Advanced Catalytic Synthesis of Ambroxol HCl for Scalable Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of Ambroxol HCl for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for high-volume mucolytic agents like Ambroxol Hydrochloride. Patent CN103073439B introduces a transformative synthesis method that addresses long-standing challenges in impurity control and process safety. This technology leverages a unique diacetyl protection strategy combined with a novel dual-catalyst system to achieve yields exceeding 67% and purity levels greater than 99.9%. For R&D directors and supply chain managers, this represents a significant opportunity to optimize the manufacturing of this critical respiratory therapeutic. The following analysis details the mechanistic advantages and commercial viability of this approach.
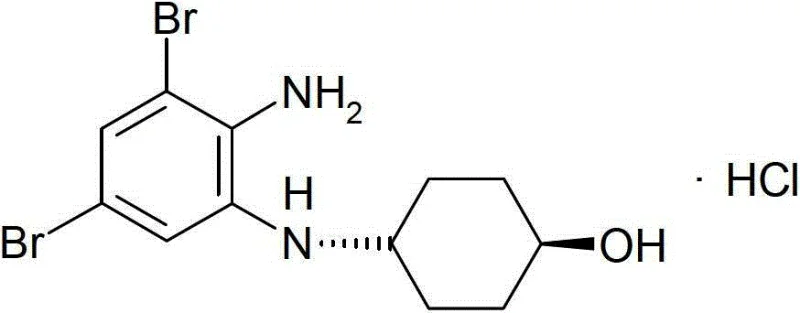
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ambroxol HCl has been plagued by low yields and complex impurity profiles, primarily due to the high reactivity of the amino group on the benzene ring. Traditional Method 1, which starts from 2-amino-3,5-dibromobenzyl bromide, suffers from uncontrolled condensation side reactions because the amino group is either unprotected or insufficiently protected. This lack of selectivity leads to the formation of polymeric by-products and quinazoline derivatives, capping overall yields at approximately 10% and purity around 92%. Furthermore, Method 2, which relies on sodium borohydride reduction of imine intermediates, introduces severe safety hazards and environmental burdens. The use of strong reducing agents necessitates rigorous safety protocols to manage hydrogen evolution, while the subsequent removal of heavy metal catalysts adds costly purification steps that complicate the supply chain.
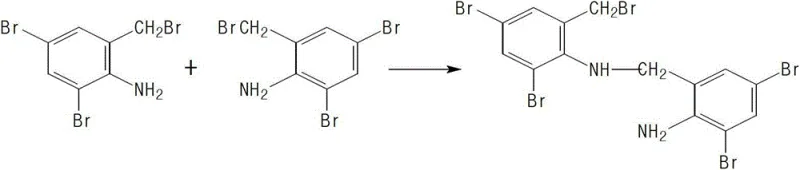
The Novel Approach
The patented methodology fundamentally reengineers the synthetic pathway by introducing a fully protected starting material, 2-diacetylamino-3,5-dibromobenzyl bromide. This strategic modification sterically and electronically deactivates the ortho-amino position, effectively shutting down the primary pathway for impurity generation. By coupling this protected electrophile with trans-4-aminocyclohexanol under mild basic conditions, the reaction proceeds with exceptional selectivity. The subsequent hydrolysis step cleanly removes the acetyl groups to reveal the active pharmaceutical ingredient. This approach not only simplifies the workflow by eliminating the need for hazardous reduction steps but also drastically improves the mass balance of the process, transforming a low-yielding, hazardous operation into a streamlined, high-efficiency manufacturing sequence.
Mechanistic Insights into Diacetyl-Protection and Dual-Catalysis
The core innovation of this synthesis lies in the synergistic effect of the diacetyl protecting group and the specific catalyst system comprising DMAP and 4-dimethylaminocyclohexanol. In the first step, the diacetyl group acts as a robust shield for the nitrogen atom, preventing it from acting as a nucleophile in unwanted intramolecular cyclizations that typically form Impurity B (the quinazoline derivative). This ensures that the nucleophilic attack comes exclusively from the cyclohexyl amine, driving the reaction toward the desired benzylamine intermediate. The presence of the acetyl groups also enhances the solubility of the starting material in organic solvents like ethanol, facilitating a homogeneous reaction phase that maximizes molecular collisions and reaction kinetics.
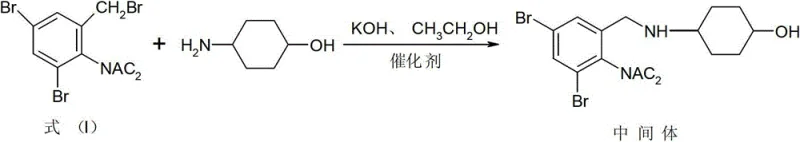
Furthermore, the catalytic system plays a pivotal role in lowering the activation energy of the nucleophilic substitution. DMAP acts as a potent nucleophilic catalyst, likely forming a transient reactive intermediate that accelerates the displacement of the bromide leaving group. Simultaneously, 4-dimethylaminocyclohexanol functions as a phase-transfer-like promoter, stabilizing the transition state and ensuring rapid conversion even at moderate temperatures of 60°C to 75°C. This dual-catalyst mechanism allows the reaction to reach near-quantitative conversion without the need for excessive reagents or harsh thermal conditions. The result is an intermediate that is sufficiently pure to proceed directly to the next step without recrystallization, a rare feat in fine chemical synthesis that significantly reduces solvent consumption and processing time.
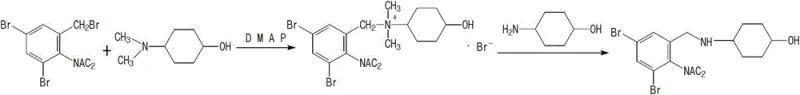
How to Synthesize Ambroxol Hydrochloride Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the protective group strategy. The process begins with the preparation of a homogeneous solution of trans-4-aminocyclohexanol in absolute ethanol, followed by the sequential addition of potassium hydroxide and the catalyst system. The protected benzyl bromide is then introduced in batches to manage exotherms, maintaining the temperature between 60°C and 75°C to ensure complete conversion while minimizing degradation. Following the condensation, the solvent is partially removed, and the intermediate is crystallized by cooling. The final step involves a straightforward acid hydrolysis in an ethanol-water-acetic acid mixture, which cleaves the acetyl groups and forms the hydrochloride salt simultaneously.
- React 2-diacetylamino-3,5-dibromobenzyl bromide with trans-4-aminocyclohexanol using a DMAP and 4-dimethylaminocyclohexanol catalyst system in ethanol.
- Isolate the intermediate via cooling crystallization without the need for complex purification steps.
- Perform acid hydrolysis and salt formation using hydrochloric acid, acetic acid, and water to yield high-purity Ambroxol HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this patented synthesis model offers profound strategic advantages beyond simple yield improvements. The elimination of sodium borohydride and high-pressure hydrogenation equipment removes significant capital expenditure requirements and reduces the dependency on specialized hazardous material handling infrastructure. This simplification of the process train directly translates to lower operational overheads and reduced insurance costs associated with chemical manufacturing. Additionally, the ability to skip intermediate purification steps means a drastic reduction in solvent usage and waste generation, aligning production with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the substantial increase in overall yield and the reduction in raw material intensity. By preventing the formation of side products, the process minimizes the loss of expensive brominated starting materials, which are often the cost drivers in halogenated API synthesis. Furthermore, the removal of the recrystallization step for the intermediate saves both time and solvent costs, creating a leaner manufacturing profile that significantly lowers the cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of stable, storable intermediates and the avoidance of reagents with volatile supply chains, such as specialized hydrogenation catalysts. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, reducing the risk of batch failures that can disrupt downstream formulation timelines. This reliability is critical for maintaining consistent inventory levels of high-demand respiratory medications in a competitive global market.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations like stirred tank reactors and crystallizers, avoiding the bottlenecks often associated with batch hydrogenation. From an environmental perspective, the significant reduction in solvent volume and the absence of heavy metal contaminants simplify wastewater treatment protocols. This ease of compliance facilitates faster regulatory approvals and reduces the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the technology's capabilities.
Q: How does the new synthesis method improve impurity profiles compared to traditional routes?
A: By utilizing a diacetyl protecting group on the starting material, the new method effectively blocks secondary reactions at the ortho-position of the benzene ring. This strategic protection prevents the formation of complex by-products like Impurity B and E, resulting in a product purity exceeding 99.9% with only trace amounts of a single known impurity.
Q: What are the safety advantages of this catalytic system over sodium borohydride reduction?
A: Traditional routes often rely on sodium borohydride reduction, which poses significant safety hazards due to hydrogen gas evolution and requires strict handling protocols. The patented condensation-hydrolysis route eliminates the need for hazardous reducing agents and high-pressure hydrogenation, significantly lowering operational risks and simplifying waste treatment.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It operates under moderate temperatures (60-75°C) and utilizes common solvents like ethanol and methanol. The elimination of difficult purification steps and the use of stable intermediates make it highly amenable to multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ambroxol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic methodologies is key to maintaining competitiveness in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like this patented route are successfully translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Ambroxol HCl meets the highest international pharmacopoeia standards, providing our partners with unwavering confidence in their supply chain.
We invite you to collaborate with us to leverage these technological advancements for your specific production needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
