Scalable Nickel-Catalyzed Synthesis of Bilastine Key Intermediate for Global Pharma Supply Chains
Scalable Nickel-Catalyzed Synthesis of Bilastine Key Intermediate for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust, cost-effective pathways for the production of high-value active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in Chinese Patent CN113292534B, which discloses a novel preparation method for a critical Bilastine intermediate, specifically 2-(4-(2-(4-(1-(2-ethoxyethyl)-1H-benzo[d]imidazol-2-yl)piperidin-1-yl)ethyl)phenyl)-2-methylpropionate (Compound II). Bilastine is a second-generation, non-sedative histamine H1 receptor antagonist widely used for treating allergic rhinitis and urticaria, known for its high bioavailability and lack of cardiotoxicity. The efficient synthesis of its key intermediates is paramount for meeting global demand. This patent introduces a transformative approach that utilizes a Nickel-complex catalyst system coupled with a Lewis acid to construct the challenging quaternary carbon atom adjacent to the phenyl ring, a structural motif that has historically plagued synthetic chemists with low yields and complex purification steps.
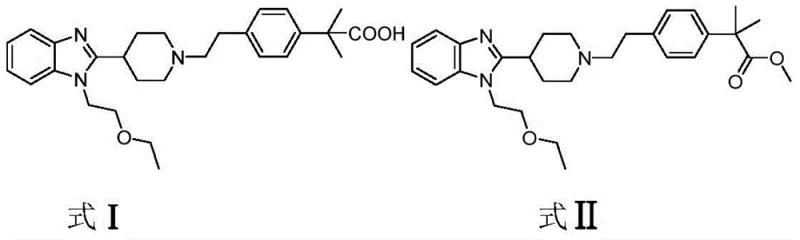
The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for the final API. Traditional methods often struggle with the steric hindrance inherent in forming the gem-dimethyl group on the propionate chain. By leveraging a nickel-catalyzed cross-coupling strategy between an aryl halide precursor and an isobutyrate ester enolate, this technology offers a streamlined alternative to legacy processes. For procurement managers and supply chain directors, this represents a shift towards more reliable pharmaceutical intermediates supplier networks, where raw material availability and process safety are prioritized over risky, multi-step sequences involving hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Bilastine intermediates have been fraught with significant technical and economic bottlenecks that hinder large-scale industrial adoption. Prior art, such as the methods described in CN105017211, relied heavily on the use of Sodium Hydride (NaH) for methylation reactions. NaH is notoriously pyrophoric and moisture-sensitive, posing severe safety hazards during industrial scale-up; consequently, production often must be halted during humid weather conditions, leading to unpredictable lead times and inflated operational costs. Furthermore, other documented pathways, including those referencing US 20119636A1 and WO2009102155, depend on Palladium-catalyzed coupling reactions. While effective on a small scale, Palladium is a precious metal with volatile pricing, and the requisite phosphine ligands add substantial expense to the bill of materials. Additionally, routes involving Friedel-Crafts acylation generate copious amounts of aluminum salt wastewater, creating a heavy burden on environmental treatment facilities and complicating regulatory compliance for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
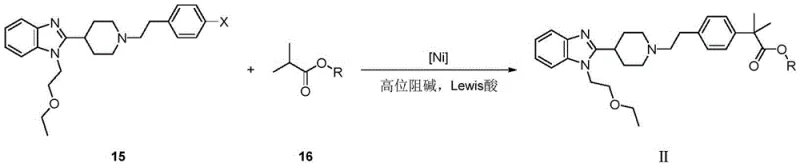
The methodology presented in Patent CN113292534B elegantly circumvents these obstacles by employing a base-metal catalytic system. The core innovation lies in the direct condensation of a halogenated aryl precursor (Compound 15) with an isobutyrate ester (Compound 16) under the influence of a Nickel complex and a Lewis acid. This single-step construction of the quaternary carbon center eliminates the need for dangerous methylation reagents like methyl iodide and avoids the use of expensive Palladium catalysts entirely. The reaction proceeds under relatively mild thermal conditions, typically around 80°C, using common organic solvents such as tetrahydrofuran (THF). This shift not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing toxic waste streams. For stakeholders focused on commercial scale-up of complex pharmaceutical intermediates, this route offers a compelling value proposition through enhanced safety profiles and superior atom economy.
Mechanistic Insights into Nickel-Catalyzed Enolate Alkylation
The success of this synthesis hinges on the precise orchestration of a catalytic cycle involving Nickel, a sterically hindered base, and a Lewis acid promoter. The mechanism initiates with the activation of the isobutyrate ester (Compound 16) by a strong, non-nucleophilic base such as Sodium Bis(trimethylsilyl)amide (NaHMDS). This deprotonation generates a reactive enolate ion, which is inherently unstable and prone to self-condensation if not properly managed. Here, the role of the Lewis acid, specifically Zinc Bromide (ZnBr2), becomes critical. The Lewis acid coordinates with the enolate oxygen, stabilizing the negative charge and modulating the nucleophilicity of the species to favor cross-coupling over side reactions. Simultaneously, the Nickel catalyst, formed in situ from salts like Ni(COD)2 and bidentate ligands such as BINAP, undergoes oxidative addition with the aryl halide bond of Compound 15. This forms an organonickel intermediate that is poised for transmetallation with the zinc-stabilized enolate.
Following transmetallation, the catalytic cycle concludes with a reductive elimination step that forges the new carbon-carbon bond, releasing the target product (Compound II) and regenerating the active Nickel(0) species. The choice of ligand is paramount; experimental data within the patent indicates that dual-ligand systems or specific bulky phosphines like BINAP significantly outperform simple monodentate ligands, likely by preventing the formation of inactive Nickel clusters and facilitating the difficult reductive elimination from the sterically crowded quaternary center. This mechanistic understanding allows for rigorous impurity control, as the specific coordination environment suppresses the formation of homocoupling byproducts (biaryls) that typically plague nickel-catalyzed reactions. The result is a high-purity crude product that requires minimal downstream purification, directly addressing the quality concerns of R&D Directors focused on impurity profiles.
How to Synthesize Bilastine Intermediate II Efficiently
Executing this synthesis requires strict adherence to anhydrous conditions and precise stoichiometric control to maximize the efficiency of the nickel catalytic cycle. The process begins with the preparation of the active catalyst species, followed by the sequential addition of reagents to manage exotherms and ensure complete conversion. The optimization of reaction parameters, including temperature, solvent choice, and molar ratios, is essential to achieving the reported high yields. Below is the structured guide for implementing this robust manufacturing protocol.
- Prepare the nickel catalyst system by mixing Ni(COD)2 with BINAP ligand in toluene at 60°C to form a dark red solution.
- Generate the enolate ion by reacting methyl isobutyrate with NaHMDS base, followed by the addition of Lewis acid ZnBr2.
- Couple the enolate with the aryl halide precursor (Compound 15) in THF at 80°C for 20 hours to yield the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the transition to this nickel-catalyzed protocol offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the decoupling of production costs from the volatile precious metals market. By substituting Palladium with Nickel, manufacturers can achieve substantial cost savings on raw materials without compromising reaction efficiency. Furthermore, the elimination of Sodium Hydride removes a significant logistical bottleneck; facilities no longer need to restrict production schedules based on humidity levels or invest in specialized containment infrastructure for pyrophoric materials. This reliability translates directly into more consistent delivery timelines and reduced risk of supply chain disruption for downstream API manufacturers.
- Cost Reduction in Manufacturing: The replacement of expensive Palladium catalysts and toxic methylating agents with abundant Nickel salts and benign esters fundamentally alters the cost structure of the intermediate. The process avoids the generation of aluminum-containing wastewater, which significantly lowers waste disposal fees and reduces the load on effluent treatment plants. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, thereby improving the overall material throughput and reducing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: By removing hazardous reagents like NaH and Methyl Iodide from the process map, the manufacturing facility becomes inherently safer and more resilient. This safety profile allows for continuous operation regardless of seasonal weather variations, ensuring a steady flow of inventory. The use of commercially available and stable starting materials, such as simple aryl halides and isobutyrate esters, further secures the supply chain against raw material shortages, making this a highly dependable source for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions are mild (80°C) and utilize standard solvents like THF, which are well-understood in large-scale chemical engineering contexts. This facilitates straightforward technology transfer from pilot plant to commercial production scales ranging from hundreds of kilograms to multi-ton batches. Moreover, the reduction in heavy metal usage and hazardous waste generation aligns with increasingly stringent global environmental regulations, future-proofing the supply chain against evolving compliance standards and enhancing the sustainability credentials of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process robustness and scalability.
Q: Why is the Nickel-catalyzed route preferred over Palladium methods for this intermediate?
A: The Nickel-catalyzed route significantly reduces raw material costs by replacing expensive Palladium catalysts with abundant Nickel salts. Furthermore, it avoids the generation of aluminum salt wastewater associated with traditional Friedel-Crafts acylation routes, offering superior environmental compliance and easier waste treatment.
Q: What are the safety advantages of this new synthesis method?
A: Unlike previous routes that utilized pyrophoric Sodium Hydride (NaH) for methylation steps—which poses severe fire risks and limits production during humid weather—this novel method employs sterically hindered bases like NaHMDS under controlled conditions, drastically improving operational safety and allowing for uninterrupted year-round manufacturing.
Q: What yields can be expected from this optimized process?
A: Under optimized conditions using Ni(COD)2, BINAP ligand, and ZnBr2 Lewis acid at 80°C, the process consistently achieves isolated yields exceeding 90%, with specific examples demonstrating up to 92% efficiency, ensuring high throughput for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a new patent must be translated into practical, commercial reality. As a premier CDMO partner, we possess the technical expertise to adapt and optimize complex catalytic pathways like the Nickel-mediated coupling described in CN113292534B. Our facilities are equipped to handle sensitive organometallic chemistry under strict inert atmospheres, ensuring that the high yields observed in the laboratory are replicated consistently in production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, maintaining stringent purity specifications throughout the process. Our rigorous QC labs employ advanced analytical techniques to monitor impurity profiles, guaranteeing that every batch of Bilastine intermediate meets the exacting standards required for global regulatory filings.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us to obtain specific COA data and route feasibility assessments, ensuring that this innovative synthesis method aligns perfectly with your project timelines and quality objectives. Let us help you secure a competitive edge in the antihistamine market through superior chemical manufacturing.
