Advanced Asymmetric Catalysis for High-Purity Axial Chiral Aryl Indole Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to access complex chiral scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN110467555B, which discloses a novel synthetic route for axial chiral aryl indole compounds. These molecules, characterized by their unique stereogenic axes formed between indole and naphthalene or benzene rings, represent a privileged structural motif in medicinal chemistry. The patent highlights that such compounds function as potent glucocorticoid receptor antagonists and HCV NS5B polymerase inhibitors, addressing urgent needs in treating inflammatory diseases and viral infections. Furthermore, recent biological evaluations included in the intellectual property reveal that specific derivatives exhibit remarkable cytotoxicity against MCF-7 breast cancer cell lines, with leading candidates demonstrating IC50 values as low as 4.44 μg/mL. This dual potential—serving as both a versatile scaffold for catalyst design and a bioactive pharmacophore—positions this technology as a high-value asset for any organization focused on reliable pharmaceutical intermediate supplier capabilities.
Historically, the construction of axially chiral indole-naphthalene and indole-benzene skeletons has been fraught with synthetic challenges. Conventional approaches often rely on direct coupling reactions between indole rings and naphthol or phenol derivatives, which frequently suffer from poor stereocontrol and require harsh reaction conditions. For instance, prior art described in literature such as Angew. Chem. Int. Ed. 2017 utilizes chiral phosphoric acids to couple 2-indolecarbinols with naphthols, but these methods can be limited by substrate scope and the stability of the carbinol precursors. Another existing strategy involves the coupling of 2-substituted indoles with azonaphthalenes, yet these routes often lack the generality required for diverse library synthesis. The limitations of these traditional methods include the necessity for cryogenic temperatures, the use of sensitive organometallic reagents, and difficulties in achieving high enantiomeric excess (ee) across a broad range of electronic variations on the aromatic rings. Consequently, there has been a persistent gap in the availability of efficient, scalable protocols for generating these high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy synthesis pathways for axial chiral biaryl systems typically involve multi-step sequences that degrade overall process efficiency. Traditional cross-coupling reactions often necessitate the use of expensive palladium or rhodium catalysts, which introduce significant cost burdens and complicate downstream purification due to stringent regulatory limits on residual heavy metals in active pharmaceutical ingredients (APIs). Moreover, the dynamic kinetic resolution of racemic precursors—a powerful strategy for converting 50% theoretical yield limits into near-quantitative outcomes—has remained largely unexplored for this specific class of indole-aryl compounds until the advent of the technology described in CN110467555B. Previous attempts to functionalize the indole C3 position with bulky aryl groups often resulted in low diastereoselectivity or required protecting group manipulations that added unnecessary steps to the manufacturing timeline. The inability to perform these transformations under ambient conditions also poses safety risks and energy consumption issues when attempting commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology presented in the patent revolutionizes this landscape by employing a direct, one-pot asymmetric addition reaction catalyzed by small organic molecules. Specifically, the process utilizes racemic indole-naphthalene or indole-benzene precursors (Formula 1 or Formula 4) reacting with azodicarboxylates (Formula 2) in the presence of a chiral phosphoric acid catalyst. This approach effectively achieves a dynamic kinetic resolution, transforming the racemic starting material into a single enantiomer of the target hydrazine derivative with exceptional efficiency. The reaction proceeds smoothly in dichloromethane at mild temperatures ranging from 20°C to 30°C, eliminating the need for energy-intensive heating or cooling cycles. By leveraging the acidity and chiral environment of the phosphoric acid, the system activates the electrophilic azo compound while simultaneously directing the nucleophilic attack of the indole species. This results in products (Formula 3 or Formula 5) with outstanding optical purity, where enantiomeric ratios (er) frequently exceed 96:4 and can reach as high as 98:2 depending on the substrate substituents.
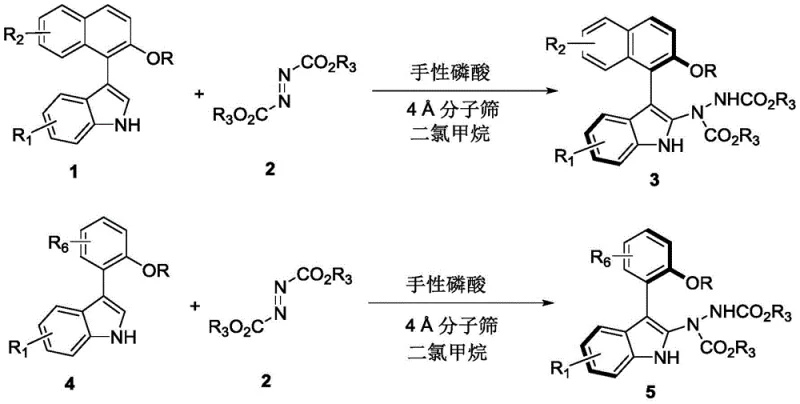
This novel pathway not only simplifies the synthetic operation but also dramatically expands the accessible chemical space. The tolerance for various functional groups—including halogens, esters, ethers, and sulfonates on both the indole and the aryl moieties—allows medicinal chemists to rapidly generate diverse analog libraries for structure-activity relationship (SAR) studies. The use of azodicarboxylates as coupling partners introduces a hydrazine linkage that can be further manipulated or retained as a key pharmacophore, adding another layer of utility to the final products. The operational simplicity, combined with the high atom economy of the addition reaction, makes this method a superior alternative to classical cross-coupling strategies for constructing these sterically hindered chiral axes.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Dynamic Kinetic Resolution
The success of this synthesis hinges on the precise molecular recognition and activation provided by the chiral phosphoric acid catalyst, specifically the binaphthyl-derived Formula 61. In this catalytic cycle, the phosphoric acid acts as a bifunctional activator. The acidic proton of the P-OH group forms a strong hydrogen bond with the nitrogen atoms of the azodicarboxylate electrophile, thereby lowering its LUMO energy and enhancing its reactivity towards nucleophilic attack. Simultaneously, the phosphoryl oxygen (P=O) engages in hydrogen bonding with the N-H proton of the indole substrate, organizing the transition state within a well-defined chiral pocket created by the bulky 3,3'-substituents on the binaphthyl backbone. These substituents, typically 2,4,6-triisopropylphenyl groups, create significant steric hindrance that forces the incoming reactants to approach from a specific trajectory, thus dictating the absolute configuration of the newly formed axis.
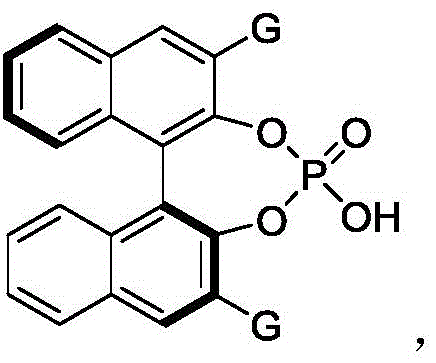
Impurity control is inherently managed through this highly organized transition state. Because the catalyst discriminates so effectively between the pro-chiral faces of the reacting species, the formation of the minor enantiomer is kinetically suppressed. The patent data indicates that even with electronically diverse substrates, such as those bearing electron-withdrawing trifluoromethyl groups or electron-donating methoxy groups, the enantioselectivity remains consistently high. For example, substrates with 5-methoxy or 6-fluoro substitutions on the naphthalene ring still achieve er values above 95:5. This robustness suggests that the steric bulk of the catalyst dominates the stereochemical outcome, rendering the process less sensitive to subtle electronic perturbations in the substrate. Furthermore, the use of 4 Å molecular sieves in the reaction mixture plays a critical role in maintaining catalyst activity by sequestering trace water, which could otherwise hydrolyze the azodicarboxylate or deactivate the phosphoric acid through competitive hydrogen bonding. This attention to moisture control ensures reproducible high yields and optical purity, which is essential for meeting the rigorous quality standards of high-purity pharmaceutical intermediates.
How to Synthesize Axial Chiral Aryl Indole Efficiently
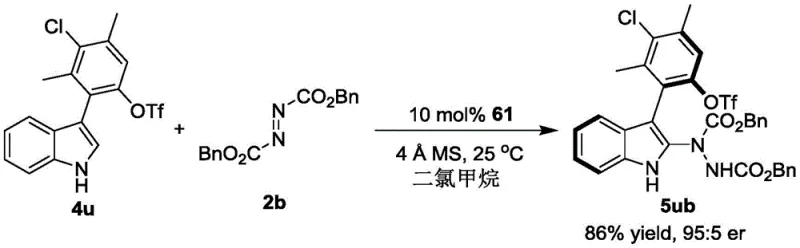
The implementation of this synthesis protocol is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of a dry reaction vessel charged with the racemic indole-aryl precursor and the azodicarboxylate coupling partner in anhydrous dichloromethane. The addition of activated 4 Å molecular sieves is a critical preparatory step to ensure the anhydrous conditions required for optimal catalyst performance. Once the mixture is equilibrated, the chiral phosphoric acid catalyst (Formula 61) is introduced, typically at a loading of 10 mol%, although optimization may allow for lower loadings in scaled processes. The reaction is then allowed to stir at ambient temperature (25°C is preferred), monitoring progress via thin-layer chromatography (TLC) until the starting materials are fully consumed. Upon completion, the solid molecular sieves are removed by filtration, and the filtrate is concentrated under reduced pressure. The crude product is subsequently purified using silica gel column chromatography, utilizing a petroleum ether and ethyl acetate gradient, to afford the target axial chiral compound as a high-purity solid. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Prepare the reaction mixture by combining the racemic indole-naphthalene or indole-benzene precursor with an azodicarboxylate derivative in dichloromethane solvent.
- Add 4 Å molecular sieves as a drying agent and introduce the chiral phosphoric acid catalyst (specifically Formula 61 derivatives) to the reaction vessel.
- Stir the mixture at ambient temperature (20-30°C) until TLC indicates completion, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic methodology offers transformative benefits over traditional metal-catalyzed routes. The most immediate impact is observed in the cost structure of the manufacturing process. By eliminating the requirement for precious transition metals such as palladium, rhodium, or iridium, the raw material costs are significantly reduced. More importantly, the absence of heavy metals removes the need for expensive and time-consuming scavenging steps to meet regulatory limits for residual metals in drug substances. This simplification of the downstream processing workflow translates directly into substantial cost savings and shorter production cycles. Additionally, the catalyst itself, being an organic small molecule, is generally more stable and easier to handle than air-sensitive metal complexes, reducing storage and handling costs associated with specialized inert atmosphere equipment.
- Cost Reduction in Manufacturing: The economic advantages extend beyond just catalyst costs. The reaction operates at ambient temperatures (20-30°C), which drastically reduces energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux. The use of dichloromethane, a common and recyclable solvent, further simplifies solvent recovery operations. Since the reaction achieves high yields (often exceeding 90%) and high selectivity in a single step, the overall material throughput is maximized, minimizing waste generation and the associated disposal costs. The atom economy of this addition reaction is inherently superior to substitution reactions that generate stoichiometric salt byproducts, aligning with green chemistry principles that are increasingly mandated by global regulatory bodies.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted indoles, naphthols, and azodicarboxylates, are commercially available from multiple global suppliers, ensuring a robust and resilient supply chain. Unlike specialized chiral ligands for metal catalysis which may have long lead times and single-source dependencies, the chiral phosphoric acid catalysts can be synthesized from readily available binaphthol precursors. This diversity in sourcing options mitigates the risk of supply disruptions. Furthermore, the mild reaction conditions reduce the risk of thermal runaways or hazardous exotherms, enhancing operational safety and ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to tonnage levels is facilitated by the simplicity of the reaction setup. The absence of gaseous reagents or high-pressure requirements allows for the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The environmental footprint is minimized due to the high selectivity of the reaction, which reduces the formation of difficult-to-separate impurities and lowers the volume of solvent required for purification. The process generates minimal hazardous waste, primarily consisting of spent molecular sieves and silica gel, which are easier to manage than heavy metal-contaminated waste streams. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for companies aiming to reduce their carbon footprint and improve their sustainability metrics in cost reduction in pharmaceutical intermediate manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphoric acid catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for project managers and technical leads evaluating this route for inclusion in their development pipelines.
Q: What is the primary advantage of using chiral phosphoric acid for this synthesis?
A: The use of chiral phosphoric acid enables a highly enantioselective dynamic kinetic resolution under mild conditions, achieving er values up to 98:2 without the need for toxic transition metals.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the process operates at ambient temperatures (25°C) using common solvents like dichloromethane and avoids hazardous reagents, making it highly suitable for commercial scale-up.
Q: What biological activities do these axial chiral compounds exhibit?
A: These compounds have demonstrated significant cytotoxic activity against MCF-7 breast cancer cells, with specific derivatives showing IC50 values as low as 4.44 μg/mL, indicating strong potential for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Supplier
The technological advancements described in CN110467555B represent a significant leap forward in the synthesis of complex chiral scaffolds, yet translating patent chemistry into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this translation, offering comprehensive CDMO services tailored to the unique challenges of asymmetric organocatalysis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields observed in the laboratory are faithfully reproduced at the manufacturing scale. We understand that the consistency of chiral phosphoric acid catalysts and the strict control of moisture levels are critical success factors, and our rigorous QC labs are equipped to monitor these parameters with precision. Our commitment to stringent purity specifications ensures that every batch of axial chiral aryl indole intermediate meets the exacting standards required for clinical and commercial API synthesis.
We invite pharmaceutical and agrochemical companies to leverage our technical capabilities to accelerate their drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic feasibility of this route against your current supply chain. We encourage you to contact our technical procurement team to request specific COA data for our catalog of chiral intermediates and to discuss route feasibility assessments for your proprietary targets. Whether you require kilogram quantities for preclinical studies or metric tons for commercial launch, NINGBO INNO PHARMCHEM is prepared to be your strategic partner in delivering high-quality, cost-effective chiral solutions.
