Advanced Enzymatic Resolution of Paroxetine Intermediates for Commercial Scale Manufacturing
Advanced Enzymatic Resolution of Paroxetine Intermediates for Commercial Scale Manufacturing
The global pharmaceutical landscape is increasingly demanding sustainable and efficient synthetic routes for high-volume antidepressants, particularly for key intermediates like those used in Paroxetine production. Patent CN101974604A introduces a groundbreaking biocatalytic methodology that leverages ionic liquids as a green reaction medium to resolve racemic piperidine derivatives. This technology addresses critical bottlenecks in traditional manufacturing, such as low yields and hazardous solvent usage, by employing lipase-catalyzed transesterification under mild conditions. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener, more cost-effective API intermediate production, offering a robust alternative to legacy chemical resolution processes that have long plagued the supply chain with inefficiency and environmental liability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral paroxetine intermediates has relied heavily on chemical resolution techniques utilizing resolving agents like (-)-di-p-toluoyl tartaric acid in volatile organic solvents such as acetone or toluene. As illustrated in the reaction scheme below, these traditional pathways suffer from inherent thermodynamic limitations, typically capping the theoretical yield at 50% and often achieving practical total yields of only 34% to 40% after neutralization. Furthermore, the reliance on large volumes of toxic, flammable solvents creates significant safety hazards and imposes heavy costs related to solvent recovery and waste disposal. The use of harsh chemicals not only complicates regulatory compliance but also introduces potential impurities that require extensive downstream purification, thereby elongating the production cycle and increasing the overall cost of goods sold for the final active pharmaceutical ingredient.
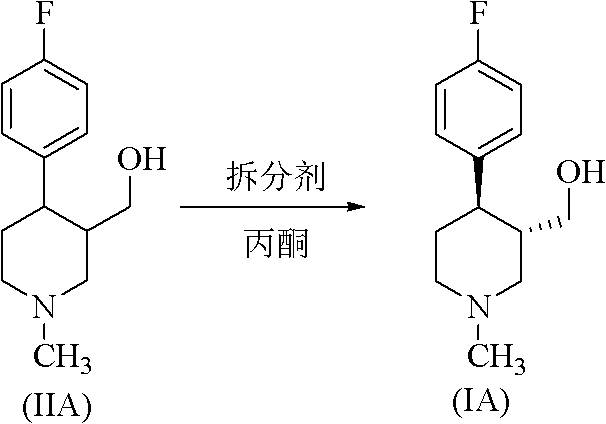
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic system where lipase enzymes facilitate the kinetic resolution of the racemic substrate within a stable ionic liquid matrix. This method eliminates the need for stoichiometric amounts of chiral resolving agents and replaces hazardous volatile organic compounds with non-flammable, designable ionic liquids like [BMIm][PF6]. The enzymatic process operates under significantly milder conditions, typically between 15°C and 60°C, which preserves the integrity of sensitive functional groups and reduces energy consumption. By shifting from a thermodynamic crystallization process to a kinetic enzymatic transformation, manufacturers can achieve superior stereocontrol and higher effective throughput, fundamentally altering the economic model of producing high-purity pharmaceutical intermediates while adhering to strict environmental, health, and safety (EHS) standards.
Mechanistic Insights into Lipase-Catalyzed Transesterification in Ionic Liquids
The core of this technological advancement lies in the precise stereoselectivity of lipases, such as Lipase B from Candida antarctica, which preferentially acylate one enantiomer of the racemic 4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine over the other. In the presence of an acyl donor like vinyl acetate, the enzyme catalyzes a transesterification reaction that converts the unwanted enantiomer into an ester, leaving the desired (3S, 4R)-alcohol unreacted or vice versa depending on the specific enzyme configuration. The ionic liquid medium plays a dual role: it acts as a solvent that maintains high substrate solubility and simultaneously stabilizes the enzyme's tertiary structure, preventing denaturation and enhancing operational stability. This synergistic interaction allows the biocatalyst to maintain high activity over extended periods, facilitating repeated batch cycles without significant loss of performance, which is a critical factor for industrial scalability.
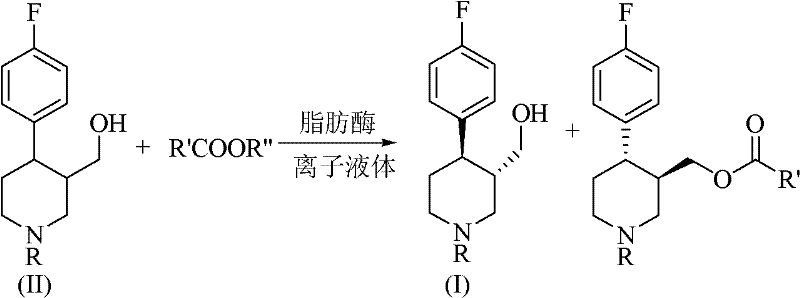
From an impurity control perspective, the specificity of the biological catalyst ensures that side reactions common in chemical catalysis, such as over-acylation or ring-opening, are virtually eliminated. The reaction progress can be meticulously monitored via HPLC, allowing for precise endpoint determination to maximize the enantiomeric excess (ee), which the patent reports can reach up to 98%. This high level of purity at the intermediate stage drastically reduces the burden on downstream purification processes, ensuring that the final API meets stringent pharmacopeial standards with minimal additional processing. The ability to tune the ionic liquid's properties by altering anions or cations further allows chemists to optimize the reaction environment for specific substrate batches, providing a level of process flexibility that rigid chemical methods cannot match.
How to Synthesize Paroxetine Intermediate Efficiently
Implementing this enzymatic resolution protocol requires careful attention to the preparation of the reaction mixture and the selection of optimal biocatalysts. The process begins by establishing a homogeneous solution of the racemic substrate in the chosen ionic liquid, followed by the addition of the acyl donor and the immobilized or free enzyme. Maintaining consistent agitation and temperature control is vital to ensure mass transfer efficiency and enzyme activity throughout the reaction duration. While the patent provides specific experimental parameters, scaling this process requires validation of mixing dynamics and heat transfer characteristics in larger reactors. The detailed standardized synthesis steps for implementing this green chemistry route are outlined in the guide below.
- Prepare the reaction system by dissolving racemic 4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine in an ionic liquid medium such as [BMIm][PF6] at a concentration of 10-150 mmol/L.
- Add the acyl donor, preferably vinyl acetate, at a molar ratio of 1: 10 relative to the substrate, and introduce the lipase catalyst, such as Lipase B from Candida antarctica.
- Maintain the reaction at 15-60°C with stirring at 100-250 rpm for 2-24 hours, then separate the enzyme and extract the product using a hexane/isopropanol mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic ionic liquid technology offers profound strategic benefits beyond mere technical superiority. The elimination of toxic volatile solvents like toluene and acetone simplifies regulatory compliance and reduces the costs associated with hazardous waste management and solvent purchasing. Moreover, the reusability of both the ionic liquid medium and the biocatalyst creates a circular economy within the manufacturing process, leading to substantial reductions in raw material consumption. This efficiency translates directly into a more resilient supply chain, as the process is less susceptible to fluctuations in the availability of specialized chemical resolving agents and is safer to operate, minimizing the risk of production stoppages due to safety incidents.
- Cost Reduction in Manufacturing: The enzymatic process significantly lowers production costs by removing the need for expensive chiral resolving agents and reducing solvent consumption through recycling. The high conversion rates and optical purity minimize the need for costly recrystallization steps and reduce material loss, leading to a more economical overall process. Additionally, the mild reaction conditions reduce energy expenditures related to heating and cooling, further enhancing the cost-efficiency profile of the manufacturing operation.
- Enhanced Supply Chain Reliability: By utilizing robust biocatalysts and stable ionic liquids, the manufacturing process becomes less dependent on volatile organic solvent markets and hazardous chemical supply chains. The ability to recycle the reaction medium ensures a consistent supply of processed material without the delays associated with solvent procurement and disposal. This stability allows for more predictable production scheduling and inventory management, ensuring that critical pharmaceutical intermediates are available to meet market demand without interruption.
- Scalability and Environmental Compliance: The green nature of this technology aligns perfectly with increasingly stringent global environmental regulations, facilitating easier permitting and expansion of production capacity. The non-flammable and non-volatile characteristics of ionic liquids make the process inherently safer for large-scale operations, reducing insurance premiums and safety infrastructure costs. Furthermore, the high selectivity of the enzyme reduces the generation of chemical waste, supporting corporate sustainability goals and enhancing the brand reputation of the manufacturing entity as a responsible producer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this method for commercial production. Understanding these details is crucial for stakeholders evaluating the integration of this green chemistry approach into their existing manufacturing portfolios.
Q: What are the advantages of using ionic liquids over traditional organic solvents in this resolution process?
A: Ionic liquids offer negligible vapor pressure, non-flammability, and superior thermal stability compared to toxic solvents like toluene or acetone. They enhance enzyme stability and stereoselectivity while allowing for solvent recycling, significantly reducing environmental pollution and operational hazards.
Q: What optical purity can be achieved using this enzymatic method?
A: According to the patent data, the enzymatic resolution method can achieve an optical purity of up to 98% ee for the desired (3S, 4R)-enantiomer, which is significantly higher than the yields obtained from traditional chemical resolution methods.
Q: Is the biocatalyst reusable in this ionic liquid system?
A: Yes, the patent highlights that both the enzyme and the ionic liquid medium can be recycled and reused. This reusability contributes to lower production costs and aligns with green chemistry principles by minimizing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic resolution technology described in patent CN101974604A for the production of high-quality paroxetine intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for antidepressant API synthesis. We are committed to leveraging advanced biocatalytic methods to deliver superior value to our global partners.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full commercial potential of this green synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this technology can optimize your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of critical pharmaceutical intermediates.
