Advanced Catalytic Synthesis of Axially Chiral Aryl Indole Intermediates for Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel scaffolds with defined stereochemistry to enhance drug efficacy and reduce off-target effects. Patent CN110467555B introduces a groundbreaking methodology for the synthesis of axially chiral aryl indole compounds, specifically targeting the indole-naphthalene and indole-benzene structural motifs. These privileged structures are increasingly recognized for their potent biological activities, including roles as glucocorticoid receptor antagonists and HCV NS5B polymerase inhibitors. The disclosed technology addresses a critical gap in organic synthesis by providing a robust, organocatalytic route to access these complex chiral architectures with exceptional optical purity. Unlike traditional methods that often struggle with racemic mixtures or require harsh conditions, this invention leverages dynamic kinetic resolution strategies to deliver high-value intermediates essential for modern drug discovery pipelines.
Historically, the construction of axially chiral indole-naphthalene and indole-benzene skeletons has been fraught with challenges, limiting their widespread adoption in medicinal chemistry. Conventional approaches typically rely on coupling reactions between indole rings and naphthol or phenol derivatives, which frequently suffer from limited substrate scope and poor stereocontrol. Furthermore, existing literature methods often necessitate the use of expensive transition metal catalysts or extreme reaction conditions that are incompatible with sensitive functional groups. The lack of efficient dynamic kinetic resolution protocols for these specific scaffolds has left a significant void in the synthetic toolbox available to process chemists. Consequently, the development of a mild, metal-free alternative that can tolerate diverse substitution patterns has been a long-standing objective for researchers aiming to optimize lead compounds.
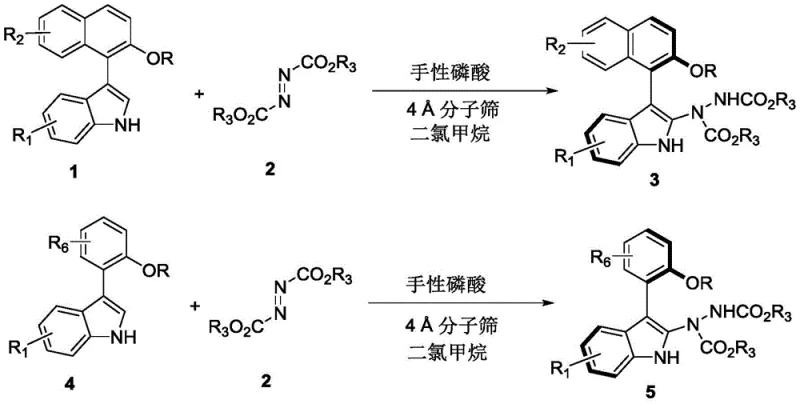
The novel approach detailed in the patent fundamentally shifts the paradigm by utilizing a chiral phosphoric acid catalyst to mediate the asymmetric addition reaction. This method transforms readily available racemic precursors into highly enriched axially chiral products in a single operational step. By employing dichloromethane as a solvent and incorporating 4Å molecular sieves, the reaction proceeds efficiently at ambient temperature (25°C), eliminating the need for energy-intensive heating or cooling cycles. The versatility of this protocol is demonstrated by its compatibility with a wide range of substituents on both the indole and naphthalene/phenyl rings, allowing for the rapid generation of diverse compound libraries. This represents a significant advancement over prior art, offering a streamlined pathway that combines high atom economy with superior enantioselectivity, making it an attractive option for both early-stage research and process development.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
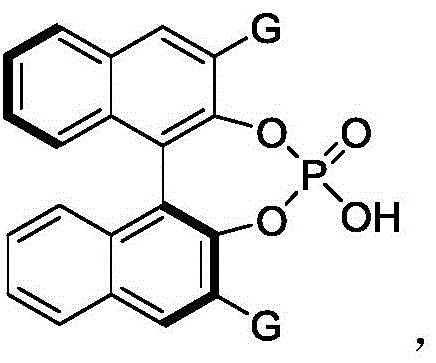
The core of this synthetic breakthrough lies in the precise activation mode of the chiral phosphoric acid catalyst, specifically the BINOL-derived derivative designated as Formula 61. This catalyst functions through a dual hydrogen-bonding network that simultaneously activates the electrophilic azodicarboxylate reagent and organizes the nucleophilic indole substrate within a well-defined chiral pocket. The bulky 2,4,6-triisopropylphenyl groups on the catalyst backbone create a sterically confined environment that dictates the trajectory of the incoming nucleophile, thereby ensuring high facial selectivity during the bond-forming event. This supramolecular recognition is critical for establishing the axial chirality, as it effectively differentiates between the enantiotopic faces of the reacting species. The result is a highly stereoselective transformation that consistently delivers products with enantiomeric ratios exceeding 96:4 across various substrates.
Impurity control is inherently managed by the specificity of the organocatalytic cycle, which minimizes side reactions common in transition metal catalysis, such as homocoupling or over-oxidation. The use of molecular sieves plays a pivotal role in maintaining the integrity of the catalytic cycle by sequestering trace water that could otherwise hydrolyze the sensitive azodicarboxylate reagent or deactivate the acidic catalyst. This careful management of the reaction environment ensures that the primary pathway dominates, leading to clean reaction profiles that simplify downstream purification. The robustness of the catalyst allows it to maintain high turnover frequencies even in the presence of potentially coordinating functional groups like halogens or esters. Such mechanistic resilience is essential for pharmaceutical manufacturing, where consistent product quality and minimal impurity profiles are non-negotiable requirements for regulatory approval.
How to Synthesize Axially Chiral Aryl Indole Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard laboratory equipment and commercially available reagents. The protocol begins with the preparation of the reaction mixture in anhydrous dichloromethane, ensuring that moisture-sensitive components remain stable throughout the process. Operators simply combine the indole substrate and the azo reagent in the specified molar ratio, typically ranging from 1:1 to 1:3, depending on the specific electronic nature of the substrates. The addition of the chiral catalyst at a loading of 10 mol% initiates the transformation, which is then allowed to proceed under stirring at room temperature. Detailed standardized synthesis steps for optimizing yield and enantioselectivity are provided in the guide below.
- Prepare the reaction mixture by combining the indole substrate (Formula 1 or 4) and the azodicarboxylate reagent (Formula 2) in dichloromethane solvent.
- Add 4Å molecular sieves as an additive to remove trace water and introduce the chiral phosphoric acid catalyst (Formula 61) at a loading of 10 mol%.
- Stir the reaction at ambient temperature (25°C) until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic methodology offers substantial strategic benefits by simplifying the sourcing of raw materials and reducing processing complexity. The reliance on organocatalysis eliminates the need for precious metal catalysts, which are subject to volatile market pricing and stringent regulatory limits regarding residual metals in final drug substances. By removing transition metals from the process entirely, manufacturers can bypass costly and time-consuming metal scavenging steps, leading to a more streamlined production workflow. This reduction in unit operations directly translates to lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry initiatives that are increasingly prioritized by global pharmaceutical buyers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification infrastructure significantly lowers the cost of goods sold for these intermediates. Furthermore, the reaction operates at ambient temperature, removing the energy costs associated with cryogenic cooling or high-temperature heating required by competing technologies. The high yields reported, often exceeding 90%, maximize the utilization of starting materials, reducing waste disposal costs and improving overall material efficiency. This economic efficiency makes the process highly competitive for large-scale production of active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: The starting materials, including substituted indoles and azodicarboxylates, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply lines to downstream drug manufacturers. Additionally, the simplicity of the workup procedure, involving basic filtration and concentration, reduces the turnaround time for production campaigns, allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The use of dichloromethane, while requiring proper handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. The absence of heavy metal waste simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. The mild reaction conditions facilitate safe scale-up from gram to kilogram scales without the need for specialized high-pressure or low-temperature reactors. This inherent scalability ensures that the supply of these critical intermediates can grow in tandem with the clinical development of the final drug candidate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axially chiral compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and reliability for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing development pipelines.
Q: What is the primary advantage of using chiral phosphoric acid catalysts for this synthesis?
A: The primary advantage is the ability to achieve high enantioselectivity (up to 98:2 er) under mild reaction conditions (25°C) without the need for toxic transition metals, simplifying downstream purification for pharmaceutical applications.
Q: Can this synthetic method be scaled for industrial production?
A: Yes, the method utilizes readily available starting materials, operates at ambient temperature, and employs simple workup procedures like filtration and column chromatography, making it highly suitable for commercial scale-up.
Q: What biological activity do these axially chiral compounds exhibit?
A: Biological testing indicates that these compounds possess cytotoxic activity against MCF-7 breast cancer cells, with specific derivatives showing IC50 values as low as 4.44 μg/mL, highlighting their potential as oncology drug leads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the chiral phosphoric acid catalysis described in CN110467555B to meet your specific volume requirements while adhering to stringent purity specifications. We operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to ensure that every batch of axially chiral intermediate meets the highest standards of optical purity and chemical identity required for clinical trials and commercial launch.
We invite you to contact our technical procurement team to discuss how we can support your project with a Customized Cost-Saving Analysis tailored to your specific target molecule. By leveraging our expertise in organocatalysis and process optimization, we can help you secure a reliable supply of high-purity pharmaceutical intermediates. Please reach out to request specific COA data and route feasibility assessments to accelerate your drug development timeline.
