Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
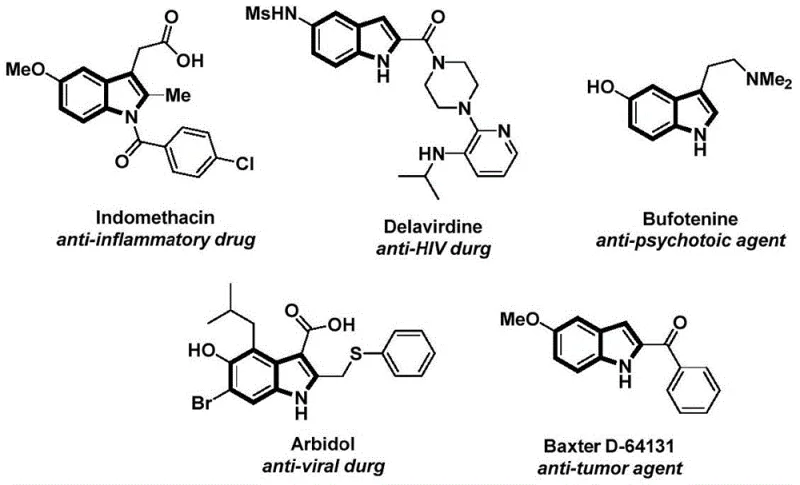
The structural motif of the indole ring is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As depicted in the reference structures of biologically active indoles, the versatility of this heterocycle is unmatched in drug discovery. However, the efficient construction of functionalized N-acyl indoles remains a significant challenge in process chemistry. Patent CN112898192B introduces a groundbreaking preparation method that addresses these synthetic bottlenecks through a novel palladium-catalyzed carbonylative cyclization strategy. This technology leverages 2-alkynyl aniline and aryl iodides as primary building blocks, utilizing a solid carbon monoxide substitute to achieve high-yield transformations under remarkably mild conditions. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this methodology represents a paradigm shift towards more sustainable and cost-effective manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to N-acyl indoles often suffer from severe operational drawbacks that hinder their application in large-scale industrial settings. Classical methods frequently rely on the direct acylation of pre-formed indoles, which can lead to regioselectivity issues, producing mixtures of N-acyl and C-acyl isomers that require difficult and expensive purification steps. Furthermore, many existing carbonylation protocols necessitate the use of gaseous carbon monoxide, a highly toxic and hazardous reagent that demands specialized high-pressure equipment and rigorous safety protocols, significantly inflating capital expenditure and operational risk. Additionally, conventional approaches often involve multi-step sequences with harsh reaction conditions, such as strong acids or elevated temperatures, which can degrade sensitive functional groups and limit the scope of substrates. These inefficiencies result in lower overall atom economy and generate substantial chemical waste, posing challenges for environmental compliance and cost reduction in API manufacturing.
The Novel Approach
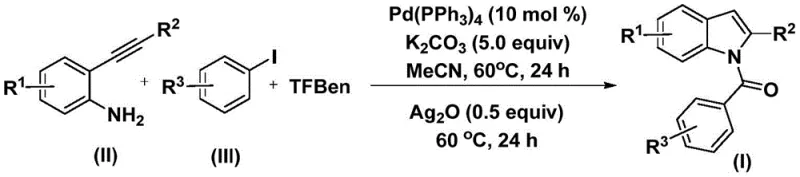
The methodology disclosed in the patent offers a transformative solution by integrating the cyclization and acylation steps into a single, streamlined operation. By employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and solid carbon monoxide surrogate, the process eliminates the need for handling dangerous CO gas, thereby drastically simplifying the reactor setup and enhancing workplace safety. The reaction proceeds efficiently at a moderate temperature of 60°C in acetonitrile, demonstrating exceptional tolerance for a wide range of functional groups including halogens, alkoxy groups, and alkyl chains. This one-pot strategy not only reduces the number of unit operations but also minimizes solvent consumption and waste generation. The use of commercially available palladium catalysts and inexpensive inorganic bases like potassium carbonate further underscores the economic viability of this route, making it an ideal candidate for the commercial scale-up of complex heterocycles required in modern drug development.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay of organometallic steps orchestrated by the palladium catalyst. The cycle initiates with the oxidative addition of the aryl iodide to the zero-valent palladium species, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an intermediate amide. The elegance of this system lies in the second stage, where the addition of silver oxide promotes an oxidative cyclization. The silver salt likely facilitates the activation of the alkyne moiety or acts as an oxidant to regenerate the active catalytic species, driving the intramolecular cyclization to form the final N-acyl indole core. This dual-catalytic or sequential additive approach ensures high conversion rates and minimizes the formation of side products, providing a robust platform for synthesizing diverse libraries of indole derivatives.
From an impurity control perspective, the specificity of the palladium insertion and the controlled release of CO from TFBen are critical for maintaining high product purity. The mild reaction conditions prevent the polymerization of the alkyne starting material, a common side reaction in transition metal catalysis. Furthermore, the use of acetonitrile as the solvent ensures excellent solubility of all reactants and intermediates, facilitating homogeneous reaction kinetics which are easier to monitor and control than heterogeneous systems. The post-treatment process is equally straightforward, involving simple filtration to remove inorganic salts and silver residues, followed by standard column chromatography. This simplicity in workup is a major advantage for process chemists aiming to reduce the environmental footprint of synthesis, as it avoids complex extraction procedures and reduces the volume of organic waste solvents requiring disposal.
How to Synthesize N-Acyl Indole Compounds Efficiently
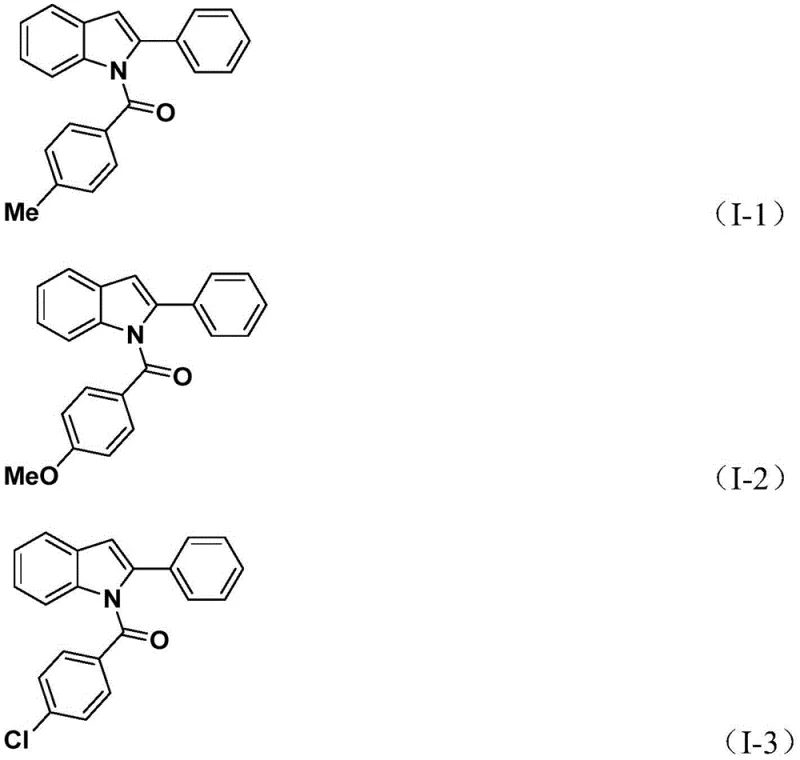
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratory and pilot-scale production. The procedure begins by charging a reaction vessel with the palladium catalyst, potassium carbonate, the CO source TFBen, the 2-alkynyl aniline substrate, and the aryl iodide in acetonitrile. The mixture is stirred and heated to 60°C for 24 hours to allow the initial carbonylation and amidation to reach completion. Following this first stage, silver oxide is introduced directly into the reaction mixture without isolation of intermediates, and the heating is continued for another 24 hours to effect the cyclization. This telescoped approach maximizes throughput and minimizes material loss. Detailed standardized synthesis steps for specific derivatives are provided in the technical guide below.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial coupling and carbonylation.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling strategic benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as aryl iodides and substituted anilines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents. The elimination of high-pressure gas infrastructure reduces the barrier to entry for contract manufacturing organizations (CMOs), allowing for more flexible production scheduling and potentially lower tolling fees. Moreover, the high substrate compatibility means that a single production line can be adapted to manufacture a wide variety of N-acyl indole analogues with minimal changeover time, enhancing asset utilization rates.
- Cost Reduction in Manufacturing: The economic profile of this process is significantly improved by the use of TFBen as a solid CO source, which removes the need for expensive and specialized high-pressure reactors required for gaseous carbon monoxide. The catalyst loading is kept relatively low, and the use of inexpensive inorganic bases like potassium carbonate instead of costly organic bases further drives down the raw material costs. Additionally, the high efficiency and selectivity of the reaction reduce the burden on downstream purification, leading to substantial savings in silica gel, solvents, and labor hours associated with chromatography and recrystallization.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-alkynyl anilines and aryl iodides, are widely available from global chemical suppliers, ensuring consistent availability and reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate moisture and air to a reasonable extent compared to sensitive organometallic reactions, simplifies logistics and storage requirements. This reliability allows for better demand forecasting and inventory management, ensuring that critical pharmaceutical intermediates can be delivered to clients with reduced lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and the absence of hazardous gases, making it safer to operate at the multi-kilogram or ton scale. The simplified workup procedure, which primarily involves filtration and standard purification, generates less aqueous and organic waste compared to traditional multi-step syntheses. This aligns with modern green chemistry principles and helps manufacturing partners meet increasingly stringent environmental regulations, avoiding potential fines and reputational damage associated with poor waste management practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this N-acyl indole synthesis method?
A: The method utilizes cheap and readily available starting materials like 2-alkynyl aniline and aryl iodides, operates under mild conditions (60°C), and avoids the use of toxic high-pressure carbon monoxide gas by using TFBen as a CO surrogate.
Q: What is the role of Silver Oxide (Ag2O) in this reaction?
A: Silver oxide is added in the second stage of the reaction to promote the oxidative cyclization of the intermediate amide, ensuring the formation of the final N-acyl indole structure with high efficiency.
Q: Is this process suitable for large-scale production?
A: Yes, the process features simple operation, good substrate compatibility, and uses common organic solvents like acetonitrile, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert process chemists has extensively evaluated the palladium-catalyzed carbonylation technology described in CN112898192B and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole derivative meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize your supply chain. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects, ensuring a secure and competitive supply of these vital pharmaceutical building blocks.
