Scalable Production of Bilastine Intermediates via Palladium-Catalyzed Heck Coupling
Scalable Production of Bilastine Intermediates via Palladium-Catalyzed Heck Coupling
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of second-generation antihistamines, particularly bilastine, which is renowned for its superior safety profile and lack of cardiotoxicity compared to earlier generations. Central to the supply chain of this critical medication is the efficient synthesis of its key precursors. Patent CN110734375A discloses a groundbreaking preparation method for the bilastine intermediate represented by Formula I, offering a significant technological leap over traditional synthetic routes. This novel approach utilizes 2-bromoethyl anisole as a readily available starting material, transforming it through a streamlined three-step sequence involving elimination, palladium-catalyzed coupling, and hydrogenation. The structural integrity and purity of the final compound, 4-(2-hydroxyethyl)-phenyl tert-butyl methyl ester, are paramount for downstream API synthesis, and this patent provides a definitive solution for achieving high-quality standards.
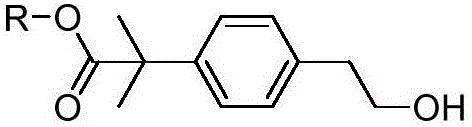
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the complex and hazardous steps associated with legacy methodologies. By establishing a reliable bilastine intermediate supplier network based on this chemistry, manufacturers can secure a consistent supply of high-purity pharmaceutical intermediates. The method not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing the reliance on toxic reagents and extreme reaction conditions, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing while maintaining rigorous quality control standards essential for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the methodology described in CN110734375A, the synthesis of similar phenethyl alcohol derivatives often relied on cumbersome and dangerous chemical transformations. As illustrated in literature reports such as those by Collier et al., traditional routes frequently employed borane dimethyl sulfide complexes for reduction steps, followed by oxidation with hydrogen peroxide. These reagents present substantial handling challenges; borane dimethyl sulfide is notoriously malodorous and poses significant air pollution risks within a production facility, requiring specialized scrubbing systems and containment protocols. Furthermore, the use of hydrogen peroxide introduces inherent safety hazards related to exothermic oxidation reactions, necessitating strict temperature controls to prevent runaway scenarios.
Additionally, alternative synthetic strategies reported in patents like WO2009/102155 and US2011/9636 often require cryogenic conditions, typically operating at temperatures as low as -78°C. Such extreme low-temperature requirements impose a heavy burden on infrastructure, demanding expensive cryogenic cooling equipment and significantly increasing energy consumption. Moreover, intermediates generated under these harsh conditions are often sensitive to moisture, leading to hydrolysis issues that complicate storage and transport. These factors collectively result in a fragile supply chain with high operational costs and increased risk of batch failure, highlighting the urgent need for a more robust and mild synthetic alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
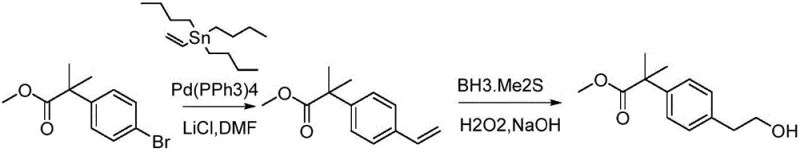
The innovative route disclosed in the patent fundamentally reshapes the synthesis landscape by introducing a mild, high-yielding three-step process that eliminates the aforementioned hazards. The strategy begins with a base-mediated elimination reaction where 2-bromoethyl anisole (Formula IV) is treated with DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) to generate vinyl anisole (Formula V). This step proceeds efficiently in solvents like tetrahydrofuran at moderate temperatures (60-75°C), avoiding the need for cryogenics. The subsequent key step involves a palladium-catalyzed Heck coupling between the newly formed vinyl anisole and a brominated aromatic ester (Formula II). This cross-coupling reaction is performed under inert atmosphere at 90-110°C, utilizing standard palladium catalysts such as tetrakis(triphenylphosphine)palladium(0), ensuring high regioselectivity and conversion rates without the sensitivity issues of previous methods.
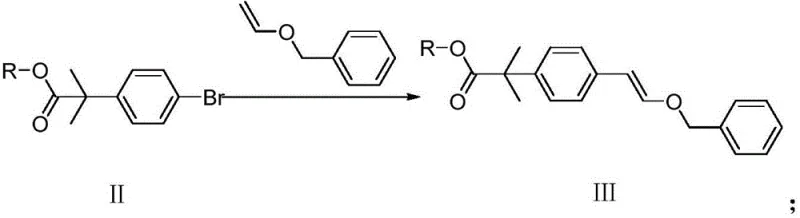
The final transformation involves a catalytic hydrogenation of the vinyl ether intermediate (Formula III) to yield the target hydroxyethyl compound (Formula I). This reduction is carried out under mild hydrogen pressure (0.09-0.12 MPa) at ambient temperatures (15-30°C) using palladium on carbon. This stands in stark contrast to the dangerous oxidation/reduction sequences of the past. The entire sequence is characterized by simple work-up procedures, typically involving filtration and concentration, which drastically simplifies the isolation of the product. For procurement managers, this translates to a process that is not only safer but also significantly more predictable in terms of lead time and output consistency, effectively reducing lead time for high-purity pharmaceutical intermediates by minimizing downtime associated with hazardous waste disposal and equipment maintenance.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling
The core of this synthetic breakthrough is the palladium-catalyzed Heck coupling reaction, a powerful tool for forming carbon-carbon bonds between aryl halides and alkenes. In this specific application, the mechanism initiates with the oxidative addition of the aryl bromide (Formula II) to the active Pd(0) species, generating an aryl-palladium(II) bromide complex. This step is facilitated by the electron-rich nature of the phosphine ligands on the catalyst, which stabilize the metal center and promote the insertion into the carbon-bromine bond. Following oxidative addition, the vinyl anisole (Formula V) coordinates to the palladium center and undergoes migratory insertion. The regioselectivity of this insertion is critical; the reaction favors the formation of the trans-alkene product where the aryl group attaches to the terminal carbon of the vinyl ether, driven by steric factors and the electronic properties of the alkoxy substituent.
Subsequent beta-hydride elimination releases the coupled product (Formula III) and regenerates a palladium-hydride species, which is then neutralized by the base (such as potassium carbonate or triethylamine) to restore the active Pd(0) catalyst, completing the catalytic cycle. From an impurity control perspective, the mild basic conditions and the specific choice of solvent (e.g., 1,4-dioxane) play a vital role in suppressing side reactions such as homocoupling of the aryl halide or isomerization of the double bond. The use of DBU in the preceding elimination step also ensures high purity of the vinyl anisole feedstock, preventing the carryover of unreacted bromide which could otherwise compete in the coupling reaction. This precise control over the reaction environment ensures that the final product meets stringent purity specifications, a key requirement for any reliable agrochemical intermediate supplier or pharmaceutical partner.
How to Synthesize 4-(2-hydroxyethyl)-phenyl tert-butyl methyl ester Efficiently
The synthesis of this critical bilastine intermediate is achieved through a logical progression of three distinct chemical transformations, each optimized for maximum yield and minimal environmental impact. The process begins with the preparation of the alkene synthon via elimination, followed by the construction of the carbon skeleton through cross-coupling, and concludes with the saturation of the double bond to reveal the primary alcohol functionality. Detailed operational parameters, including specific solvent ratios, catalyst loadings, and temperature profiles, are essential for replicating the high success rates reported in the patent examples. The following guide outlines the standardized synthesis steps derived from the proprietary data, providing a clear roadmap for process implementation.
- Perform base-mediated elimination of 2-bromoethyl anisole using DBU in a suitable solvent like tetrahydrofuran to generate vinyl anisole.
- Execute a palladium-catalyzed Heck coupling reaction between the generated vinyl anisole and methyl 3-bromophenyl tert-butyl ester under inert atmosphere.
- Conduct catalytic hydrogenation of the resulting vinyl ether intermediate using palladium on carbon to yield the final hydroxyethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this novel synthetic route offers profound strategic advantages beyond mere technical feasibility. The elimination of hazardous reagents like borane dimethyl sulfide and the avoidance of cryogenic temperatures directly correlate to a drastic simplification of the manufacturing infrastructure. Facilities no longer require specialized containment for malodorous gases or expensive low-temperature reactors, which significantly lowers the barrier to entry for production and reduces capital expenditure. Furthermore, the stability of the intermediates allows for more flexible inventory management, as the materials are less prone to degradation during storage compared to moisture-sensitive compounds generated by older methods. This robustness enhances supply chain reliability, ensuring that production schedules are met without unexpected interruptions due to reagent instability or safety incidents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the removal of expensive and difficult-to-handle reagents. By replacing borane complexes and cryogenic cooling with standard palladium catalysts and moderate heating, the operational expenditure is substantially decreased. The high yields reported in each step (exceeding 90% in many cases) minimize raw material waste, leading to a more efficient atom economy. Additionally, the simplified work-up procedures, which often involve simple filtration and solvent removal rather than complex chromatographic separations, reduce labor costs and solvent consumption, contributing to overall cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 2-bromoethyl anisole and methyl 3-bromophenyl tert-butyl ester, mitigates the risk of raw material shortages. Unlike specialized reagents that may have long lead times or single-source suppliers, the inputs for this process are commodity chemicals with robust global supply chains. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without the need for highly specialized equipment, ensuring continuity of supply even in the face of regional disruptions or facility maintenance.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with peroxide oxidations or pyrophoric reagents. The generation of three wastes is significantly minimized, aligning with increasingly stringent environmental regulations. The use of recyclable palladium catalysts and common organic solvents facilitates waste treatment and recovery, making the process environmentally sustainable. This compliance not only avoids potential regulatory fines but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious partners in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of bilastine intermediates using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implications of adopting this route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their broader API synthesis strategies.
Q: Why is this new synthetic route considered safer than conventional methods?
A: Conventional methods often utilize hazardous reagents such as borane dimethyl sulfide, which is malodorous and poses air pollution risks, or hydrogen peroxide, which presents oxidation hazards. The novel route described in patent CN110734375A eliminates these dangerous reagents, relying instead on mild palladium catalysis and standard hydrogenation, significantly improving operational safety.
Q: What are the typical yields achieved in this three-step process?
A: The patented process demonstrates exceptional efficiency across all stages. Experimental data indicates yields of approximately 97% for the elimination step, 96.7% for the Heck coupling, and 91% for the final hydrogenation, resulting in a highly efficient overall production capability suitable for industrial scale-up.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is specifically designed for industrial applicability. It operates under mild reaction temperatures (ranging from ambient to 110°C) and avoids extreme conditions like cryogenic cooling (-78°C), making it compatible with standard glass-lined or stainless steel reactors found in most fine chemical facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the timely delivery of life-saving medications. Our technical team has thoroughly analyzed the synthetic pathway described in CN110734375A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life on an industrial scale. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of bilastine intermediate performs consistently in your downstream processes. Our facility is equipped to handle the palladium-catalyzed reactions and hydrogenation steps with the highest safety and efficiency standards.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. By leveraging our expertise in this specific synthetic route, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can support your goal of bringing effective antihistamine therapies to patients worldwide with greater speed and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
