Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing privileged scaffolds, among which the indole nucleus stands out as a cornerstone structure found in numerous bioactive molecules. As detailed in the recent patent CN112898192B, a novel preparation method for N-acyl indole compounds has been developed that addresses critical bottlenecks in traditional synthetic routes. This technology leverages a palladium-catalyzed carbonylation cyclization strategy, utilizing readily available 2-alkynylanilines and aryl iodides as starting materials. The significance of this advancement cannot be overstated, given that N-acyl indoles serve as pivotal intermediates for a wide array of therapeutic agents, including anti-inflammatory, anti-tumor, and anti-viral drugs. By shifting away from hazardous gaseous reagents towards safer solid alternatives, this process not only enhances laboratory safety but also streamlines the path toward industrial application, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
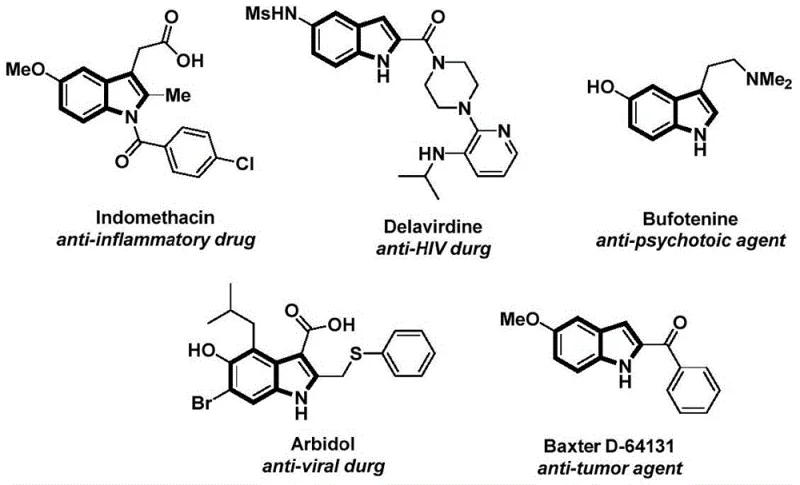
Indole derivatives are ubiquitous in medicinal chemistry, forming the core structure of blockbuster drugs such as Indomethacin, an anti-inflammatory agent, and Delavirdine, used in HIV treatment. The ability to efficiently functionalize the nitrogen atom of the indole ring with an acyl group is a frequent requirement in late-stage drug development and process chemistry. However, conventional methods often suffer from limitations regarding atom economy, safety hazards associated with carbon monoxide gas, or harsh reaction conditions that compromise sensitive functional groups. The methodology disclosed in patent CN112898192B offers a transformative approach by integrating carbonylation and cyclization into a streamlined sequence. This innovation is particularly relevant for R&D directors focused on purity and impurity profiles, as the mild conditions employed minimize side reactions and degradation, thereby simplifying downstream purification processes and ensuring high-quality output for complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing N-acyl indole scaffolds frequently rely on the direct use of carbon monoxide gas under high pressure, which necessitates specialized autoclave equipment and rigorous safety protocols that significantly inflate capital expenditure and operational complexity. Furthermore, many existing protocols require stoichiometric amounts of toxic heavy metals or harsh acidic conditions that are incompatible with base-sensitive substrates, leading to poor yields and difficult waste disposal challenges. The reliance on gaseous CO also introduces variability in mass transfer rates, which can result in inconsistent reaction kinetics and batch-to-batch reproducibility issues, a major concern for supply chain heads managing large-scale production campaigns. Additionally, multi-step sequences often required to install the acyl group post-cyclization increase the overall process time and material loss, driving up the cost of goods sold and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and efficient solid carbon monoxide substitute, effectively eliminating the need for high-pressure gas infrastructure while maintaining high reaction efficiency. This method operates under relatively mild thermal conditions, typically around 60°C, which preserves the integrity of sensitive functional groups such as halogens and esters that might otherwise decompose under more vigorous conditions. The one-pot nature of the reaction, where carbonylation and subsequent cyclization occur sequentially in the same vessel, drastically reduces solvent consumption and manual handling time, aligning perfectly with green chemistry principles. By employing a palladium catalyst system combined with silver oxide as a promoter, the process achieves excellent conversion rates and broad substrate scope, making it a versatile tool for cost reduction in pharmaceutical intermediate manufacturing without sacrificing product quality or safety standards.
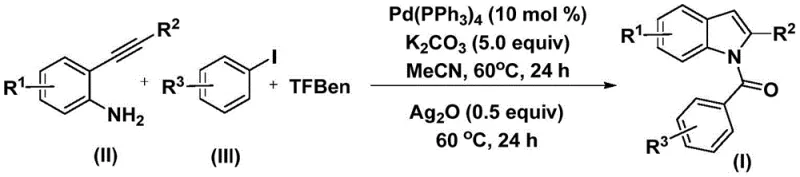
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between palladium catalysis and oxidative cyclization, beginning with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the aryl iodide substrate. This generates a reactive aryl-palladium intermediate which subsequently undergoes migratory insertion of carbon monoxide released in situ from the decomposition of TFBen, forming a key acyl-palladium species. The 2-alkynylaniline then coordinates and inserts into this acyl-palladium bond, followed by reductive elimination to yield an amide intermediate. This sequence is highly dependent on the ligand environment and the precise stoichiometry of the base, typically potassium carbonate, which neutralizes the HI byproduct and drives the equilibrium forward. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters to suppress potential side reactions, such as homocoupling of the alkyne or hydrodehalogenation, ensuring a clean impurity profile essential for regulatory compliance in API production.
Following the formation of the amide intermediate, the addition of silver oxide triggers the final cyclization step, likely through activation of the alkyne moiety or oxidation of the palladium center to facilitate intramolecular nucleophilic attack by the indole nitrogen. This dual-catalyst system ensures that the reaction proceeds to completion with high regioselectivity, favoring the formation of the desired N-acyl indole over potential C-acylated isomers. The tolerance for diverse substituents on the aromatic rings, including electron-withdrawing groups like fluorine and chlorine as well as electron-donating groups like methoxy and methyl, underscores the robustness of this catalytic cycle. For R&D teams, this mechanistic clarity provides a strong foundation for troubleshooting and optimization, enabling the rapid adaptation of this protocol to synthesize novel analogues for structure-activity relationship studies or scale-up campaigns for commercial supply.
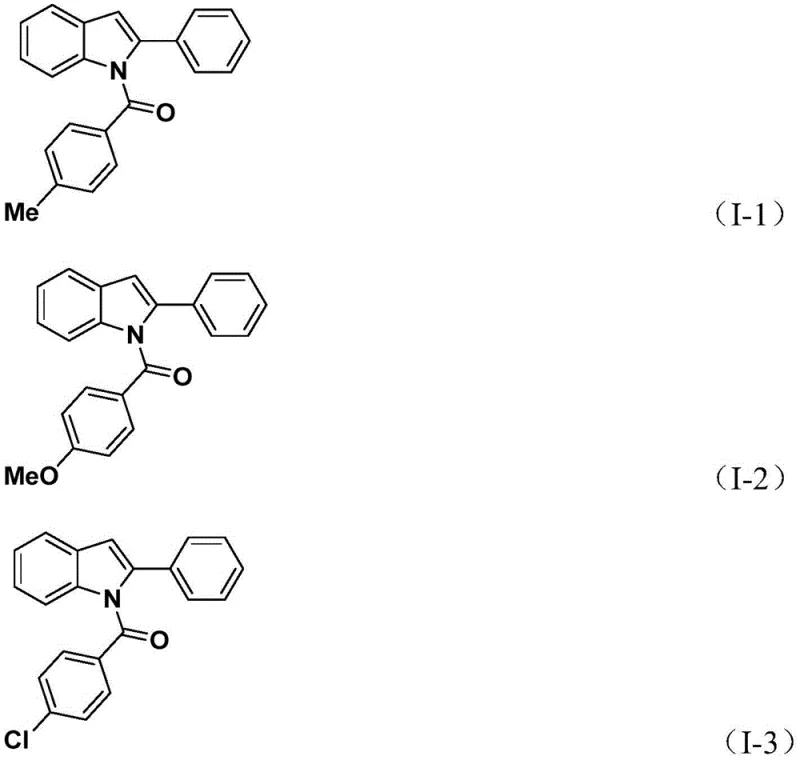
How to Synthesize N-Acyl Indole Compounds Efficiently
The practical execution of this synthesis is designed to be straightforward and accessible, requiring standard laboratory glassware and commercially available reagents that do not demand specialized storage or handling facilities. The protocol typically involves charging a reaction vessel with the palladium catalyst, base, solid CO source, and substrates in a polar aprotic solvent such as acetonitrile, followed by heating to initiate the carbonylation phase. After the initial reaction period, the oxidant is introduced to drive the cyclization, after which the mixture is worked up via simple filtration and purification techniques like column chromatography. This operational simplicity reduces the barrier to entry for adoption in both academic and industrial settings, facilitating the rapid generation of compound libraries. For detailed procedural specifics regarding reagent ratios, temperature profiles, and workup instructions, please refer to the standardized synthesis guide below.
- Combine palladium catalyst, potassium carbonate, solid CO source (TFBen), 2-alkynylaniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling reaction.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial strategic benefits by decoupling production capabilities from the logistical constraints associated with hazardous gases and complex high-pressure reactors. The shift to solid reagents and ambient pressure conditions significantly lowers the barrier for contract manufacturing organizations to adopt this route, thereby increasing the number of qualified vendors and enhancing supply chain resilience against disruptions. Moreover, the use of inexpensive and widely available starting materials, such as substituted aryl iodides and alkynes, ensures a stable raw material supply base that is less susceptible to market volatility compared to exotic or proprietary reagents. This stability translates directly into more predictable pricing models and reliable delivery schedules for downstream pharmaceutical clients who depend on consistent availability of critical intermediates for their own production timelines.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements and the use of a solid carbon monoxide surrogate drastically reduce capital investment and maintenance costs associated with reactor infrastructure. Furthermore, the high atom economy and single-pot nature of the reaction minimize solvent usage and waste generation, leading to significant savings in waste disposal fees and raw material consumption. By avoiding the need for cryogenic conditions or expensive protecting group strategies often required in alternative routes, the overall cost of goods is optimized, allowing for more competitive pricing in the global marketplace for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on shelf-stable solid reagents like TFBen and common inorganic bases mitigates the risks associated with the transportation and storage of compressed gases, which are often subject to strict regulatory controls and shipping delays. This logistical flexibility enables manufacturers to maintain higher inventory levels of key reagents without safety concerns, ensuring continuous production capability even during periods of supply chain stress. Additionally, the robustness of the reaction conditions means that minor variations in utility supplies, such as cooling water temperature or steam pressure, are less likely to cause batch failures, thereby improving overall equipment effectiveness and on-time delivery performance.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic gaseous emissions make this process inherently safer and easier to scale from kilogram to multi-ton quantities without extensive re-engineering of the process flow. The reduced environmental footprint, characterized by lower energy consumption and minimized hazardous waste, aligns with increasingly stringent global environmental regulations and corporate sustainability goals. This compliance advantage not only reduces the risk of regulatory fines but also enhances the brand reputation of manufacturers as responsible partners in the pharmaceutical supply chain, appealing to eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. By addressing these points, we aim to clarify the operational advantages and help stakeholders make informed decisions about integrating this methodology into their existing manufacturing frameworks.
Q: What are the advantages of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment and enhances operational safety, making the process more suitable for standard laboratory and pilot plant settings.
Q: What is the role of Silver Oxide (Ag2O) in the reaction mechanism?
A: Silver oxide acts as an oxidant and promoter in the second stage of the reaction, facilitating the intramolecular cyclization of the amide intermediate to form the final N-acyl indole structure efficiently.
Q: Can this method tolerate diverse functional groups on the substrates?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical industry. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the Pd-catalyzed carbonylation route are successfully translated into robust manufacturing processes. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch consistency and regulatory compliance for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project, whether it involves custom synthesis of novel analogues or the commercial supply of established intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed transformations can drive efficiency and value for your organization.
