Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Pharmaceutical Manufacturing
Introduction to Advanced N-Acyl Indole Synthesis
The structural motif of the indole ring is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to antiviral drugs such as Delavirdine and anti-tumor compounds. As illustrated in the diverse pharmacological profiles presented in 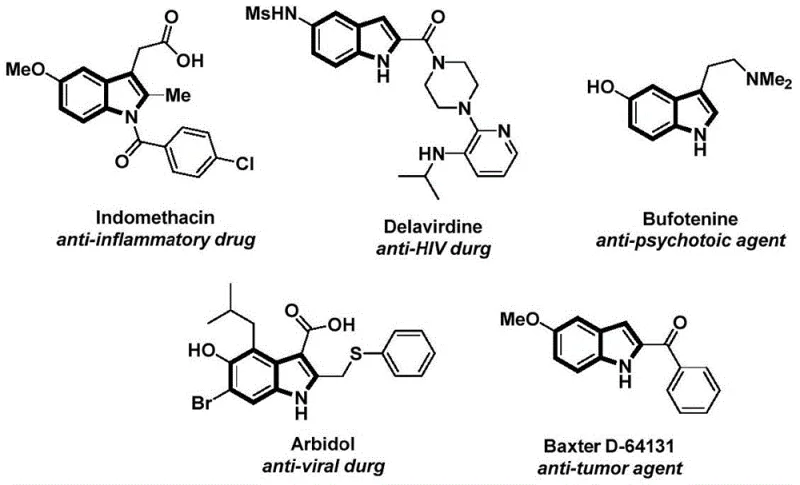 , the functionalization of the indole nitrogen atom is a critical transformation for modulating biological activity and physicochemical properties. Patent CN112898192B discloses a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This novel protocol utilizes a palladium-catalyzed carbonylation cyclization strategy, leveraging readily available 2-alkynyl anilines and aryl iodides to construct the N-acyl indole backbone in a single, streamlined operation. For pharmaceutical manufacturers and R&D directors seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in process intensification, offering a robust pathway to high-value heterocyclic intermediates without the logistical burdens associated with traditional high-pressure carbonylation techniques.
, the functionalization of the indole nitrogen atom is a critical transformation for modulating biological activity and physicochemical properties. Patent CN112898192B discloses a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This novel protocol utilizes a palladium-catalyzed carbonylation cyclization strategy, leveraging readily available 2-alkynyl anilines and aryl iodides to construct the N-acyl indole backbone in a single, streamlined operation. For pharmaceutical manufacturers and R&D directors seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in process intensification, offering a robust pathway to high-value heterocyclic intermediates without the logistical burdens associated with traditional high-pressure carbonylation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles via carbonylation has been hindered by significant practical and safety constraints. Traditional methods often rely on the direct use of carbon monoxide gas, which is highly toxic and requires specialized high-pressure reactors to achieve sufficient solubility and reaction rates. These stringent conditions not only escalate capital expenditure for manufacturing facilities but also introduce complex safety protocols that can slow down production timelines and increase operational risks. Furthermore, conventional palladium-catalyzed routes frequently suffer from limited substrate scope, where sensitive functional groups on the aromatic rings may degrade under harsh thermal or pressure conditions. The necessity for expensive ligands or stoichiometric amounts of toxic metal promoters in older methodologies further complicates the downstream purification processes, leading to lower overall yields and higher waste generation. Consequently, the industry has long sought a milder, safer, and more versatile alternative that maintains high reaction efficiency while simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology described in patent CN112898192B revolutionizes this landscape by employing a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), which releases CO in situ under mild thermal conditions. As depicted in the general reaction scheme in 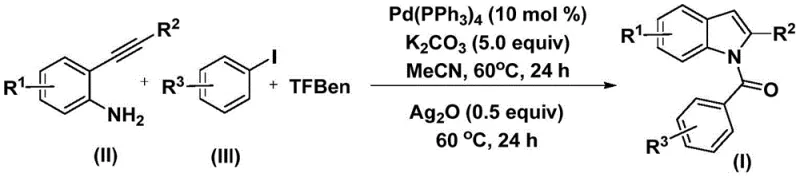 , this approach allows the reaction to proceed at a moderate temperature of 60°C in an open or standard sealed vessel, completely eliminating the need for high-pressure gas infrastructure. The process involves a tandem sequence where the palladium catalyst first mediates the coupling of the aryl iodide with the in situ generated CO, followed by cyclization with the 2-alkynyl aniline. A distinct feature of this novel approach is the sequential addition of silver oxide in the second stage, which acts as a crucial promoter for the final cyclization step, ensuring high conversion and selectivity. This two-stage, one-pot protocol not only enhances safety by avoiding gaseous CO but also dramatically improves the atom economy and operational simplicity, making it an ideal candidate for cost reduction in API manufacturing where safety and efficiency are paramount.
, this approach allows the reaction to proceed at a moderate temperature of 60°C in an open or standard sealed vessel, completely eliminating the need for high-pressure gas infrastructure. The process involves a tandem sequence where the palladium catalyst first mediates the coupling of the aryl iodide with the in situ generated CO, followed by cyclization with the 2-alkynyl aniline. A distinct feature of this novel approach is the sequential addition of silver oxide in the second stage, which acts as a crucial promoter for the final cyclization step, ensuring high conversion and selectivity. This two-stage, one-pot protocol not only enhances safety by avoiding gaseous CO but also dramatically improves the atom economy and operational simplicity, making it an ideal candidate for cost reduction in API manufacturing where safety and efficiency are paramount.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensure high fidelity in bond formation. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Simultaneously, the thermal decomposition of the solid CO source, TFBen, releases carbon monoxide into the reaction medium, which subsequently inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate is then intercepted by the amine group of the 2-alkynyl aniline, undergoing a nucleophilic attack to form an amide linkage while regenerating the palladium catalyst for the next turnover. The structural versatility of this mechanism is evident in the tolerance for various substituents on the starting materials, as defined by the general formulas (II) and (III) shown in  and
and  , allowing for the incorporation of electron-donating and electron-withdrawing groups without compromising the catalytic cycle. The final cyclization to form the indole ring, facilitated by silver oxide, likely involves the activation of the alkyne moiety or the stabilization of the transition state, driving the equilibrium towards the thermodynamically stable N-acyl indole product shown in formula (I) in
, allowing for the incorporation of electron-donating and electron-withdrawing groups without compromising the catalytic cycle. The final cyclization to form the indole ring, facilitated by silver oxide, likely involves the activation of the alkyne moiety or the stabilization of the transition state, driving the equilibrium towards the thermodynamically stable N-acyl indole product shown in formula (I) in 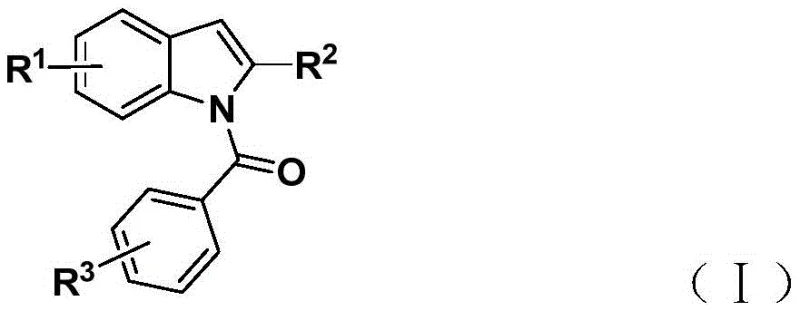 . This precise control over the reaction trajectory minimizes the formation of regioisomers or oligomeric byproducts, resulting in a clean impurity profile that is highly desirable for regulatory compliance in drug substance manufacturing.
. This precise control over the reaction trajectory minimizes the formation of regioisomers or oligomeric byproducts, resulting in a clean impurity profile that is highly desirable for regulatory compliance in drug substance manufacturing.
Furthermore, the choice of silver oxide as a secondary additive plays a pivotal role in the reaction's success, acting not merely as a base but potentially as a soft Lewis acid that coordinates with the alkyne or the carbonyl oxygen to facilitate the intramolecular cyclization. This dual-catalyst system (Pd/Ag) ensures that even sterically hindered substrates can undergo cyclization efficiently, a common bottleneck in indole synthesis. The use of acetonitrile as the solvent further optimizes the solubility of both the organic substrates and the inorganic salts, creating a homogeneous reaction environment that maximizes mass transfer and heat distribution. By understanding these mechanistic nuances, process chemists can fine-tune reaction parameters such as catalyst loading and stoichiometry to achieve optimal yields, as demonstrated by the broad scope of examples where yields consistently exceed 50% and reach up to 82% for optimized substrates. This deep mechanistic understanding provides a solid foundation for scaling the process from gram-scale laboratory synthesis to multi-kilogram pilot production with predictable outcomes.
How to Synthesize N-Acyl Indole Efficiently
The synthesis of N-acyl indole compounds via this palladium-catalyzed protocol is designed for operational ease and reproducibility, making it accessible for both research and production environments. The procedure typically begins with the charging of a reaction vessel with the palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, along with potassium carbonate as the base and the solid CO source TFBen. To this mixture, the 2-alkynyl aniline and the aryl iodide are added in acetonitrile, and the system is heated to 60°C for an initial period of 24 hours to allow for the carbonylation and amidation steps to reach completion. Following this first stage, silver oxide is introduced directly into the reaction mixture without the need for intermediate workup, and the heating is continued for another 24 hours to drive the cyclization to completion.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent like acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to promote cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere chemical yield. The shift from gaseous carbon monoxide to a solid surrogate fundamentally alters the risk profile of the manufacturing process, removing the need for specialized high-pressure autoclaves and the associated regulatory hurdles for handling toxic gases. This simplification of infrastructure translates directly into reduced capital expenditure and lower insurance costs, facilitating faster deployment of production lines for new drug candidates. Moreover, the use of commercially available starting materials such as aryl iodides and 2-alkynyl anilines ensures a robust and resilient supply chain, as these precursors are widely sourced from global chemical vendors, mitigating the risk of raw material shortages. The mild reaction conditions (60°C) also imply lower energy consumption compared to high-temperature alternatives, contributing to a smaller carbon footprint and aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of a solid CO source significantly lower the barrier to entry for producing these valuable intermediates. By avoiding the complexities of gas handling systems, manufacturers can reduce both CAPEX and OPEX, while the high substrate compatibility minimizes the need for expensive protecting group strategies, leading to substantial cost savings in the overall synthetic route.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like TFBen and common aryl halides ensures that production schedules are not disrupted by the logistics of hazardous gas delivery. The robustness of the reaction across a wide range of substituents means that supply chain disruptions for specific substituted anilines can often be mitigated by switching to alternative analogs without re-optimizing the entire process, providing flexibility in sourcing.
- Scalability and Environmental Compliance: The one-pot nature of the reaction reduces solvent usage and waste generation associated with intermediate isolation steps. The mild conditions and absence of toxic gas emissions make the process inherently safer and more environmentally compliant, simplifying the permitting process for scale-up and ensuring continuity of supply in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this N-acyl indole synthesis method. These insights are derived directly from the experimental data and scope studies detailed in the patent literature, providing a clear picture of the method's capabilities and limitations for potential adopters. Understanding these aspects is crucial for process development teams evaluating the feasibility of this route for their specific target molecules.
Q: What are the advantages of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup compared to traditional carbonylation methods.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent substrate tolerance, accommodating various functional groups including halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and alkoxy groups on both the 2-alkynyl aniline and aryl iodide starting materials.
Q: How does the two-stage temperature profile affect the reaction yield?
A: Maintaining a consistent mild temperature of 60°C for both the initial coupling (24 hours) and the subsequent silver oxide-mediated cyclization (24 hours) ensures high conversion rates and minimizes side reactions, leading to isolated yields ranging from 44% to 82%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-acyl indole intermediate meets the exacting standards required by global regulatory bodies. Our commitment to technological innovation allows us to offer customized synthesis solutions that leverage advanced methodologies like the palladium-catalyzed carbonylation described herein, providing our partners with a competitive edge in time-to-market.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this synthetic route for their specific projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to contact us today to discuss your specific needs,索取 specific COA data, and receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies.
