Advanced Molybdenum-Copper Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Molybdenum-Copper Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical gaps in current synthetic capabilities. This technology leverages a dual catalytic system involving molybdenum hexacarbonyl and cuprous acetate to facilitate a highly efficient cycloaddition reaction. The significance of this development cannot be overstated, as the 1,2,4-triazole scaffold is a privileged structure found in numerous blockbuster drugs, including antiviral agents and DPP-4 inhibitors like Sitagliptin. By enabling the direct construction of these motifs from readily available trifluoroethylimidoyl chloride and functionalized isonitriles, this patent offers a streamlined pathway that bypasses the limitations of traditional multi-step syntheses.
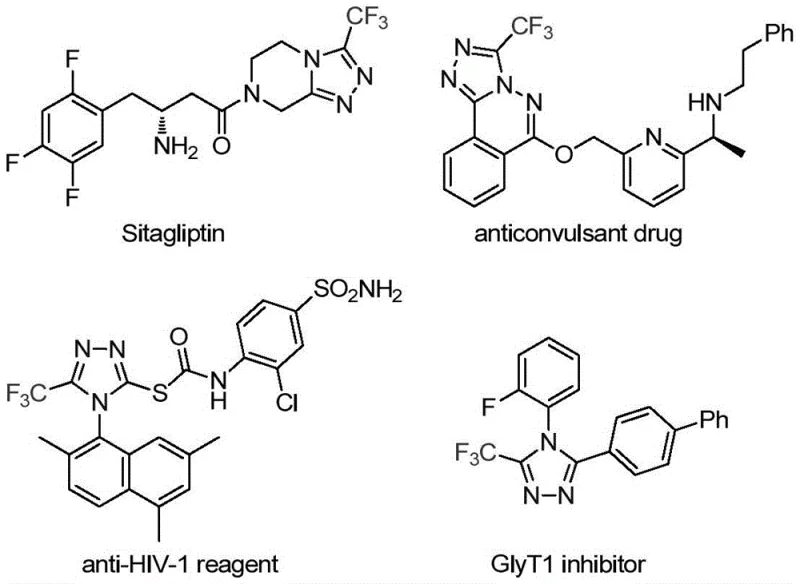
The strategic incorporation of the trifluoromethyl group into the triazole ring significantly alters the physicochemical properties of the parent molecule, improving lipophilicity and electronegativity which are crucial for drug-receptor interactions. For R&D directors and process chemists, the ability to access these diversified scaffolds through a single, operationally simple protocol represents a major advancement in medicinal chemistry toolkits. The method described in the patent not only simplifies the synthetic route but also ensures high reaction efficiency and excellent functional group tolerance, making it an attractive option for the rapid generation of compound libraries for biological screening. Furthermore, the mild reaction conditions and the use of commercially available starting materials position this technology as a viable candidate for immediate adoption in both discovery and process research settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has relied on several conventional strategies that often suffer from significant drawbacks regarding operational complexity and reagent availability. Traditional approaches frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that typically require harsh reaction conditions and generate substantial amounts of chemical waste. Another common route involves copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane; however, the handling of diazo compounds poses serious safety risks due to their potential explosiveness and instability, which complicates scale-up efforts in a manufacturing environment. Additionally, methods relying on the cyclization of trifluoroethylimidoyl chloride with aldehyde hydrazones and hydrazides often result in fully substituted triazoles, limiting the structural diversity available for 3,4-disubstituted variants which are highly sought after in drug design. These legacy methods often struggle with low atom economy, difficult purification protocols, and the necessity for expensive or hazardous reagents, creating bottlenecks in the supply chain for high-purity intermediates.
The Novel Approach
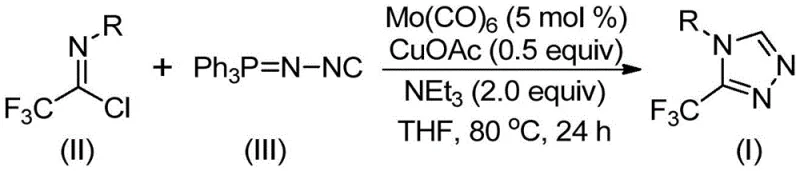
In stark contrast to these cumbersome traditional methods, the novel approach disclosed in patent CN113307778A utilizes a sophisticated yet practical co-catalytic system to achieve the direct synthesis of 3-trifluoromethyl-1,2,4-triazoles. This innovative strategy employs molybdenum hexacarbonyl as a metal activator in conjunction with cuprous acetate to promote a [3+2] cycloaddition between functionalized isonitriles and trifluoroethylimidoyl chloride. The reaction proceeds under remarkably mild thermal conditions, typically between 70°C and 90°C, in common organic solvents like tetrahydrofuran (THF), eliminating the need for extreme temperatures or pressures. A key advantage of this methodology is its exceptional substrate scope, allowing for the introduction of various substituents such as alkyl, alkoxy, halogen, and nitro groups on the aromatic ring without compromising yield. The process is designed to be operationally simple, requiring only standard laboratory equipment and avoiding the use of dangerous diazo reagents, thereby significantly enhancing workplace safety and reducing the regulatory burden associated with hazardous material handling. This shift towards a safer, more efficient catalytic cycle represents a paradigm shift in how these valuable heterocyclic building blocks are manufactured.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic pathway of this transformation is a fascinating interplay between molybdenum and copper species that orchestrates the formation of the triazole ring with high precision. Initially, the molybdenum hexacarbonyl interacts with the functionalized isonitrile to form a reactive metal-isocyanide complex, which serves to activate the isonitrile carbon towards nucleophilic attack. Simultaneously, the cuprous acetate likely facilitates the coordination of the trifluoroethylimidoyl chloride, lowering the energy barrier for the subsequent bond-forming events. The reaction is proposed to proceed through a copper-promoted [3+2] cycloaddition where the activated isonitrile adds across the imidoyl chloride moiety to generate a five-membered ring intermediate. This intermediate then undergoes a crucial elimination step where triphenylphosphine oxide is removed, driven by the presence of water in the system or during workup, to yield the final aromatic 3-trifluoromethyl-substituted 1,2,4-triazole product. Understanding this dual-catalyst mechanism is vital for process optimization, as the precise ratio of Mo to Cu and the choice of base (triethylamine) are critical parameters that influence the rate of complex formation and the overall turnover number of the catalytic cycle.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy pathways that often lead to complex mixtures of byproducts. The concerted nature of the cycloaddition, guided by the metal centers, ensures high regioselectivity, predominantly forming the desired 1,2,4-triazole isomer rather than alternative 1,2,3-triazole or 1,3,4-triazole congeners. The use of molecular sieves in the reaction mixture further aids in maintaining anhydrous conditions during the initial stages, preventing premature hydrolysis of the sensitive imidoyl chloride starting material, which is a common source of yield loss in similar reactions. Post-reaction, the removal of the triphenylphosphine oxide byproduct is straightforward, typically achieved through standard silica gel chromatography, resulting in a product with a clean impurity profile. For quality control teams, this predictability in the reaction outcome translates to easier validation and more consistent batch-to-batch reproducibility, which is essential for meeting the stringent purity specifications required for pharmaceutical intermediates intended for clinical use.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, one must adhere to the specific stoichiometric ratios and environmental controls outlined in the patent data to maximize yield and minimize side reactions. The process begins with the careful weighing of molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), and triethylamine (2.0 equiv), which are added to a reaction vessel containing the trifluoroethylimidoyl chloride and the functionalized isonitrile substrate. It is imperative to use dry, degassed tetrahydrofuran (THF) as the solvent to ensure the stability of the metal catalysts and the reactivity of the isonitrile species. The reaction mixture is then heated to a controlled temperature range of 80°C and maintained for approximately 24 hours, although monitoring via TLC or HPLC is recommended to determine the exact endpoint for different substrates. Detailed standardized synthesis steps follow below to guide the laboratory execution of this protocol.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu-catalyzed methodology presents a compelling value proposition centered around cost efficiency and supply reliability. The primary economic driver is the utilization of inexpensive and commercially abundant starting materials, such as trifluoroethylimidoyl chloride and functionalized isonitriles, which are significantly cheaper than the specialized diazo reagents or complex hydrazines required by older methods. Furthermore, the catalyst system relies on molybdenum and copper salts, which are far more cost-effective than precious metals like palladium or rhodium often used in cross-coupling reactions, leading to a drastic reduction in raw material costs per kilogram of product. The simplified workup procedure, which avoids complex extraction sequences or specialized scavenging resins for heavy metals, further reduces the operational expenditure associated with downstream processing. By streamlining the synthesis into a single pot with high atom economy, manufacturers can achieve substantial cost savings in both material consumption and labor hours, directly impacting the bottom line of API manufacturing budgets.
- Cost Reduction in Manufacturing: The elimination of hazardous diazo compounds and expensive precious metal catalysts fundamentally alters the cost structure of producing these intermediates. Since the reaction utilizes base metals and avoids the need for cryogenic conditions or high-pressure equipment, the capital expenditure (CAPEX) for setting up production lines is significantly lowered. The high reaction efficiency reported in the patent, with yields reaching up to 99% for certain substrates, means that less raw material is wasted, maximizing the output from every batch. Additionally, the reduced generation of toxic waste simplifies disposal protocols, leading to lower environmental compliance costs and fewer fees associated with hazardous waste treatment facilities.
- Enhanced Supply Chain Reliability: Sourcing consistency is a critical concern for supply chain leaders, and this method excels by relying on commodity chemicals that are widely available from multiple global vendors. Unlike proprietary reagents that may be subject to single-source bottlenecks or long lead times, trifluoroethylimidoyl chloride and triethylamine are standard inventory items for most chemical distributors. This abundance ensures that production schedules are not disrupted by raw material shortages, providing a stable foundation for long-term supply agreements. Moreover, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their production sites geographically to mitigate regional risks and ensure continuous availability of these critical pharmaceutical intermediates for their clients.
- Scalability and Environmental Compliance: The patent explicitly demonstrates that the reaction can be successfully expanded to gram-level scales without loss of efficiency, indicating a clear path towards kilogram and ton-scale commercial production. The use of THF as a solvent is advantageous as it is a well-understood industrial solvent with established recovery and recycling protocols, aligning with green chemistry principles. The absence of explosive reagents removes a major safety barrier to scale-up, allowing for larger batch sizes in standard stainless steel reactors without the need for specialized blast-proof infrastructure. This scalability, combined with a cleaner reaction profile, facilitates easier regulatory approval for new drug applications, as the impurity profile is well-defined and manageable, ensuring that the supply chain remains compliant with increasingly strict global environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method, derived directly from the experimental data and disclosures within the document. These answers are designed to provide clarity for technical teams evaluating the feasibility of integrating this route into their existing workflows. By understanding the specific nuances of catalyst loading, substrate compatibility, and purification requirements, stakeholders can make informed decisions about process adoption. The data supports the versatility of the method across a wide range of electronic and steric environments on the aromatic ring.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents on the aromatic ring.
Q: Is this process suitable for large-scale production?
A: The patent explicitly states the method can be expanded to gram-level reactions with high efficiency, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Mo/Cu-catalyzed synthesis of 3-trifluoromethyl-1,2,4-triazoles for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of fluorinated reagents and the precise control of reaction parameters necessary to maintain stringent purity specifications. With our rigorous QC labs and commitment to quality, we guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards, providing you with a reliable foundation for your drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can reduce your overall COGS. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom targets. Let us help you accelerate your timeline to market with a supply chain partner that combines technical expertise with commercial reliability.
