Advanced Fluorination Technology for Scalable Florfenicol Intermediate Manufacturing
Advanced Fluorination Technology for Scalable Florfenicol Intermediate Manufacturing
The pharmaceutical and veterinary industries are constantly seeking robust synthetic pathways that balance high purity with economic viability, a challenge explicitly addressed in the recent disclosure of patent CN111423391A. This intellectual property introduces a groundbreaking preparation method for a critical florfenicol intermediate, specifically focusing on the fluorination of a cyclic oxazoline compound to install the essential fluoromethyl group. Unlike traditional methodologies that rely on the expensive and complex Ishikawa reagent derived from hexafluoropropylene, this novel approach leverages a fluorinating agent synthesized from tetrafluoroethylene and dimethylamine. For R&D directors and procurement strategists, this shift represents a pivotal opportunity to optimize the supply chain for one of the world's most important broad-spectrum antibacterial drugs, offering a route that is not only chemically elegant but also commercially superior in terms of raw material accessibility and process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of florfenicol intermediates has been bottlenecked by the reliance on the Ishikawa reagent, a fluorinating agent that necessitates the use of hexafluoropropylene as a key starting material. Hexafluoropropylene is a high-cost specialty gas that introduces significant volatility into the supply chain and inflates the overall production costs of the final API. Furthermore, the preparation of the Ishikawa reagent itself is a cumbersome multi-step process that often requires stringent low-temperature controls and extended reaction times, such as stirring for 18 hours at room temperature after initial mixing. Subsequent fluorination steps in these legacy processes frequently demand harsh thermal conditions, often reaching temperatures around 100°C, which can exacerbate impurity formation and pose safety challenges in large-scale manufacturing environments. These factors collectively create a high barrier to entry for cost-effective production, limiting the ability of manufacturers to respond agilely to market demand fluctuations.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111423391A circumvents these economic and operational hurdles by employing a fluorinating reagent generated from the readily available and inexpensive tetrafluoroethylene gas. This reagent is prepared in situ by introducing tetrafluoroethylene into a solution of dimethylamine in dichloromethane at a mild temperature of -5°C, a process that is significantly simpler and faster than the Ishikawa counterpart. The subsequent fluorination of the cyclic oxazoline substrate proceeds efficiently at moderate temperatures ranging from 75°C to 85°C, eliminating the need for the extreme thermal stress associated with older technologies. This streamlined approach not only simplifies the operational workflow but also inherently reduces the cost of goods sold by substituting a premium raw material with a commodity chemical, thereby enhancing the overall competitiveness of the manufacturing process.
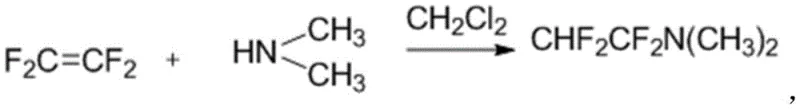
Mechanistic Insights into Tetrafluoroethylene-Mediated Fluorination
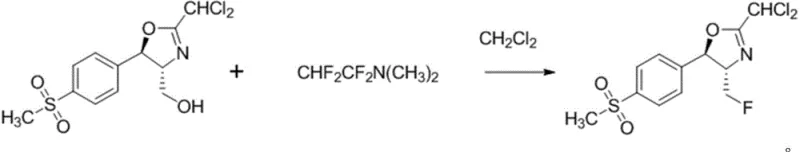
The core chemical innovation of this patent lies in the generation and utilization of a difluoroamino-species derived from tetrafluoroethylene, which acts as a potent nucleophilic fluorinating agent. Mechanistically, the reaction involves the activation of the hydroxymethyl group on the (4R,5R)-2-(1,1-dichloromethyl)-4-hydroxymethyl-5-(4-methylsulfonylphenyl)-4,5-oxazoline ring system. The fluorinating reagent, likely forming a reactive intermediate such as CHF2CF2N(CH3)2, facilitates the displacement of the hydroxyl functionality with a fluorine atom while maintaining the integrity of the sensitive oxazoline ring. This transformation is critical because the introduction of the fluorine atom significantly enhances the lipophilicity and metabolic stability of the final antibiotic, yet it must be achieved without epimerizing the adjacent chiral centers at the C4 and C5 positions. The patent data indicates that this specific reagent system achieves this delicate balance, delivering the desired fluoro-product with high stereochemical fidelity.
Furthermore, the purification strategy described in the patent is meticulously designed to manage the impurity profile, which is a paramount concern for regulatory compliance in veterinary drug manufacturing. Following the fluorination reaction, the crude mixture undergoes a rigorous workup procedure involving aqueous washing to remove amine salts and residual reagents, followed by solvent recovery. The final purification step utilizes methanol recrystallization in the presence of activated carbon, which effectively adsorbs colored impurities and trace organic by-products. This combination of selective reaction chemistry and robust downstream processing ensures that the resulting florfenicol intermediate meets stringent purity specifications, often exceeding 97% content as demonstrated in the experimental examples, thus providing a reliable feedstock for the final hydrolysis and acylation steps required to produce florfenicol.
How to Synthesize Florfenicol Intermediate Efficiently
The synthesis protocol detailed in the patent offers a clear roadmap for implementing this technology at an industrial scale, beginning with the precise preparation of the fluorinating agent under controlled low-temperature conditions to ensure reagent stability. Operators must carefully manage the addition of tetrafluoroethylene gas into the dimethylamine solution to prevent exothermic runaway, followed by the immediate transfer of this reagent to the reactor containing the cyclic substrate. The reaction is then driven to completion by heating the mixture to the optimal range of 85°C for approximately 2 hours, after which the product is isolated through a sequence of phase separations and crystallizations. For a comprehensive understanding of the specific stoichiometric ratios, solvent volumes, and safety parameters required for successful execution, please refer to the standardized synthesis guidelines provided below.
- Preparation of the fluorinating reagent by introducing tetrafluoroethylene gas into a cooled solution of dimethylamine in dichloromethane at -5°C.
- Execution of the fluorination reaction by heating the cyclic oxazoline compound with the prepared reagent in dichloromethane at 75-85°C for 1-4 hours.
- Purification of the crude product through aqueous washing, solvent recovery, and recrystallization from methanol with activated carbon decolorization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this tetrafluoroethylene-based fluorination route offers profound advantages that extend far beyond simple yield improvements. By decoupling the synthesis from the hexafluoropropylene supply chain, manufacturers can insulate themselves from the price volatility and availability constraints often associated with specialized fluorinated gases. This shift to commodity-grade raw materials translates directly into a more predictable and stable cost structure, allowing for better long-term financial planning and pricing competitiveness in the global veterinary pharmaceutical market. Additionally, the simplified reagent preparation eliminates an entire class of expensive precursors, effectively reducing the bill of materials and freeing up capital for other areas of process optimization or capacity expansion.
- Cost Reduction in Manufacturing: The elimination of the Ishikawa reagent synthesis step removes a significant cost center from the production ledger, as the requisite hexafluoropropylene is replaced by the far more economical tetrafluoroethylene. This substitution avoids the complex and energy-intensive preparation of the traditional fluorinating agent, leading to substantial savings in both raw material procurement and utility consumption. Furthermore, the milder reaction conditions reduce the thermal load on the manufacturing equipment, potentially extending vessel lifespan and lowering maintenance costs associated with high-temperature operations. These cumulative efficiencies result in a leaner manufacturing process that maximizes margin potential without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Utilizing tetrafluoroethylene, a widely produced industrial chemical, significantly mitigates the risk of supply disruptions that can occur with niche reagents like hexafluoropropylene. This broader supplier base ensures a continuous flow of materials, which is critical for maintaining uninterrupted production schedules and meeting tight delivery deadlines for downstream API manufacturers. The robustness of the supply chain is further reinforced by the simplicity of the reagent generation process, which can be performed on-site or sourced from multiple vendors, thereby reducing dependency on single-source suppliers and enhancing overall operational resilience against market shocks.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as gas-liquid mixing and crystallization, which are well-understood and easily replicated in large-scale reactors. The use of dichloromethane as a solvent, while requiring careful management, allows for efficient recovery and recycling, minimizing waste generation and aligning with modern green chemistry principles. Moreover, the avoidance of heavy metal catalysts or exotic reagents simplifies the waste treatment profile, reducing the environmental footprint of the facility and easing the regulatory burden associated with hazardous waste disposal. This alignment with sustainability goals makes the technology attractive for companies aiming to improve their environmental, social, and governance (ESG) ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluorination technology, drawing directly from the experimental data and process descriptions found in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this route for the production of high-purity florfenicol intermediates. By understanding the nuances of the reagent system and the purification workflow, stakeholders can make informed decisions about integrating this methodology into their existing manufacturing portfolios.
Q: What is the primary cost advantage of this new fluorination method compared to the Ishikawa reagent route?
A: The primary advantage lies in the raw material substitution; this method utilizes tetrafluoroethylene and dimethylamine, which are significantly more abundant and cost-effective than the hexafluoropropylene required for synthesizing the traditional Ishikawa reagent, thereby drastically reducing the overall bill of materials.
Q: Does this process maintain the critical stereochemistry required for florfenicol activity?
A: Yes, the process is designed to preserve the (4R,5R) stereochemical configuration of the cyclic oxazoline precursor, ensuring that the resulting fluorinated intermediate retains the necessary chiral integrity for downstream conversion into biologically active florfenicol.
Q: How does the reaction condition profile of this patent compare to prior art methods?
A: This patent describes milder reaction conditions, operating effectively at temperatures between 75°C and 85°C, whereas conventional methods often require higher thermal inputs around 100°C, leading to improved energy efficiency and reduced safety risks associated with high-pressure high-temperature operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN111423391A and possess the technical expertise to translate this laboratory-scale innovation into a robust commercial reality. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with the necessary corrosion-resistant reactors and cryogenic capabilities to handle tetrafluoroethylene chemistry safely, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of florfenicol intermediate meets the highest international standards.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can optimize your bottom line. We encourage you to reach out today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments that will secure your position as a leader in the competitive veterinary antibiotics market.
