Advanced Zeolite-Catalyzed Synthesis of 4-Aminopyrrolo[2,1-f][1,2,4]triazine for Scalable Antiviral Production
Advanced Zeolite-Catalyzed Synthesis of 4-Aminopyrrolo[2,1-f][1,2,4]triazine for Scalable Antiviral Production
The rapid evolution of antiviral therapeutics has placed immense pressure on the global supply chain for critical heterocyclic intermediates, particularly those required for nucleoside analogs like Remdesivir. Patent CN114478533A introduces a transformative methodology for the preparation of 4-aminopyrrolo[2,1-f][1,2,4]triazine, a pivotal building block in the synthesis of broad-spectrum antiviral agents. This innovation addresses longstanding bottlenecks in medicinal chemistry by replacing hazardous stoichiometric reagents with a robust, heterogeneous zeolite catalytic system. For R&D directors and procurement strategists, this shift represents not merely a synthetic variation but a fundamental restructuring of the production landscape, offering a pathway to higher yields, reduced environmental liabilities, and enhanced supply chain resilience. The technology leverages the unique acidic properties of zeolite molecular sieves to drive the condensation of readily available precursors, effectively bypassing the multi-step, waste-intensive protocols that have historically plagued this sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminopyrrolo[2,1-f][1,2,4]triazine has been fraught with operational inefficiencies and safety hazards that complicate commercial scale-up. Prior art, such as the method disclosed in CN 111533747 A, relies on the use of chlorosulfonic acid isocyanate for cyanation and O-[4-nitro-2-(trifluoromethyl)phenyl]hydroxylamine for amination. These reagents are not only expensive and hazardous to handle on a metric-ton scale but also generate substantial quantities of toxic waste, leading to yield limitations around 66%. Furthermore, alternative routes like those in CN110092787A require prolonged reflux times of up to 16 hours in ethanol, resulting in mediocre yields of approximately 55.9%. The WO2007056170 protocol exacerbates these issues with reaction times extending to 18 hours and the generation of massive amounts of phosphate solid waste due to the excessive use of potassium phosphate. These legacy processes create significant burdens for supply chain heads, who must manage complex waste streams and endure long cycle times that restrict production throughput.
The Novel Approach
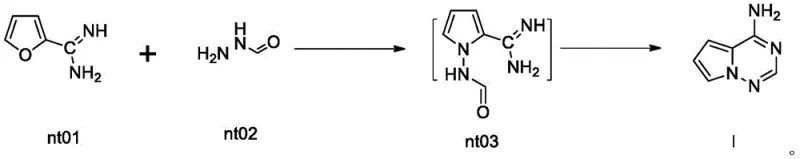
In stark contrast, the methodology outlined in CN114478533A streamlines the synthesis into a direct, catalytic condensation between compound nt01 and compound nt02. By utilizing zeolite molecular sieves—specifically medium silica types like H-Y or high silica types like H-ZSM-5—as the catalyst, the process eliminates the need for dangerous stoichiometric reagents and harsh acidic conditions. The reaction proceeds efficiently at temperatures between 100°C and 200°C, completing within a mere 0.5 to 5 hours, which is a drastic reduction compared to the day-long cycles of conventional methods. This approach not only boosts yields to over 80% but also simplifies the workup procedure to a simple filtration and recrystallization. The ability to recycle the solid catalyst further enhances the economic viability of this route, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize cost structures without compromising quality.
![Reaction scheme showing the novel synthesis of 4-aminopyrrolo[2,1-f][1,2,4]triazine from nt01 and nt02](/insights/img/4-aminopyrrolo-triazine-synthesis-pharma-supplier-20260313084845-06.webp)
Mechanistic Insights into Zeolite-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise activation of the reactants by the Brønsted acid sites present within the zeolite framework. When compound nt01 and compound nt02 interact within the porous structure of the catalyst, the zeolite facilitates a concerted cyclization mechanism that avoids the formation of unstable intermediates common in solution-phase chemistry. The reaction likely proceeds through a transition state, designated as compound nt03 in the patent literature, where the formamidine moiety of nt02 attacks the nitrile group of nt01, followed by intramolecular cyclization and aromatization. The shape-selective nature of the zeolite pores may also play a role in suppressing side reactions, thereby enhancing the selectivity for the desired 4-aminopyrrolo[2,1-f][1,2,4]triazine scaffold. This mechanistic efficiency is critical for R&D teams focused on impurity control, as it minimizes the generation of closely related structural analogs that are difficult to separate.
Furthermore, the heterogeneous nature of the catalysis ensures that the active sites remain distinct from the product stream, preventing product degradation that often occurs with strong liquid acids. The stability of the zeolite lattice under the reaction conditions (up to 200°C) allows for consistent performance over multiple batches. For quality assurance professionals, this translates to a highly reproducible process where batch-to-batch variability is minimized. The mechanism supports a clean reaction profile, evidenced by HPLC data showing purity levels exceeding 99% after simple recrystallization. This level of purity is essential for downstream coupling reactions in the synthesis of complex nucleoside therapeutics, where trace impurities can poison subsequent catalytic steps or compromise the safety profile of the final API.
How to Synthesize 4-Aminopyrrolo[2,1-f][1,2,4]triazine Efficiently
Implementing this novel synthesis route requires careful attention to catalyst selection and reaction parameters to maximize the benefits of the zeolite system. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment beyond standard stainless steel reactors capable of handling mild nitrogen pressure. Operators should focus on maintaining the optimal molar ratio of reactants, typically between 1:1 and 1:2, to ensure complete conversion while minimizing excess reagent waste. The choice of solvent, whether toluene, ethanol, or DMF, can be tuned based on solubility profiles and downstream processing requirements, offering flexibility for different manufacturing setups. Detailed standardized synthesis steps see the guide below.
- Charge a reactor with compound nt01, compound nt02, a solvent such as toluene or ethanol, and a zeolite molecular sieve catalyst (e.g., H-Y or H-ZSM-5).
- Heat the reaction mixture to a temperature between 100°C and 200°C under nitrogen pressure and stir for 0.5 to 5 hours to facilitate cyclization.
- Filter the reaction mixture to recover the solid catalyst, concentrate the filtrate, and purify the crude product via recrystallization with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zeolite-catalyzed process offers a compelling value proposition centered on cost reduction in pharmaceutical intermediate manufacturing and risk mitigation. The elimination of hazardous reagents like chlorosulfonic acid isocyanate removes the need for specialized storage facilities and expensive safety protocols, directly lowering overhead costs. Additionally, the shift from stoichiometric reagents to a catalytic system drastically reduces the volume of raw materials required per kilogram of product, creating substantial cost savings in material procurement. The simplified workup, which replaces complex aqueous extractions and salt removals with a straightforward filtration, significantly reduces solvent consumption and energy usage during concentration steps. These factors combine to create a leaner, more cost-effective production model that enhances margin potential for high-volume contracts.
- Cost Reduction in Manufacturing: The transition to a recyclable heterogeneous catalyst fundamentally alters the cost structure of production. By recovering and reusing the zeolite molecular sieve multiple times without significant loss of activity, the effective cost of the catalyst per batch approaches zero over time. This stands in sharp contrast to traditional methods that consume expensive phosphates or amines in stoichiometric quantities, generating waste that incurs disposal fees. The reduction in waste treatment costs, combined with the higher reaction yields, leads to a significant decrease in the overall cost of goods sold, allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, compound nt01 and compound nt02, ensures a robust supply chain free from the volatility associated with specialized, hazardous reagents. The short reaction time of 0.5 to 5 hours dramatically increases plant throughput, allowing manufacturers to respond rapidly to surges in demand for antiviral intermediates. This agility is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on schedule. The robustness of the process also means fewer batch failures, guaranteeing a continuous flow of material to customers.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method generates minimal waste, primarily consisting of the solid catalyst which is non-hazardous and recyclable, and avoids the generation of heavy metal or phosphate-laden wastewater. This aligns perfectly with green chemistry principles and simplifies the permitting process for commercial scale-up of complex pharmaceutical intermediates. Facilities can operate with a smaller environmental footprint, reducing the risk of regulatory shutdowns and enhancing the corporate sustainability profile for partners in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zeolite-catalyzed synthesis. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement officers negotiating supply agreements based on process efficiency.
Q: What are the advantages of using zeolite molecular sieves over traditional catalysts for this synthesis?
A: Zeolite molecular sieves, such as H-ZSM-5 and H-Y types, offer significant advantages including heterogeneous catalysis which allows for easy filtration and recycling. Unlike homogeneous acid catalysts or stoichiometric reagents that generate large amounts of salt waste, zeolites minimize environmental impact and reduce downstream purification costs while maintaining high catalytic activity over multiple cycles.
Q: How does the purity of the product compare to prior art methods?
A: The novel zeolite-catalyzed process described in patent CN114478533A achieves high purity levels, with HPLC analysis showing results exceeding 99%. This is a substantial improvement over older methods which often required complex workups to remove inorganic salts and byproducts, thereby streamlining the supply chain for high-purity pharmaceutical intermediates.
Q: Is the catalyst reusable in this industrial process?
A: Yes, the solid zeolite catalyst can be recovered by simple filtration after the reaction. Experimental data indicates that the catalyst retains good activity even after being recycled and reused multiple times, which significantly lowers the cost of goods sold (COGS) and enhances the sustainability profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminopyrrolo[2,1-f][1,2,4]triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN114478533A can be fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 4-aminopyrrolo[2,1-f][1,2,4]triazine meets the exacting standards required for antiviral drug synthesis. Our commitment to quality ensures that your downstream processes remain uninterrupted by impurity-related issues.
We invite you to collaborate with us to optimize your supply chain for next-generation antiviral therapeutics. By leveraging our expertise in zeolite catalysis and process intensification, we can help you achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
