Advanced Synthesis of Carbon-14 Labeled Ibrutinib for Environmental and Pharmacokinetic Studies
The pharmaceutical industry faces increasing regulatory pressure to understand the environmental fate of active pharmaceutical ingredients (APIs) post-excretion. Patent CN113024565B introduces a groundbreaking methodology for the synthesis of radioactive isotope carbon-14 labeled ibrutinib, a critical tool for addressing these environmental safety concerns. This innovation provides a robust tracer agent essential for basic research into the environmental safety problems associated with ibrutinib, a widely used Bruton's Tyrosine Kinase (BTK) inhibitor. By enabling precise tracking of tissue distribution, metabolite structure identification, and mass balance, this technology supports comprehensive radioactive pharmacokinetics research. The method ensures that the labeled atoms remain firmly attached to the molecular skeleton, thereby eliminating the paradoxical information often obtained from less stable isotope labeling tests. This technical advancement represents a significant leap forward for pharmaceutical companies aiming to comply with stringent environmental regulations while maintaining rigorous drug safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbon-14 labeled ibrutinib has relied on labeling the carbonyl carbon within the acryloyl fragment of the molecule. While this conventional approach offers short synthesis steps and relatively low initial costs, it suffers from severe technical and safety drawbacks that hinder its utility in long-term environmental studies. The primary intermediate required, carbonyl-14C acryloyl chloride, is a low-boiling point substance that is highly volatile during experimental operations. This volatility creates a substantial risk of inadvertent inhalation by experimenters, leading to potential internal irradiation hazards that compromise laboratory safety standards. Furthermore, the trace amounts of this substance are prone to deterioration, which can affect the consistency and reliability of the final labeled product. Most critically, the carbon-14 labeled acryloyl fragment in these literature-reported markers is chemically unstable and can easily detach from the molecular skeleton during metabolic processes. This detachment results in incomplete tracking data, making it impossible to accurately monitor the motion change trace of the main fragment of the molecule, thus rendering the isotope labeling test ineffective for rigorous environmental safety analysis.
The Novel Approach
In stark contrast to previous methodologies, the novel approach detailed in the patent selects the pyrazolo [3,4-d] pyrimidine fragment within the ibrutinib molecule as the labeling unit, specifically targeting the 3-position carbon atom. This strategic selection leverages the inherent aromaticity and chemical stability of the pyrazolo pyrimidine core, ensuring that the carbon-14 label is firm and not easy to fall off during complex biological or environmental tracing tests. The synthesis route begins with cost-effective [14C] barium carbonate as the starting radioisotope material, which is converted into carbon dioxide for subsequent carboxylation. This process avoids the use of hazardous volatile intermediates, as all radioactive gas handling is conducted in a strictly sealed system that prevents escape. The resulting intermediates are viscous pastes or non-sublimable solids, which drastically reduces the risk of operator exposure to radioactive materials. By securing the label at a metabolically stable site, this method provides viable, economical, and safe access to high-quality tracers that can reveal accurate movement change information of ibrutinib in complex systems.
Mechanistic Insights into C-14 Carboxylation and Cyclization
The core of this synthesis lies in the precise construction of the labeled pyrazolo pyrimidine ring system through a series of controlled chemical transformations. The process initiates with the reaction of 4-phenoxyphenyl lithium with [14C] carbon dioxide generated from barium carbonate at low temperatures below -70°C, yielding 4-phenoxybenzene [14C] formic acid with high radiochemical efficiency. This labeled acid is subsequently converted into an acyl chloride and condensed with malononitrile in the presence of sodium hydride to form a key malononitrile intermediate. The mechanism then proceeds through a methylation step using trimethylsilyldiazomethane, followed by a cyclization reaction with hydrazine hydrate to construct the pyrazole ring. Finally, the pyrimidine ring is closed via reaction with formamide at elevated temperatures. Each step is optimized to minimize the loss of radioactive material, ensuring that the isotopic integrity is maintained throughout the multi-step sequence. The use of inert gas protection and specific temperature controls at each stage prevents side reactions that could compromise the position of the label or reduce the overall yield.
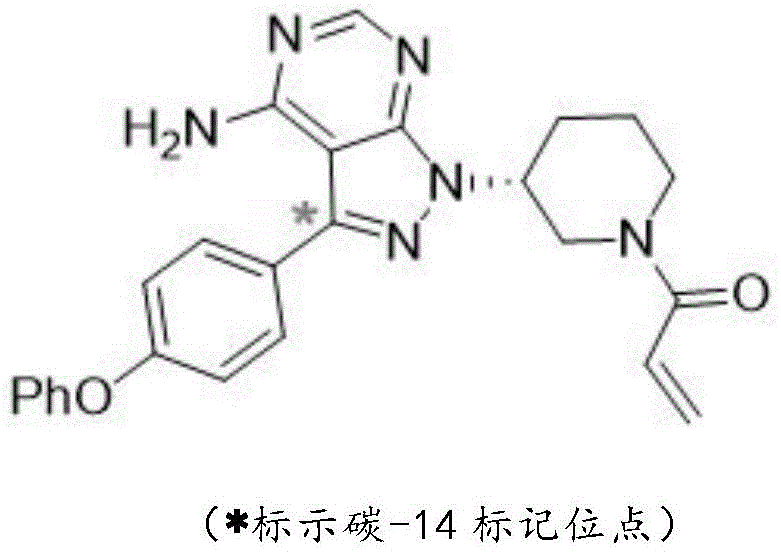
Controlling the impurity profile in radiochemical synthesis is paramount, especially when the final product is intended for sensitive pharmacokinetic studies. The described method incorporates rigorous purification protocols, including conventional post-treatment and column chromatography at multiple stages, to ensure the removal of unlabeled byproducts and chemical impurities. The stability of the label at the 3-position of the pyrazolo pyrimidine fragment means that the impurity spectrum is significantly cleaner compared to methods labeling the acryloyl group, where degradation products often mimic the tracer signal. By avoiding the use of volatile radioactive intermediates, the process also reduces the risk of cross-contamination in the synthesis equipment, which is a common source of isotopic impurities in other methods. The final product achieves a chemical purity of more than 98% and a radiochemical purity of more than 99%, meeting the stringent specifications required for regulatory submission and high-level research. This high level of purity ensures that the data generated from environmental safety studies is accurate and free from artifacts caused by label detachment or impurity interference.
How to Synthesize Carbon-14 Labeled Ibrutinib Efficiently
Implementing this synthesis route requires a deep understanding of radiochemical handling and multi-step organic synthesis under inert conditions. The process is designed to be scalable while maintaining the high specific activity required for tracer studies. Operators must adhere to strict safety protocols regarding the handling of [14C] barium carbonate and the generation of radioactive carbon dioxide. The detailed standardized synthesis steps involve eight distinct stages, ranging from initial carboxylation to the final acryloylation of the piperidine moiety. Each stage requires precise control of reaction parameters such as temperature, stoichiometry, and reaction time to maximize the radiochemical yield. For a comprehensive breakdown of the specific reagents, conditions, and workup procedures required to replicate this high-purity synthesis, please refer to the standardized guide provided below.
- Carboxylation of 4-phenoxyphenyl lithium with C-14 CO2 to form labeled formic acid.
- Condensation with malononitrile and cyclization to form the pyrazolo pyrimidine core.
- Mitsunobu reaction and deprotection to attach the piperidine moiety and final acryloyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers substantial advantages over traditional methods, primarily driven by enhanced safety and operational efficiency. The elimination of volatile radioactive intermediates translates directly into reduced safety infrastructure costs, as specialized containment for high-volatility gases is not required to the same extent. This simplification of the safety profile allows for more flexible manufacturing setups and reduces the regulatory burden associated with handling hazardous radioactive materials. Furthermore, the use of barium carbonate as a starting material provides a cost-effective entry point for the synthesis, avoiding the premium pricing often associated with more complex labeled building blocks. The robustness of the chemical steps ensures consistent batch-to-batch quality, which is critical for maintaining supply continuity in long-term environmental monitoring programs. These factors combine to create a supply chain that is both resilient and economically viable for large-scale research initiatives.
- Cost Reduction in Manufacturing: The synthesis route achieves cost optimization by utilizing readily available starting materials and eliminating the need for expensive purification steps associated with volatile intermediates. By avoiding the use of low-boiling point radioactive substances, the process reduces the loss of valuable isotopic material due to evaporation or degradation, thereby improving the overall material efficiency. The high radiochemical yield across the multi-step sequence ensures that the cost per unit of the final labeled product is significantly lower than methods with poor retention of the isotope. Additionally, the stability of the intermediates allows for simpler storage and handling requirements, further reducing overhead costs related to specialized containment and monitoring. These qualitative efficiencies contribute to a more sustainable cost structure for the production of complex radiochemical tracers.
- Enhanced Supply Chain Reliability: The operational safety of this method directly enhances supply chain reliability by minimizing the risk of production stoppages due to safety incidents. Since the process avoids hazardous volatile intermediates, the likelihood of accidental release or contamination events that could shut down a facility is drastically reduced. The use of stable solid or viscous intermediates facilitates easier logistics and transportation of materials between synthesis stages, ensuring a smoother workflow. This stability also means that the synthesis is less sensitive to minor fluctuations in environmental conditions, leading to more predictable production schedules. For procurement managers, this translates into a dependable source of high-purity labeled compounds that can be delivered consistently to support critical research timelines without unexpected delays.
- Scalability and Environmental Compliance: This synthesis method is inherently designed for scalability, with reaction conditions that can be adapted from laboratory to commercial scale without significant re-engineering. The sealed system approach for carbon dioxide handling ensures that emissions are contained, aligning with strict environmental compliance standards for radioactive waste management. The reduction in hazardous waste generation, due to the absence of volatile radioactive byproducts, simplifies the disposal process and reduces the environmental footprint of the manufacturing operation. This alignment with environmental, social, and governance (ESG) goals makes the supply of these intermediates more attractive to multinational corporations with rigorous sustainability mandates. The ability to scale while maintaining high purity and safety standards ensures that the supply chain can grow to meet increasing demand for environmental safety data.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of carbon-14 labeled ibrutinib. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data. Understanding these details is crucial for research directors and procurement specialists evaluating the feasibility of incorporating this tracer into their drug development or environmental safety programs. The information provided here clarifies the stability advantages, safety protocols, and scalability potential of this specific synthesis route.
Q: Why is the 3-position pyrazolo pyrimidine chosen for C-14 labeling?
A: The 3-position within the aromatic pyrazolo pyrimidine fragment offers superior chemical and metabolic stability compared to carbonyl labeling, preventing isotope detachment during environmental tracing.
Q: What are the safety advantages of this synthesis method?
A: The process utilizes a strictly sealed system for C-14 CO2 handling and avoids volatile radioactive intermediates like acryloyl chloride, significantly reducing internal irradiation risks for operators.
Q: Is this method suitable for large-scale environmental safety studies?
A: Yes, the route uses cost-effective starting materials like Barium Carbonate-14 and provides high radiochemical purity, making it viable for extensive mass balance and metabolite identification studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering unparalleled expertise in translating complex patent routes like CN113024565B into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even the most intricate radiochemical syntheses can be manufactured with precision and consistency. We understand the critical nature of stringent purity specifications in pharmaceutical research, and our rigorous QC labs are equipped to verify every batch against the highest international standards. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and regulatory compliance, allowing you to focus on your core research objectives without worrying about material quality or availability.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our capabilities, we are ready to provide comprehensive route feasibility assessments. Our goal is to become your strategic partner in securing high-quality intermediates that drive your innovation forward. Contact us today to request an evaluation and discover how our manufacturing prowess can enhance your research and development efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
