Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as Carbon Source
Introduction: A Breakthrough in Biomass-Derived Heterocycle Synthesis
The pharmaceutical and fine chemical industries are constantly seeking sustainable, efficient pathways to access privileged heterocyclic scaffolds. Patent CN113880781A introduces a transformative methodology for the preparation of 3-trifluoromethyl-substituted 1,2,4-triazole compounds, a core structure prevalent in numerous bioactive molecules. As illustrated in the structural diversity of modern therapeutics, the 1,2,4-triazole ring is a critical pharmacophore found in blockbuster drugs such as Sitagliptin and various kinase inhibitors, underscoring the immense commercial value of efficient access to this motif.
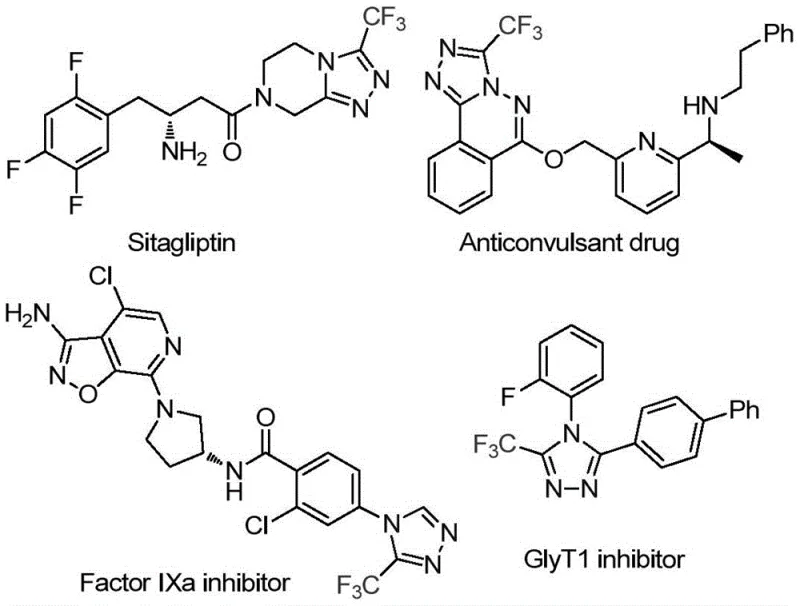
This patented approach distinguishes itself by leveraging D-glucose, a ubiquitous and renewable biomass resource, as the primary carbon source. Unlike traditional petrochemical-derived routes, this method capitalizes on the natural abundance of glucose to generate key aldehyde intermediates in situ. The process is characterized by its operational simplicity, utilizing a trifluoromethanesulfonic acid-catalyzed cascade reaction that proceeds under relatively mild thermal conditions. For R&D directors and procurement specialists alike, this represents a significant shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has relied on multi-step sequences involving hazardous reagents and stringent reaction conditions. Conventional protocols often necessitate the use of pre-functionalized aldehyde precursors, which can be expensive and unstable, requiring careful storage and handling. Furthermore, many existing cyclization strategies demand anhydrous and anaerobic environments to prevent side reactions or catalyst deactivation, thereby increasing the complexity of the reactor setup and the overall energy consumption of the process. The reliance on transition metal catalysts in some traditional routes also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet the rigorous purity specifications required for API manufacturing.
The Novel Approach
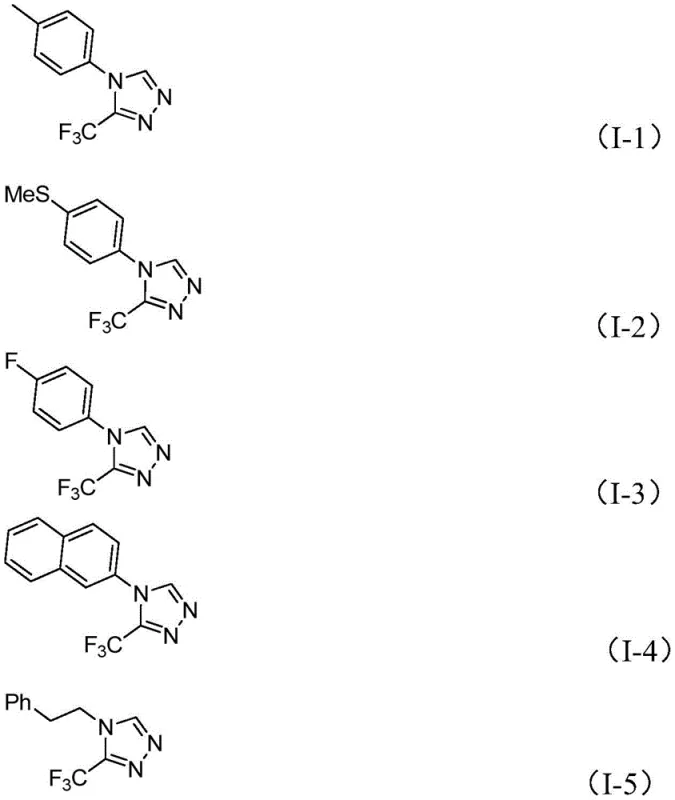
In stark contrast, the method disclosed in CN113880781A offers a streamlined, one-pot solution that bypasses these logistical hurdles. By employing glucose as a latent aldehyde source, the process eliminates the need for isolating sensitive aldehyde intermediates. The reaction utilizes a robust catalytic system comprising trifluoromethanesulfonic acid and tert-butyl hydroperoxide, which facilitates a smooth cascade from hydrazone formation to cyclization and final aromatization. This approach not only tolerates the presence of water but actually incorporates it as a beneficial additive, fundamentally altering the safety and cost profile of the synthesis. The substrate scope is remarkably broad, accommodating various aryl and phenethyl substituents, as demonstrated by the successful synthesis of derivatives I-1 through I-5, making it a versatile platform for library generation.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
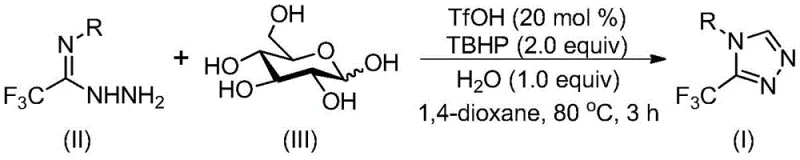
The elegance of this synthesis lies in its mechanistic pathway, which efficiently converts a simple sugar into a complex nitrogen-rich heterocycle. The reaction initiates with the acid-catalyzed cleavage of glucose, generating reactive aldehyde species in situ. These aldehydes immediately undergo condensation with trifluoroacetimidoyl hydrazide to form a hydrazone intermediate. This step is crucial as it sets the stage for the subsequent intramolecular nucleophilic addition. The presence of the strong acid catalyst, trifluoromethanesulfonic acid, is pivotal in activating the glucose backbone and promoting the condensation equilibrium towards the hydrazone.
Following hydrazone formation, the system undergoes an intramolecular cyclization to close the triazole ring. The final and decisive step involves oxidative aromatization driven by tert-butyl hydroperoxide (TBHP). This oxidation ensures the formation of the fully aromatic 1,2,4-triazole system, stabilizing the final product. The general reaction scheme highlights the convergence of these distinct chemical events into a single operational sequence, minimizing waste and maximizing atom economy. This mechanistic understanding allows process chemists to fine-tune parameters such as temperature (70-90°C) and stoichiometry to optimize yields for specific substrates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis requires careful attention to reagent ratios and thermal control to ensure maximum conversion. The protocol is designed to be robust, utilizing commercially available solvents like 1,4-dioxane which effectively dissolve both the organic hydrazide and the carbohydrate starting material. The reaction is typically conducted in a Schlenk tube or standard reactor, reflecting its adaptability to both laboratory and pilot-scale environments. While the general procedure is straightforward, precise control over the oxidant addition and reaction time is essential to prevent over-oxidation or decomposition of the sensitive hydrazide starting material.
- Combine trifluoromethanesulfonic acid, tert-butyl hydroperoxide (70% aq), water, trifluoroacetimidoyl hydrazide, and glucose in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for 2 to 4 hours to allow the cascade cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to a glucose-based synthesis model offers profound strategic advantages beyond mere chemical novelty. The primary driver for adoption is the drastic simplification of the raw material supply chain. Glucose is a commodity chemical produced on a massive global scale for the food and fermentation industries, ensuring a stable, low-cost, and continuous supply that is immune to the volatility often seen in specialized petrochemical intermediates. This stability translates directly into predictable pricing models and reduced risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive, pre-functionalized aldehyde precursors results in substantial raw material cost savings. Furthermore, the removal of the requirement for anhydrous and anaerobic conditions significantly lowers capital expenditure on specialized reactor equipment and reduces energy costs associated with drying solvents and maintaining inert atmospheres. The use of aqueous TBHP and the tolerance of water in the reaction mixture further simplify the solvent recovery and waste treatment processes, leading to a leaner, more cost-efficient manufacturing operation.
- Enhanced Supply Chain Reliability: By relying on biomass-derived starting materials, manufacturers can diversify their supply base away from fossil-fuel-dependent sources. Glucose is readily available from multiple geographic regions and suppliers, mitigating the risk of geopolitical supply disruptions. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental controls, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream API producers.
- Scalability and Environmental Compliance: The process has been demonstrated to be effective at the gram scale with clear potential for kilogram and tonne-scale expansion. The absence of heavy metal catalysts simplifies the regulatory approval process for pharmaceutical applications, as there is no need for extensive and costly heavy metal clearance testing. The use of greener reagents and the generation of benign byproducts align with increasingly stringent environmental regulations, positioning this technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this glucose-mediated triazole synthesis. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature, providing a clear picture of the method's capabilities and limitations for industrial application.
Q: What is the primary carbon source used in this novel triazole synthesis?
A: The process utilizes D-glucose, a widely available and renewable biomass feedstock, which acts as the carbon synthon to generate the necessary aldehyde intermediate in situ.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the key advantages of this method is that it operates under mild conditions without the need for rigorous anhydrous or oxygen-free environments, simplifying operational requirements.
Q: What catalyst and oxidant system is employed?
A: The reaction employs trifluoromethanesulfonic acid (TfOH) as the acid catalyst to activate glucose and tert-butyl hydroperoxide (TBHP) as the oxidant to drive the final aromatization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biomass-derived synthetic routes in modern pharmaceutical manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this glucose-based cyclization can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite pharmaceutical partners and chemical distributors to collaborate with us on optimizing this technology for your specific supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your target molecules. We are prepared to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timelines and reduce overall manufacturing costs.
