Revolutionizing Aminoanisole Production: A Deep Dive into Clean Catalytic Hydrogenation Processes
The global demand for high-purity aromatic amines, specifically aminoanisoles, continues to surge as they serve as critical building blocks for a vast array of pharmaceutical active ingredients and agrochemical formulations. In response to the industry's pressing need for greener and more efficient manufacturing pathways, patent CN1861570A introduces a transformative technology for preparing amino benz methyl-phenoxide (aminoanisole) through the catalytic hydrogenation of nitro methyl-phenoxide mixtures. This innovation represents a paradigm shift away from legacy reduction chemistries that have long plagued the fine chemical sector with severe environmental liabilities. By leveraging advanced heterogeneous catalysis in a methanol solvent system, this process not only achieves exceptional conversion rates but also fundamentally alters the economic and ecological footprint of aminoanisole production. For R&D directors and technical decision-makers, understanding the nuances of this patent is essential, as it offers a robust framework for scaling up production while adhering to increasingly stringent global environmental regulations. The technology described herein provides a comprehensive solution that addresses both the quality imperatives of modern drug synthesis and the cost-efficiency mandates of contemporary chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-aminoanisole and 4-aminoanisole has relied heavily on stoichiometric reduction methods, primarily utilizing sodium sulfide or iron powder as the reducing agents. These conventional pathways are fraught with significant operational and environmental drawbacks that render them increasingly obsolete in a modern regulatory landscape. The use of sodium sulfide, for instance, generates substantial quantities of sulfur-containing wastewater, which is notoriously difficult and expensive to treat due to its high chemical oxygen demand and toxicity. Similarly, iron powder reduction produces massive volumes of iron sludge, a solid waste stream that requires complex disposal procedures and often leads to secondary pollution risks if not managed with extreme care. Furthermore, these traditional methods often suffer from poor selectivity and lower product quality, necessitating extensive downstream purification steps that erode overall process yield. The corrosive nature of the reaction byproducts also accelerates equipment degradation, leading to higher maintenance costs and unplanned downtime, thereby compromising supply chain reliability for downstream pharmaceutical manufacturers who depend on consistent intermediate availability.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in patent CN1861570A employs a catalytic hydrogenation strategy that fundamentally eliminates the generation of stoichiometric inorganic waste. By utilizing hydrogen gas as the clean reducing agent in the presence of efficient catalysts such as Raney-Ni or Pd-C, the process converts nitro groups directly to amino groups with water as the primary byproduct. This approach drastically simplifies the workup procedure, as there is no need to filter out tons of iron sludge or treat sulfide-laden effluent. The use of methanol as a solvent further enhances the process efficiency by reducing the viscosity of the reaction system, which improves the dispersion of the catalyst and facilitates superior heat transfer during the exothermic hydrogenation reaction. This novel route not only ensures a safer operating environment by mitigating the risks associated with handling hazardous reducing agents but also delivers a product profile with significantly higher purity. The ability to operate in either continuous fixed-bed reactors or batch stirred tanks provides unparalleled flexibility, allowing manufacturers to tailor the production scale to market demand without sacrificing process control or product consistency.
Mechanistic Insights into Raney-Ni Catalyzed Hydrogenation
The core of this technological advancement lies in the sophisticated interaction between the nitroanisole substrate and the heterogeneous catalyst surface under hydrogen pressure. In this mechanism, hydrogen molecules dissociate on the active sites of the Raney-Ni or Pd-C catalyst, forming reactive atomic hydrogen species that subsequently attack the nitrogen-oxygen bonds of the nitro group. The reaction proceeds through a series of intermediate reduction steps, ultimately yielding the desired amine functionality while preserving the sensitive methoxy ether linkage on the aromatic ring. The choice of methanol as the solvent is mechanistically critical; beyond merely dissolving the reactants, it acts as a heat sink to manage the exotherm of the hydrogenation, preventing thermal runaway that could lead to side reactions or catalyst deactivation. Moreover, the solvent properties of methanol facilitate the mass transfer of hydrogen gas into the liquid phase, ensuring that the catalyst surface remains saturated with reactants for optimal turnover frequency. This precise control over reaction kinetics is what enables the single-pass conversion rates to exceed 85%, a metric that underscores the high efficiency of the catalytic system compared to diffusion-limited stoichiometric reductions.
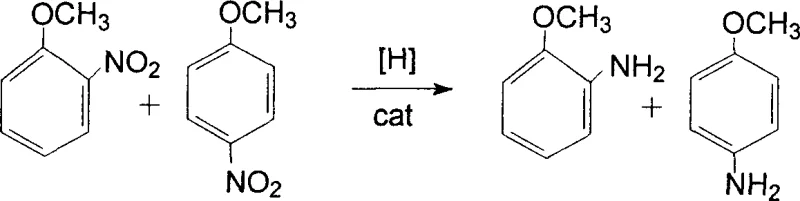
Following the reduction phase, the process incorporates a rigorous purification protocol designed to eliminate trace impurities and colored byproducts that often compromise the quality of fine chemical intermediates. The addition of adsorption agents such as activated carbon, diatomaceous earth, or molecular sieves serves as a critical polishing step, capturing residual catalyst fines and organic impurities through physical adsorption mechanisms. This decolorization operation is conducted at controlled temperatures between 40°C and 80°C to maximize adsorption capacity without inducing product degradation. Subsequent solid-liquid separation via filtration ensures that the liquid phase contains only the dissolved aminoanisole isomers, methanol, and water, setting the stage for the final isolation step. The separation of the ortho and para isomers is achieved through a clever exploitation of their differential solubility profiles in methanol at varying temperatures. By cooling the solution to specific crystallization points, such as 10°C for the para-isomer and 0°C for the ortho-isomer, the process achieves high-purity separation without the need for energy-intensive distillation columns, thereby preserving the thermal stability of the sensitive amine products.
How to Synthesize Aminoanisole Efficiently
The implementation of this synthesis route requires a coordinated sequence of unit operations that balance reaction kinetics with separation thermodynamics to maximize yield and purity. The process begins with the preparation of the feed solution, where the nitroanisole mixture is dissolved in methanol and pumped into the reactor system containing the pre-activated catalyst. Detailed standard operating procedures regarding pressure ramping, temperature control, and hydrogen flow rates are critical to maintaining the safety and efficiency of the hydrogenation step. Following the reaction completion, indicated by the cessation of hydrogen uptake, the mixture undergoes a specialized filtration process to recover the valuable catalyst for reuse, minimizing raw material consumption. The subsequent purification and crystallization stages require precise thermal management to ensure the selective precipitation of the target isomers. For a comprehensive breakdown of the specific operational parameters, equipment specifications, and safety protocols required to execute this synthesis at scale, please refer to the standardized technical guide provided below.
- Dissolve the nitroanisole mixture in methanol and introduce hydrogen gas in the presence of a Raney-Ni or Pd-C catalyst within a fixed-bed or stirred reactor.
- Execute solid-liquid separation to recover the catalyst using a blade filter system, allowing the catalyst to be recycled for subsequent batches.
- Purify the crude product through adsorption decolorization followed by fractional crystallization to separate 2-aminoanisole and 4-aminoanisole isomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this catalytic hydrogenation technology translates into tangible strategic advantages that extend far beyond simple unit cost calculations. The elimination of heavy metal waste streams and sulfur contaminants significantly reduces the burden on wastewater treatment facilities, leading to substantial operational expenditure savings related to environmental compliance and waste disposal fees. Furthermore, the high efficiency of the catalyst recovery system, which allows for repeated recycling of the Raney-Ni or Pd-C, drastically lowers the consumption of expensive noble or transition metals, directly impacting the variable cost of goods sold. The robustness of the process equipment, particularly the use of corrosion-resistant fixed-bed reactors, ensures longer asset life and reduced maintenance intervals, contributing to a more predictable and reliable supply schedule. By transitioning to this cleaner technology, companies can mitigate the risk of production stoppages caused by environmental regulatory crackdowns, thereby securing a more resilient supply chain for their downstream pharmaceutical customers who prioritize sustainability in their vendor selection criteria.
- Cost Reduction in Manufacturing: The shift from stoichiometric reagents to catalytic hydrogenation eliminates the need for purchasing large volumes of iron powder or sodium sulfide, which are subject to volatile commodity pricing. Additionally, the recovery and regeneration of the decolorization agents and the high-efficiency recycling of the catalyst create a closed-loop system that minimizes raw material loss. The simplified downstream processing, which avoids complex extraction and neutralization steps associated with traditional methods, reduces utility consumption including steam and cooling water, further driving down the overall manufacturing cost structure.
- Enhanced Supply Chain Reliability: The versatility of the reactor design, which supports both batch and continuous operations, allows manufacturers to scale production capacity rapidly in response to market fluctuations without requiring entirely new infrastructure. The use of methanol, a widely available and globally traded solvent, ensures that raw material sourcing remains stable and unaffected by regional supply disruptions. Moreover, the high selectivity of the reaction reduces the formation of hard-to-remove impurities, decreasing the likelihood of batch failures and ensuring a consistent flow of high-quality intermediates to the production line.
- Scalability and Environmental Compliance: This process is inherently designed for industrial scale-up, with reactor configurations that maintain consistent heat and mass transfer characteristics from pilot plant to full commercial production. The significant reduction in hazardous waste generation aligns perfectly with green chemistry principles, facilitating easier permitting and regulatory approval in jurisdictions with strict environmental laws. The ability to integrate with onsite hydrogen generation technologies further enhances the self-sufficiency of the production facility, reducing dependence on external gas suppliers and optimizing the overall carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic hydrogenation technology for aminoanisole production. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the feasibility of this route. Understanding these details is crucial for technical teams assessing the compatibility of this process with existing manufacturing assets and for commercial teams negotiating supply agreements based on quality and sustainability metrics.
Q: What are the primary environmental benefits of this hydrogenation process compared to traditional reduction methods?
A: Unlike traditional sodium sulfide or iron powder reduction methods which generate massive amounts of toxic wastewater and iron sludge, this catalytic hydrogenation process utilizes methanol as a clean solvent and produces minimal hazardous waste, significantly reducing environmental compliance costs.
Q: How does the process ensure high purity of the final aminoanisole products?
A: The process incorporates a dedicated adsorption decolorization step using agents like activated carbon or molecular sieves, followed by precise temperature-controlled crystallization, which effectively separates the ortho and para isomers to achieve purities exceeding 98%.
Q: Is this technology suitable for large-scale continuous production?
A: Yes, the patent explicitly supports the use of three-phase fixed-bed or fluidized bed reactors, which are inherently designed for continuous operation, offering superior heat transfer and scalability compared to batch processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like the one described in patent CN1861570A requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from laboratory concept to industrial reality. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aminoanisole delivered meets the exacting standards required for pharmaceutical synthesis. We are committed to delivering not just a chemical product, but a comprehensive value proposition that includes process optimization, regulatory support, and unwavering supply security.
We invite you to engage with our technical procurement team to discuss how we can tailor this catalytic hydrogenation process to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener production method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and sustainability in your supply chain.
