a sophisticated yet operationally simple solution that fundamentally reshapes the synthetic landscape for N-acyl indoles. This novel approach employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and effective solid carbon monoxide substitute, effectively mitigating the safety risks associated with handling CO gas while maintaining high reaction efficiency. The process integrates a palladium-catalyzed carbonylation with a subsequent silver-mediated cyclization in a sequential one-pot fashion, significantly reducing the need for intermediate isolation and purification steps. As illustrated in the general reaction scheme, the transformation proceeds under mild thermal conditions (60°C) in acetonitrile, demonstrating exceptional compatibility with a wide range of substituents including methyl, methoxy, tert-butyl, and various halogens. This strategic innovation not only simplifies the workflow but also enhances the overall practicality of the synthesis, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic elegance of this transformation lies in its dual-catalytic nature, orchestrating a seamless sequence of organometallic events to construct the indole core with precision. The reaction is initiated by the oxidative addition of the zero-valent palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, the carbon monoxide released in situ from the decomposition of TFBen inserts into the palladium-carbon bond, forming an acyl-palladium species. This electrophilic acyl complex then undergoes nucleophilic attack by the amino group of the 2-alkynylaniline, followed by reductive elimination to yield an intermediate amide compound. The final and crucial step involves the addition of silver oxide, which acts as an oxidant to promote the intramolecular cyclization of the amide, closing the ring to furnish the desired N-acyl indole product. This intricate dance of coordination chemistry ensures high regioselectivity and minimizes the formation of undesired byproducts, a critical factor for R&D directors focused on impurity control.
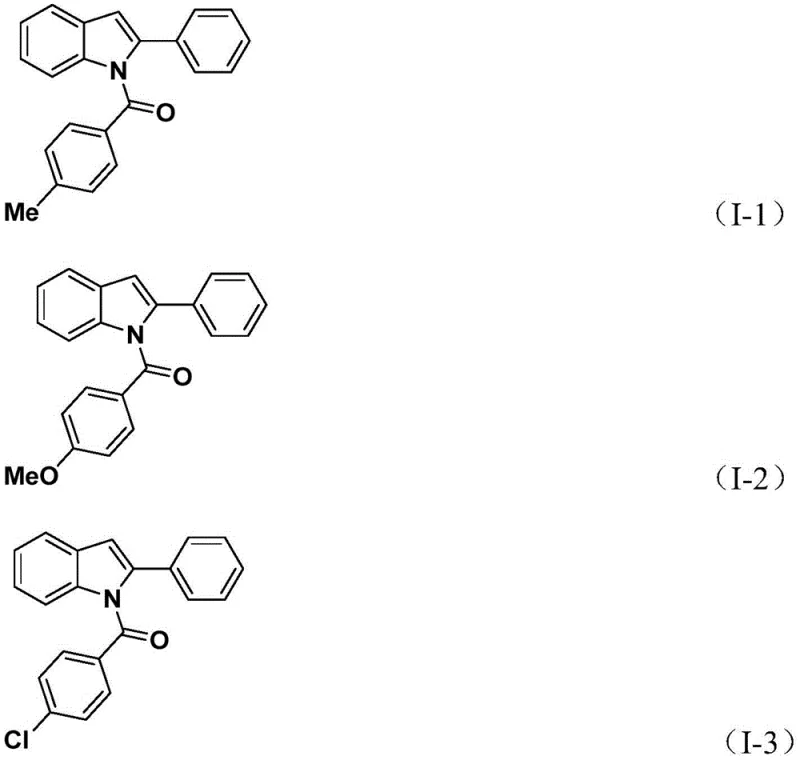
From an impurity profile perspective, the use of mild conditions and specific reagents like potassium carbonate as a base helps to suppress side reactions such as homocoupling of the alkyne or hydrodehalogenation of the aryl iodide. The choice of acetonitrile as the solvent further optimizes the solubility of all reactants and intermediates, ensuring a homogeneous reaction environment that facilitates consistent conversion rates. Experimental data from the patent indicates that this method tolerates diverse electronic environments on the aromatic rings, with yields ranging significantly depending on the steric and electronic nature of the substituents. For instance, substrates bearing electron-donating groups like methoxy or alkyl chains generally proceed smoothly, while the system also accommodates electron-withdrawing halogens, demonstrating the robustness required for synthesizing diverse libraries of bioactive analogs. The ability to achieve yields up to 82% for specific derivatives underscores the efficiency of this catalytic cycle in converting starting materials into valuable chemical entities.
How to Synthesize N-Acyl Indole Efficiently
The practical implementation of this synthesis protocol is designed to be accessible for laboratory-scale optimization while retaining the potential for industrial translation. The procedure involves a straightforward charging of reagents into a reaction vessel, followed by a controlled thermal profile that drives the two distinct stages of the transformation. Operators simply combine the palladium catalyst, base, CO source, and substrates in the organic solvent, allowing the carbonylation to proceed before introducing the oxidant for the cyclization phase. This modular approach allows for fine-tuning of reaction parameters to maximize yield for specific substrate combinations. For detailed standard operating procedures and precise stoichiometric ratios validated by experimental data, please refer to the synthesis guide below.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynylaniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate carbonylation and amide formation.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling strategic advantages that extend beyond mere chemical novelty. The shift towards using solid CO surrogates like TFBen eliminates the logistical complexities and safety liabilities associated with storing and transporting high-pressure carbon monoxide cylinders, thereby streamlining facility operations and reducing regulatory burdens. Furthermore, the reliance on commercially available starting materials such as 2-alkynylanilines and aryl iodides ensures a stable and resilient supply chain, minimizing the risk of production delays caused by raw material shortages. The mild reaction conditions (60°C) and the use of common solvents like acetonitrile contribute to significant energy savings and simplified waste management protocols, aligning with modern green chemistry principles and environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements and the use of a one-pot sequential process drastically reduce capital expenditure and operational overheads. By avoiding the need for specialized autoclaves and complex gas handling systems, manufacturers can achieve substantial cost savings in CAPEX and maintenance. Additionally, the high atom economy and reduced number of isolation steps lower the consumption of solvents and silica gel for purification, directly impacting the cost of goods sold (COGS) and improving overall margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: The broad substrate compatibility of this method means that a single standardized protocol can be applied to synthesize a wide variety of N-acyl indole derivatives, reducing the need for multiple specialized manufacturing lines. This flexibility allows suppliers to respond rapidly to changing market demands for different API intermediates without extensive retooling. The robustness of the reaction against moisture and air, typical of palladium-catalyzed systems optimized with phosphine ligands, further ensures consistent batch-to-batch quality, fostering trust and long-term partnerships with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The transition from bench-scale to commercial production is facilitated by the simplicity of the workup procedure, which involves basic filtration and column chromatography, techniques that are easily adaptable to large-scale processing. The avoidance of toxic CO gas significantly improves the safety profile of the manufacturing site, reducing insurance premiums and enhancing worker safety. Moreover, the efficient conversion rates minimize the generation of chemical waste, supporting sustainability goals and ensuring compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational parameters and chemical capabilities of the method for potential partners and technical evaluators.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid carbon monoxide substitute, eliminating the need for handling hazardous high-pressure CO gas while ensuring efficient carbonyl insertion.
Q: What is the role of Silver Oxide (Ag2O) in the reaction?
A: Silver oxide acts as a crucial oxidant in the second stage of the reaction, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest industry standards, providing our clients with the confidence needed to advance their clinical programs.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can optimize your supply chain and drive value for your organization.
