Optimizing Acyclovir Production Through Advanced N2-Formylguanine Alkylation Technology
Optimizing Acyclovir Production Through Advanced N2-Formylguanine Alkylation Technology
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiviral agents, and the production of 9-(2-hydroxy)-ethoxymethylguanine, universally known as Acyclovir, remains a cornerstone of herpes treatment protocols. The technical landscape for synthesizing this vital compound has evolved significantly, moving away from cumbersome early methods toward more efficient, regioselective processes. A pivotal advancement in this domain is documented in Chinese Patent CN1041632C, which outlines a superior methodology utilizing N2-formylguanine as a key intermediate. This approach addresses longstanding challenges regarding isomeric purity and overall yield that have plagued previous generations of synthesis. By leveraging the unique electronic properties of the formyl group, manufacturers can achieve exceptional control over the alkylation site on the purine ring. This report analyzes the technical merits of this patented process, offering strategic insights for R&D directors and procurement specialists aiming to optimize their antiviral supply chains.
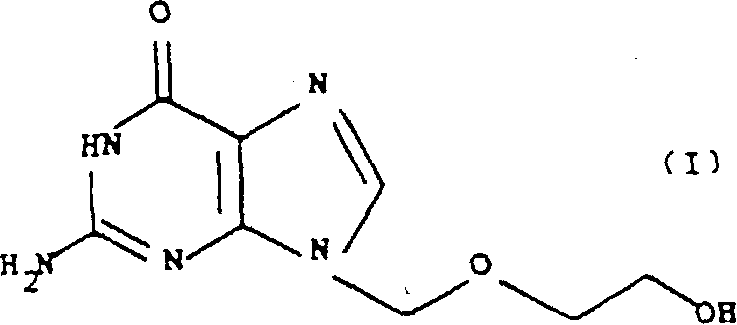
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of acyclovir relied heavily on silylation strategies, such as those described in Belgian Patent 833,006. These conventional routes typically involve the trisilylation of guanine at the 2-amino, 6, and 9 positions to activate the molecule for subsequent alkylation. While chemically feasible, this method suffers from severe economic and technical drawbacks that impact commercial viability. The process necessitates the use of a vast excess of silylating reagents, which not only drives up raw material costs but also generates substantial chemical waste, complicating environmental compliance and disposal logistics. Furthermore, the lack of precise regiocontrol often leads to the formation of the unwanted 7-substituted isomer in pharmaceutically unacceptable quantities. Removing this structural impurity requires additional, costly purification steps that erode profit margins and extend production lead times, making these legacy processes less attractive for modern, high-volume manufacturing environments.
The Novel Approach
In stark contrast to the silylation-heavy legacy methods, the novel approach detailed in the patent data introduces a streamlined pathway centered on N2-formylguanine. This innovative strategy bypasses the need for excessive silylation by employing a formyl group to protect the exocyclic amino function. The brilliance of this method lies in its ability to direct the alkylation of 2-oxa-1,4-diacetoxybutane specifically to the N9 position of the purine ring. By avoiding the formation of the problematic 7-isomer almost entirely, the process simplifies the downstream purification burden significantly. The reaction conditions are milder and more manageable, utilizing standard oxidants and alkylating agents rather than moisture-sensitive silyl chlorides. This shift represents a fundamental improvement in process chemistry, offering a cleaner, more direct route to the target molecule that aligns perfectly with the principles of green chemistry and cost-effective industrial production.
Mechanistic Insights into N2-Formylguanine Alkylation
The core of this technological breakthrough rests on the specific reactivity of the N2-formylguanine intermediate. The formyl group acts as a temporary protecting group that modulates the electron density of the purine ring system. When guanine is converted to its N2-formyl derivative, either through the oxidation of a glyoxal-guanine adduct or direct formylation, the resulting molecule exhibits a strong preference for nucleophilic attack at the N9 nitrogen atom. This regioselectivity is crucial because the biological activity of acyclovir is strictly dependent on the side chain being attached at the N9 position; the N7 isomer is biologically inactive and considered a toxic impurity. The patent data highlights that reacting N2-formylguanine with 2-oxa-1,4-diacetoxybutane yields the N9-alkylated product in yields exceeding 90%, with only trace amounts of the 7-isomer detected. This high degree of selectivity is a direct result of the steric and electronic influence of the formyl moiety, which effectively shields the N7 position while activating N9 for the substitution reaction.
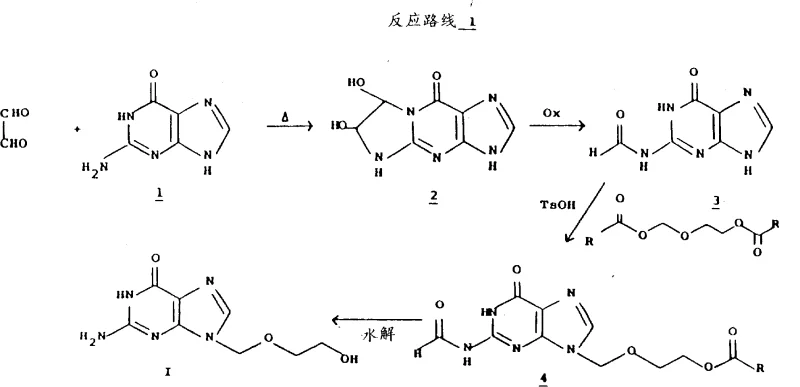
Furthermore, the mechanism includes a sophisticated purification protocol that leverages anion exchange chromatography to ensure ultimate product quality. Even with high regioselectivity, trace amounts of starting material or hydrolysis byproducts like guanine can persist. The patented process addresses this by dissolving the crude acyclovir in an alkaline solution and passing it through a strong anion exchange resin, such as Amberlite IRA-400. Under these basic conditions, the guanine impurity interacts differently with the resin compared to the acyclovir molecule, allowing for effective separation. The elution profile is carefully controlled using a mixture of lower alcohol and aqueous alkali, ensuring that the target product is recovered with exceptional purity. This mechanistic understanding of both the synthetic transformation and the purification physics allows process engineers to design reactors and columns that maximize throughput while maintaining stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Acyclovir Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the oxidation and alkylation stages. The process begins with the preparation of the glyoxal-guanine adduct, which is subsequently oxidized using sodium periodate at a controlled pH range of 1.8 to 2.2 to generate the critical N2-formylguanine intermediate. Following isolation, this intermediate undergoes alkylation with 2-oxa-1,4-diacetoxybutane in the presence of an acid catalyst like p-toluenesulfonic acid. The reaction is typically conducted under reduced pressure to facilitate the removal of acetic acid byproduct, driving the equilibrium toward the desired N9-alkylated species. Finally, a two-stage hydrolysis removes the protecting groups to reveal the active pharmaceutical ingredient. For a comprehensive, step-by-step technical guide including exact stoichiometry and safety protocols, please refer to the standardized operating procedure below.
- Oxidation of glyoxal-guanine adduct to N2-formylguanine using sodium periodate at pH 1.8-2.2.
- Regioselective alkylation of N2-formylguanine with 2-oxa-1,4-diacetoxybutane to form the N9-substituted intermediate.
- Hydrolysis of the intermediate followed by anion exchange chromatography to remove guanine impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this N2-formylguanine-based process offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on expensive and hazardous silylating agents, manufacturers can source cheaper, more stable commodities like formic acid derivatives and diacetoxybutane. This shift reduces the volatility of input costs and mitigates the risks associated with handling moisture-sensitive reagents that require specialized storage and transport. Additionally, the high regioselectivity of the reaction means that less material is wasted on inactive isomers, directly improving the mass balance of the production line. This efficiency translates into a lower cost of goods sold (COGS) and a more competitive pricing structure for the final API intermediate, providing a distinct market advantage.
- Cost Reduction in Manufacturing: The elimination of expensive silylation reagents and the reduction in solvent usage for purification significantly lowers the overall production cost. Since the process avoids the formation of difficult-to-remove 7-isomers, the need for repetitive recrystallization or complex chromatographic separations is minimized, leading to substantial savings in energy and labor. The high yield of the alkylation step ensures that the maximum amount of starting material is converted into valuable product, optimizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Sourcing N2-formylguanine or its precursors is more straightforward than managing the supply of specialized silyl chlorides, which can be subject to market fluctuations. The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, reducing the risk of production delays caused by failed runs or off-spec material. This reliability is critical for maintaining continuous supply to downstream API manufacturers, ensuring that medication production schedules are met without interruption due to intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor equipment and avoiding extreme temperatures or pressures that complicate scale-up. From an environmental perspective, the reduction in chemical waste and the avoidance of heavy metal catalysts or toxic silyl byproducts simplify wastewater treatment and regulatory compliance. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced acyclovir synthesis technology. These answers are derived directly from the experimental data and claims found in the underlying patent literature, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does the N2-formyl protection strategy improve regioselectivity compared to silylation?
A: Unlike traditional silylation methods which often result in significant 7-isomer formation requiring complex purification, the N2-formyl group effectively blocks the N7 position sterically and electronically, directing alkylation almost exclusively to the N9 position with yields exceeding 90%.
Q: What purification method ensures USP-grade purity for the final acyclovir product?
A: The process utilizes strong anion exchange resin chromatography (e.g., Amberlite IRA-400) to separate acyclovir from unreacted guanine, achieving impurity levels below 0.1%, which is significantly purer than standard commercial grades.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the reaction conditions utilize common reagents like sodium periodate and diacetoxybutane without requiring cryogenic temperatures or exotic catalysts, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyclovir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the N2-formylguanine alkylation process requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory bench to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace isomers at parts-per-million levels. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance guarantees that every kilogram of intermediate meets the highest international standards.
We invite you to collaborate with us to leverage these technological advancements for your antiviral drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of acyclovir intermediates that will strengthen your market position and accelerate your time to market.
