Advanced Synthetic Route for Anagrelide Intermediates: Enhancing Purity and Scalability for Global Supply Chains
Advanced Synthetic Route for Anagrelide Intermediates: Enhancing Purity and Scalability for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with environmental sustainability, particularly for critical cardiovascular therapeutics. Patent CN102256981A introduces a transformative methodology for the preparation of Anagrelide and its analogues, specifically targeting the synthesis of 6,7-dichloro-1,5-dihydroimidazo[2,1-b]quinazolin-2(3H)-one. This intellectual property represents a significant departure from legacy synthetic routes that have long relied on hazardous reagents and inefficient purification protocols. By shifting the synthetic strategy towards a nitrobenzyl sulfonate ester intermediate approach, the invention addresses critical pain points regarding toxicity, waste generation, and process scalability. For R&D directors and procurement specialists evaluating long-term supply strategies, understanding the mechanistic advantages of this patent is essential for securing a reliable API intermediate supplier capable of delivering consistent quality.
The core innovation lies in the strategic manipulation of the aromatic ring substitution pattern prior to the formation of the imidazoquinazoline core. Traditional methods often struggle with regioselectivity and the handling of unstable intermediates, leading to variable yields and complex impurity profiles. In contrast, the disclosed process leverages the unexpected preferential nitration at the 6-position of 2,3-dichlorobenzaldehyde, a finding that streamlines the entire upstream synthesis. This level of control over the molecular architecture not only enhances the chemical purity of the final active pharmaceutical ingredient but also reduces the operational burden on manufacturing facilities. As we delve deeper into the technical specifics, it becomes clear that this process offers a viable solution for cost reduction in pharmaceutical manufacturing while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial production of Anagrelide has been plagued by reliance on intermediates such as ethyl-N-(6-amino-2,3-dichlorobenzyl)glycine, which is typically derived from 2,3-dichloro-6-nitrobenzonitrile. This precursor is notorious for its high toxicity and potential for causing severe skin irritation, posing significant occupational health risks in a production environment. Furthermore, alternative routes involving the free radical halogenation of tolyl groups are notoriously difficult to control on a large scale, often resulting in mixtures of halogenated products that require extensive and costly separation processes. Perhaps most critically, the reduction of nitro groups in these conventional pathways frequently employs tin chloride in strong acid conditions. This generates substantial quantities of stanniferous waste, creating a heavy environmental burden and complicating the disposal logistics for chemical plants. Additionally, the acidic conditions can promote unwanted side reactions, such as the further chlorination of the aromatic ring, leading to difficult-to-remove trichloro impurities that compromise the safety profile of the final drug substance.
The Novel Approach
The methodology outlined in CN102256981A circumvents these issues by initiating the synthesis from the more benign and commercially accessible 2,3-dichlorobenzaldehyde. Through a carefully controlled nitration process, the invention achieves high selectivity for the 6-nitro isomer, which can be easily purified via crystallization, thereby establishing a high-purity foundation for subsequent steps. Instead of relying on hazardous halogenation or toxic nitriles, the process converts the resulting nitrobenzyl alcohol into a sulfonate ester, such as a mesylate. This activated intermediate serves as a superior electrophile for coupling with glycine derivatives, proceeding under much milder conditions than traditional alkyl halides. The elimination of tin-based reductants in favor of catalytic hydrogenation or other clean reduction methods further underscores the environmental superiority of this route. By minimizing the formation of heavy metal waste and avoiding aggressive acidic conditions, this novel approach ensures a cleaner reaction profile and simplifies the downstream purification of the quinazoline core.
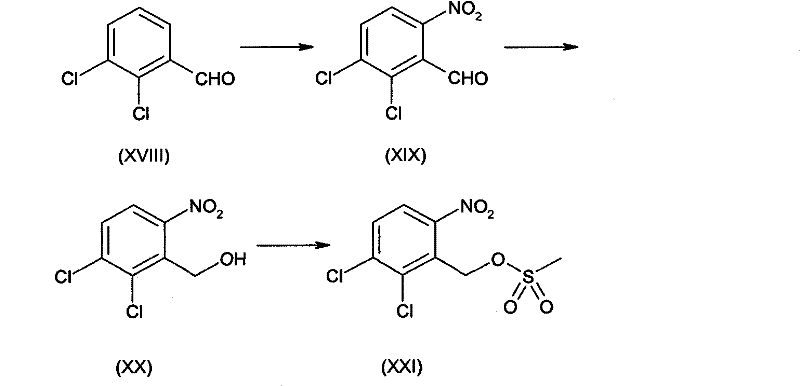
Mechanistic Insights into Nitrobenzyl Sulfonate Activation and Cyclization
A detailed examination of the reaction mechanism reveals why this pathway offers such distinct advantages for the commercial scale-up of complex quinazoline derivatives. The initial nitration of 2,3-dichlorobenzaldehyde is counter-intuitive, as one might expect substitution at other positions; however, the specific electronic effects of the dichloro and aldehyde substituents direct the nitro group predominantly to the 6-position. This regioselectivity is the cornerstone of the process efficiency, as it avoids the formation of isomeric byproducts that would otherwise dilute the yield and complicate purification. Following the reduction of the aldehyde to the alcohol, the conversion to the sulfonate ester (Compound XIII in the patent general formula) activates the benzylic position for nucleophilic attack without introducing the instability associated with benzylic halides. This stability allows the intermediate to be isolated or carried forward with minimal degradation, ensuring a robust supply of material for the coupling step.
The subsequent coupling with glycine derivatives and the final cyclization steps are equally optimized for purity. The use of organic bases like triethylamine facilitates the displacement of the sulfonate group by the amine nitrogen of the glycine derivative, forming the crucial carbon-nitrogen bond. Following this, the reduction of the aromatic nitro group to an amine can be achieved using catalytic hydrogenation over palladium on carbon, a method that produces only water as a byproduct, in stark contrast to the metal sludge generated by tin reduction. The final cyclization to form the imidazoquinazoline ring is executed under conditions that minimize the hydrolysis of the lactam nucleus, a common failure point in older methods where acidolysis competed with salt formation. This precise control over the reaction environment ensures that the final Anagrelide base is obtained with exceptional purity, often exceeding 99.8% as determined by HPLC, before being converted to the stable hydrochloride or hydrobromide salt forms.
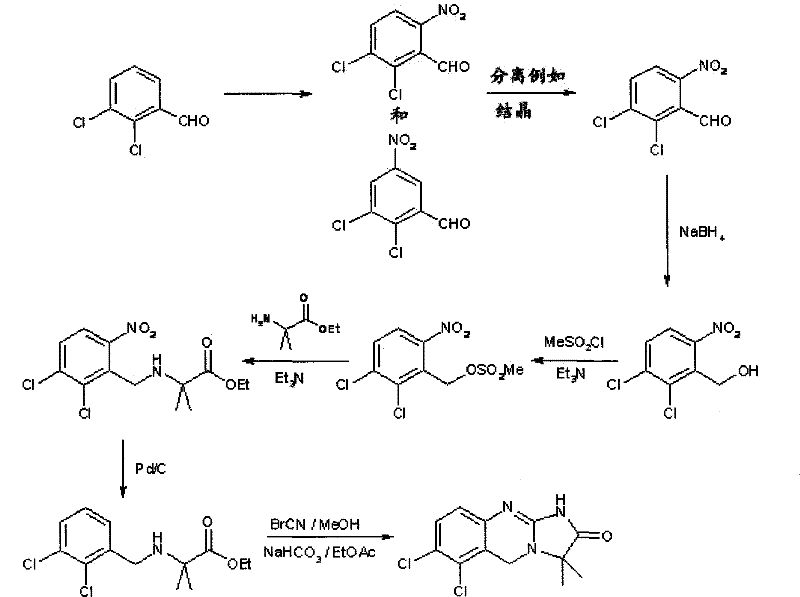
How to Synthesize Anagrelide Intermediates Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly temperature control during the nitration and activation steps to maintain selectivity. The process begins with the nitration of the aldehyde precursor in a sulfuric acid medium, followed by reduction to the alcohol using sodium borohydride. The critical activation step involves reacting the alcohol with methanesulfonyl chloride in the presence of a tertiary amine base at low temperatures to prevent decomposition. Subsequent coupling with the appropriate glycine ester and reduction of the nitro group sets the stage for the final ring closure. While the general principles are straightforward, the specific stoichiometry and solvent choices detailed in the patent are vital for achieving the reported high yields and purity levels. For a comprehensive understanding of the exact operational parameters required to replicate this success in a pilot or production setting, please refer to the standardized synthesis guide below.
- Nitration of 2,3-dichlorobenzaldehyde to form 2,3-dichloro-6-nitrobenzaldehyde, followed by reduction to the corresponding benzyl alcohol.
- Activation of the benzyl alcohol using alkyl sulphonyl halide (e.g., mesyl chloride) to form a stable nitrobenzyl sulfonate ester intermediate.
- Coupling with glycine derivatives, followed by catalytic hydrogenation and cyclization to yield the final quinazolinone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic pathway offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost reduction in pharmaceutical manufacturing driven by the simplification of the waste management infrastructure. By eliminating the need for tin chloride and the associated heavy metal waste treatment, facilities can significantly lower their operational expenditures related to environmental compliance and hazardous waste disposal. Furthermore, the use of readily available starting materials like 2,3-dichlorobenzaldehyde reduces dependency on specialized, toxic precursors that may be subject to supply volatility or restrictive regulatory controls. This shift enhances supply chain reliability by sourcing from a broader, more stable market of commodity chemicals, thereby mitigating the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as tin chloride and toxic nitriles directly translates to lower raw material costs and reduced waste disposal fees. The ability to purify key intermediates via simple crystallization rather than complex chromatography or extensive distillation further drives down processing costs. Additionally, the higher selectivity of the nitration step minimizes the loss of valuable material to isomeric byproducts, effectively increasing the overall mass efficiency of the plant. These factors combine to create a more economically viable production model that can withstand market fluctuations better than legacy processes reliant on inefficient, multi-step purifications.
- Enhanced Supply Chain Reliability: Sourcing 2,3-dichlorobenzaldehyde is significantly less risky than procuring specialized nitrobenzonitriles or managing the logistics of radical halogenation reagents. The robustness of the sulfonate ester intermediate means it can potentially be stockpiled or transported with greater safety margins compared to unstable halides. This stability allows for more flexible production scheduling and inventory management, ensuring that high-purity cardiovascular drug intermediates are available to meet demand without the bottlenecks often caused by difficult-to-control exothermic reactions or hazardous material handling requirements in the supply line.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing unit operations such as filtration and crystallization that are easily transferred from the laboratory to multi-ton reactors. The avoidance of heavy metal contaminants simplifies the validation process for GMP manufacturing, as there is no need for rigorous testing and removal of residual tin. This aligns perfectly with modern green chemistry initiatives, reducing the facility's environmental footprint and ensuring long-term regulatory compliance. The cleaner reaction profile also means less downtime for equipment cleaning between batches, maximizing asset utilization and throughput for commercial scale-up of complex quinazoline derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent specification, offering clarity on how this method outperforms traditional synthesis routes. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing the quality credentials of potential suppliers.
Q: How does this new process improve upon conventional Anagrelide synthesis methods?
A: The novel process eliminates the need for toxic 2,3-dichloro-6-nitrobenzonitrile precursors and avoids the generation of heavy stanniferous waste associated with tin chloride reductions, significantly simplifying waste management and purification.
Q: What are the key advantages of using sulfonate ester intermediates in this pathway?
A: Using sulfonate esters allows for milder reaction conditions compared to radical halogenation, improves regioselectivity during nitration, and facilitates easier purification of intermediates through crystallization, leading to higher overall purity.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes readily available starting materials like 2,3-dichlorobenzaldehyde and employs robust steps like catalytic hydrogenation, making it highly scalable and environmentally compliant for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anagrelide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, high-quality supply to truly benefit our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN102256981A is executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect even trace levels of impurities, guaranteeing that every batch of Anagrelide intermediate meets the exacting standards required for cardiovascular drug formulation. Our commitment to process excellence means we can deliver the cost and quality benefits of this advanced synthetic route to your global supply chain without compromise.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient pathway. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of high-purity Anagrelide intermediates into your manufacturing pipeline.
