Optimizing Ibrutinib Manufacturing: Advanced Catalytic Routes For Commercial Scale-Up And Purity Control
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with Bruton's tyrosine kinase (Btk) inhibitors representing a cornerstone in managing hematological malignancies. Patent CN113816962A provides a critical technical disclosure regarding the synthesis of ibrutinib, detailing innovative pathways that address longstanding challenges in constructing the complex pyrazolo-pyrimidine scaffold. This document serves as a vital resource for understanding how modern catalytic methods can replace legacy stoichiometric approaches, offering a roadmap for manufacturers seeking to enhance process efficiency. By leveraging these disclosed methodologies, production facilities can achieve superior control over stereochemistry and impurity profiles, which are paramount for regulatory approval. The strategic implementation of these synthetic routes allows for a more robust supply chain capable of meeting the escalating global demand for high-quality kinase inhibitors without compromising on safety or efficacy standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the biaryl ether linkage found in ibrutinib often rely on harsh nucleophilic aromatic substitution conditions that require elevated temperatures and strong bases. These aggressive parameters frequently lead to the formation of difficult-to-remove byproducts and can compromise the integrity of sensitive functional groups elsewhere in the molecule. Furthermore, legacy processes often involve multiple protection and deprotection steps that drastically increase the overall step count and reduce the cumulative yield of the manufacturing campaign. The reliance on expensive palladium catalysts in some conventional cross-coupling methods also introduces significant cost burdens and necessitates rigorous metal scavenging operations to meet strict residual metal limits. Consequently, these inefficiencies translate into higher production costs and extended lead times, creating bottlenecks for procurement teams aiming to secure reliable inventory levels for clinical and commercial needs.
The Novel Approach
In contrast, the methodology outlined in the patent introduces a streamlined coupling strategy that utilizes copper catalysis to forge the critical carbon-oxygen bond under milder reaction conditions. This approach significantly reduces the thermal stress on the intermediate molecules, thereby preserving chiral integrity and minimizing degradation pathways that generate complex impurity spectra. The use of readily available phenylboronic acids or halide precursors allows for greater flexibility in sourcing raw materials, which enhances supply chain resilience against market fluctuations. Additionally, the process design incorporates efficient workup procedures that facilitate the removal of catalytic residues without requiring extensive chromatographic purification. This technological shift represents a paradigm change in process chemistry, enabling manufacturers to produce high-purity intermediates with a reduced environmental footprint and improved economic viability for large-scale operations.
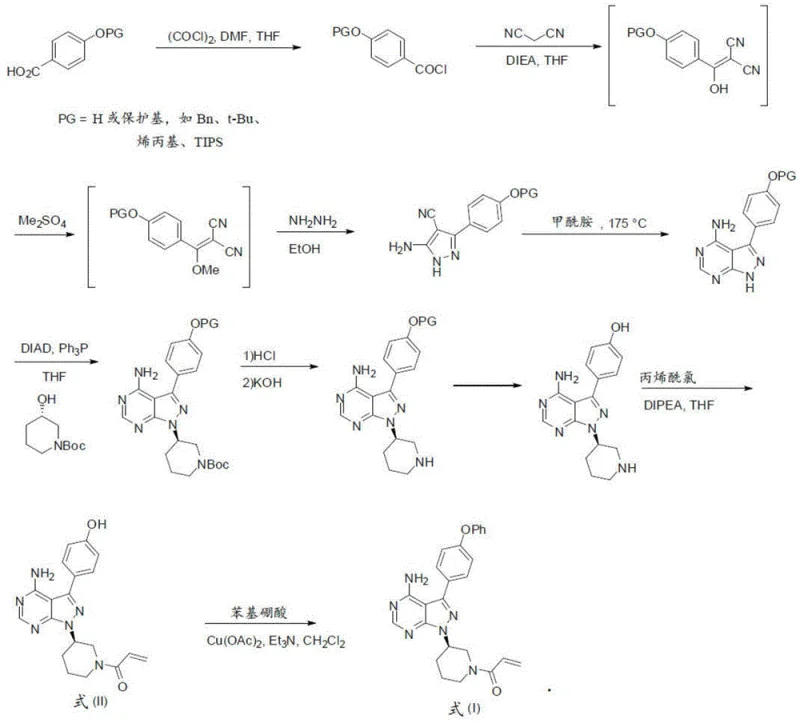
Mechanistic Insights into Copper-Catalyzed Biaryl Ether Formation
The core of this innovative synthesis lies in the mechanistic elegance of the copper-catalyzed coupling reaction between the hydroxyl-substituted pyrazolo-pyrimidine intermediate and the phenyl derivative. The catalytic cycle likely involves the oxidative addition of the copper species to the aryl halide or the activation of the boronic acid, followed by coordination with the phenolic oxygen of the heterocyclic core. This coordination facilitates the transmetallation or substitution event that constructs the biaryl ether bond with high regioselectivity, ensuring that the reaction occurs exclusively at the desired position on the aromatic ring. The presence of specific ligands or base additives further stabilizes the catalytic complex, preventing the precipitation of inactive copper species and maintaining turnover numbers throughout the reaction duration. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters such as temperature and stoichiometry to maximize yield while suppressing the formation of homocoupled side products.
Impurity control is meticulously addressed through the selection of reaction solvents and quenching agents that selectively partition unwanted byproducts into the aqueous phase. The patent describes specific washing protocols using citric acid and carbonate solutions that effectively remove residual amines and acidic impurities generated during the coupling and subsequent elimination steps. This level of detail in the purification strategy ensures that the final product maintains a clean impurity profile, which is essential for passing stringent quality control tests required by regulatory agencies. Moreover, the process avoids the use of genotoxic reagents wherever possible, aligning with modern safety guidelines for pharmaceutical manufacturing. By integrating these purification logic into the early stages of process development, manufacturers can avoid costly reprocessing steps later in the production timeline, ensuring a smoother path from kilogram-scale batches to commercial tonnage.
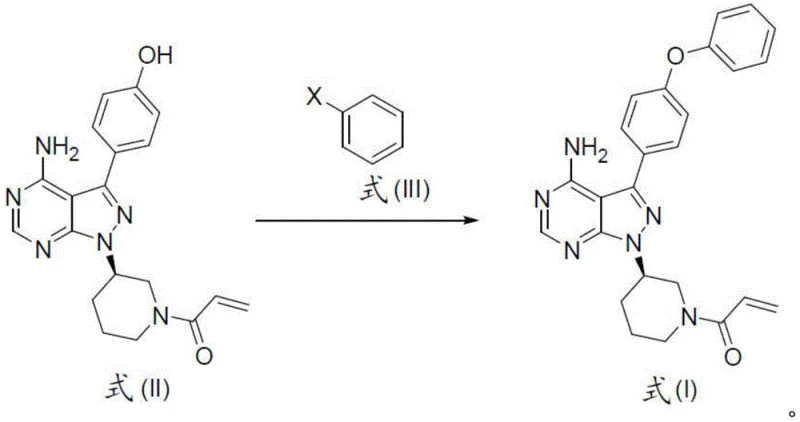
How to Synthesize Ibrutinib Efficiently
Executing this synthesis requires precise adherence to the reaction conditions specified to ensure reproducibility and safety across different manufacturing scales. The process begins with the preparation of the key heterocyclic intermediate, followed by the critical coupling step that installs the phenoxyphenyl moiety essential for biological activity. Operators must maintain strict control over reaction temperatures and addition rates, particularly during the exothermic acylation and elimination phases, to prevent thermal runaways that could compromise product quality. The detailed standardized synthesis steps see the guide below provide a framework for translating these laboratory discoveries into robust manufacturing protocols. Adhering to these guidelines ensures that the final active pharmaceutical ingredient meets all necessary specifications for potency and purity.
- Prepare the pyrazolo-pyrimidine core intermediate (Formula II) through condensation and cyclization reactions ensuring high regioselectivity.
- Execute the key coupling reaction between Formula II and phenylboronic acid or halide derivatives using copper catalysis to form the biaryl ether linkage.
- Perform beta-elimination on the acylated piperidine precursor (Formula XVII) using non-nucleophilic bases like DBU to generate the terminal acrylamide moiety.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial benefits that directly impact the bottom line and operational reliability of pharmaceutical supply chains. The reduction in step count and the use of cheaper, more abundant catalysts translate into significant cost savings in raw material procurement and waste disposal management. Procurement managers will find that the reliance on commodity chemicals rather than specialized proprietary reagents reduces exposure to supply disruptions and price volatility in the fine chemical market. Furthermore, the improved yield and purity profiles mean that less starting material is required to produce the same amount of final API, effectively increasing the capacity of existing manufacturing infrastructure without capital investment. These efficiencies create a more competitive pricing structure for the final drug product, allowing healthcare providers to access life-saving medications more affordably while maintaining healthy margins for manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts in favor of copper-based systems drastically lowers the cost of goods sold associated with catalytic materials. By avoiding complex purification steps required to remove trace palladium, the process reduces solvent consumption and processing time, leading to lower utility and labor costs per kilogram of output. This economic advantage is compounded by the higher overall yield, which maximizes the value extracted from every batch of raw materials purchased. Consequently, the total manufacturing cost is significantly optimized, providing a strong buffer against market pressures and enabling more aggressive pricing strategies in competitive tender processes.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that production schedules are not held hostage by the limited availability of exotic reagents. This accessibility allows for the qualification of multiple secondary suppliers for key inputs, thereby de-risking the supply chain against single-source failures or geopolitical disruptions. The robustness of the chemical process also means that batch failure rates are minimized, ensuring a consistent flow of material to downstream formulation sites. For supply chain heads, this reliability translates into better inventory management and the ability to commit to long-term delivery contracts with confidence, knowing that production capacity is stable and scalable.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents that are easier to recover and recycle, thus reducing the environmental burden of manufacturing operations. The mild reaction conditions reduce energy consumption for heating and cooling, aligning with corporate sustainability goals and regulatory requirements for carbon footprint reduction. Scalability is inherent in the design, as the reaction kinetics remain favorable even when transitioning from pilot reactors to large-scale production vessels. This ensures that increasing volume to meet market demand does not introduce new technical challenges or safety risks, facilitating a smooth ramp-up to commercial supply levels.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in a GMP environment. These answers are derived directly from the technical disclosures and experimental data provided within the patent documentation to ensure accuracy and relevance. Understanding these aspects is critical for stakeholders evaluating the feasibility of adopting this route for their own manufacturing portfolios. The insights provided here aim to clarify the operational implications and quality benefits associated with this advanced chemical process.
Q: How does the novel copper-catalyzed coupling improve impurity profiles compared to traditional methods?
A: The patented process utilizes specific copper salts and base combinations that minimize side reactions such as homocoupling or over-oxidation, resulting in a cleaner crude profile that simplifies downstream purification and reduces solvent consumption during recrystallization.
Q: What are the scalability advantages of the beta-elimination step described in the patent?
A: The elimination protocol employs commercially available reagents like DBU and ethyl acetate under mild temperature conditions, avoiding cryogenic requirements or hazardous reagents, which significantly enhances operational safety and facilitates seamless transfer from pilot plant to multi-ton commercial production.
Q: Can this synthesis route support the stringent purity specifications required for oncology APIs?
A: Yes, the route incorporates robust crystallization steps and aqueous workup procedures specifically designed to remove residual metals and organic impurities, ensuring the final active pharmaceutical ingredient meets rigorous international pharmacopoeia standards for genotoxic impurities and residual solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring complex synthetic routes like this to life on a global scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Ibrutinib intermediate or API complies with international regulatory standards. Our commitment to quality assurance means that we proactively manage impurity profiles and residual solvents, delivering products that are ready for immediate formulation without additional remediation.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis technology to your specific volume and cost requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this optimized manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate our capabilities against your project milestones. Let us collaborate to secure a sustainable and efficient supply chain for your oncology portfolio.
