Advanced Metal Complex Intermediates for Scalable Cannabidiol Manufacturing and Purification
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to enhance the production efficiency of high-value active compounds, particularly within the cannabinoid sector. Patent CN115724739A introduces a significant technological advancement by disclosing a class of resorcinol carboxylate metal complexes, eutectics, or salts that serve as superior intermediates for the preparation of Cannabidiol (CBD) and its analogues. Traditional synthetic routes often struggle with the physical properties of key intermediates, which frequently exist as viscous liquids that are notoriously difficult to purify to the stringent standards required for pharmaceutical applications. This innovation addresses these critical bottlenecks by transforming liquid intermediates into stable, crystalline solid forms through coordination with specific metal ions. The ability to isolate these intermediates as solids allows for conventional purification techniques such as recrystallization and filtration, which are far more scalable and cost-effective than chromatographic methods. For R&D directors and process chemists, this represents a pivotal shift towards more controllable and reproducible manufacturing processes, ensuring that the final API or intermediate meets rigorous quality specifications regarding impurity profiles and chemical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
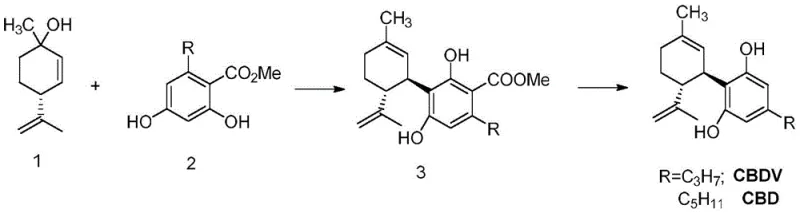
Historically, the synthesis of Cannabidiol and its structural analogues has relied heavily on Friedel-Crafts alkylation reactions followed by de-esterification steps to yield the final phenolic products. A significant drawback identified in prior art, such as the methods described in CN 111183135, is the physical state of the key intermediate compounds generated during these sequences. These intermediates typically exist as liquids with high boiling points, rendering them incompatible with standard purification techniques like distillation or simple crystallization. Consequently, manufacturers are often forced to rely on column chromatography or other labor-intensive purification methods to remove side products and unreacted starting materials. This reliance not only increases the operational complexity and time required for production but also introduces significant variability in batch-to-batch consistency. Furthermore, the liquid nature of these intermediates complicates quality control analysis, as impurities may be trapped within the viscous matrix, leading to potential issues with the final product's impurity spectrum. For supply chain managers, these inefficiencies translate into longer lead times and higher production costs, making the commercial scale-up of such processes economically challenging.
The Novel Approach
The novel approach detailed in patent CN115724739A circumvents these traditional limitations by introducing a metal complexation step that fundamentally alters the physical properties of the synthesis intermediate. By reacting the resorcinol carboxylate precursor with specific metal sources such as magnesium, calcium, or zinc salts, the process generates a metal complex that possesses a well-defined solid crystal structure. This transformation from a difficult-to-handle liquid to a manageable solid is a game-changer for process chemistry. The solid form allows for easy isolation via filtration, and impurities can be effectively washed away using appropriate solvents, significantly enhancing the purity of the intermediate before it proceeds to the final conversion step. This method not only simplifies the operational workflow but also improves the overall yield of the process by minimizing product loss associated with complex purification techniques. The robustness of this solid-state intermediate ensures that the subsequent conversion to Cannabidiol is performed with a higher quality starting material, thereby reducing the burden on downstream purification and ensuring a more consistent final product quality.
Mechanistic Insights into Resorcinol Carboxylate Metal Coordination
The core of this technological breakthrough lies in the coordination chemistry between the resorcinol carboxylate ligand and the selected metal centers. The resorcinol moiety contains phenolic hydroxyl groups that act as Lewis bases, capable of donating electron pairs to metal ions such as Magnesium (Mg), Calcium (Ca), or Zinc (Zn). In the presence of a base or specific metal alkyl/halide reagents, deprotonation of the phenolic hydroxyl groups occurs, facilitating the formation of strong coordination bonds. This interaction creates a stable chelate or complex structure, often represented by general formula I in the patent documentation. The formation of these bonds restricts the molecular motion and organizes the molecules into a regular lattice structure, which is the driving force behind the crystallization of the intermediate. The choice of metal is critical; for instance, magnesium complexes may form via reaction with Grignard reagents or magnesium alkoxides, while calcium and zinc complexes can be formed using their respective salts in aqueous or alcoholic media. This versatility allows process chemists to select the most cost-effective and operationally safe metal source for their specific manufacturing environment.
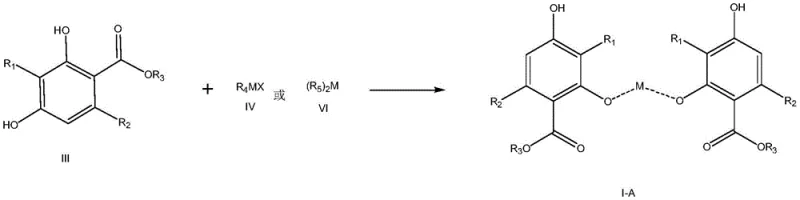
Furthermore, the stability of these metal complexes plays a crucial role in impurity control. By locking the intermediate into a solid coordination complex, the reactivity of the functional groups is temporarily modulated, preventing unwanted side reactions that might occur in the free liquid state. This stabilization is particularly important during the isolation and storage phases, where the intermediate might otherwise degrade or react with atmospheric moisture. The purification mechanism relies on the differential solubility of the metal complex compared to organic impurities. While the complex precipitates out of the solution as a solid, many organic by-products remain dissolved in the mother liquor and are removed during filtration. This selective precipitation acts as a powerful purification step, effectively narrowing the impurity profile before the final deprotection step. For R&D teams, understanding this mechanistic advantage is key to optimizing reaction conditions such as temperature, solvent choice, and stoichiometry to maximize the crystallinity and purity of the complex.
How to Synthesize Cannabidiol Intermediates Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure the formation of the desired metal complex polymorph. The process generally involves dissolving the resorcinol carboxylate precursor in a suitable solvent such as tetrahydrofuran, toluene, or alcohols, followed by the controlled addition of the metalating agent. Temperature control is paramount, with reactions often conducted at low temperatures ranging from -30°C to 10°C to manage exotherms and ensure selective complexation. Following the reaction, the solvent is concentrated, and a pulping solvent is added to induce crystallization. The resulting solid is then filtered and dried to yield the pure metal complex. Detailed standardized synthesis steps see the guide below.
- Prepare the resorcinol carboxylate precursor and react with a metal source such as magnesium, calcium, or zinc salts under controlled basic conditions.
- Isolate the resulting metal complex intermediate which forms a stable solid crystal structure, allowing for efficient filtration and washing to remove impurities.
- Convert the purified metal complex into the final Cannabidiol product through deprotection and acidification steps in a suitable solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal complex technology offers substantial advantages for procurement and supply chain operations within the fine chemical and pharmaceutical sectors. The primary value driver is the significant simplification of the purification process, which directly correlates to reduced manufacturing costs and improved throughput. By eliminating the need for extensive chromatographic purification of liquid intermediates, manufacturers can reduce solvent consumption, waste generation, and labor hours. This efficiency gain allows for a more competitive pricing structure for the final Cannabidiol intermediates, making it an attractive option for buyers looking to optimize their cost of goods sold. Additionally, the solid nature of the intermediate enhances supply chain reliability by improving the stability and shelf-life of the material during storage and transportation. Liquid intermediates often require specialized handling and are prone to degradation, whereas solid metal complexes are robust and easier to manage in a warehouse environment.
- Cost Reduction in Manufacturing: The transition from liquid to solid intermediates fundamentally changes the cost structure of the synthesis. Conventional methods often incur high costs due to the need for specialized equipment to handle viscous liquids and the expensive solvents required for chromatography. In contrast, the metal complex method utilizes standard filtration and crystallization equipment, which is widely available and less capital intensive. The reduction in solvent usage for purification also lowers waste disposal costs, contributing to a more sustainable and economically viable production model. Furthermore, the higher yields associated with easier purification mean that less raw material is wasted, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability of complex chemical processes. The robustness of the metal complex intermediate mitigates this risk by providing a stable checkpoint in the synthesis workflow. Because the intermediate can be isolated, characterized, and stored as a solid, it decouples the upstream synthesis from the downstream conversion. This flexibility allows manufacturers to build inventory of the stable intermediate, ensuring that final product delivery schedules can be met even if there are fluctuations in the final conversion step. For procurement managers, this means a more reliable supplier capable of meeting demand without the frequent delays associated with troubleshooting difficult liquid-phase reactions.
- Scalability and Environmental Compliance: Scaling up chemical processes from the lab to commercial production is fraught with challenges, particularly when dealing with difficult-to-purify liquids. The solid-state nature of these metal complexes makes the process inherently more scalable, as filtration and crystallization are unit operations that scale linearly and predictably. This ease of scale-up reduces the time and investment required to bring new products to market. Additionally, the process aligns well with environmental compliance goals by reducing the volume of organic waste solvents generated. The ability to use greener solvents for crystallization and the reduction in overall solvent intensity supports corporate sustainability initiatives and regulatory compliance regarding waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal complex technology in Cannabidiol production. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing frameworks.
Q: Why are metal complex intermediates superior to liquid intermediates in CBD synthesis?
A: Liquid intermediates often suffer from high boiling points and difficulty in purification via conventional methods like distillation or crystallization. Metal complexes transform these intermediates into stable solid forms with defined crystal structures, significantly simplifying the purification process and improving overall product purity.
Q: Which metals are suitable for forming these resorcinol carboxylate complexes?
A: The patent specifies magnesium, calcium, and zinc as effective metals for coordination. These metals form stable coordination bonds with the phenolic hydroxyl groups and carboxylate moieties, creating robust complexes that can be easily isolated and handled in large-scale production.
Q: Does this method impact the final stereochemistry of the Cannabidiol?
A: The process is designed to maintain the structural integrity of the cannabinoid scaffold. The coordination chemistry focuses on the phenolic and ester functionalities without disrupting the chiral centers established in the earlier Friedel-Crafts alkylation steps, ensuring the final product meets stereochemical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabidiol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical intermediate market. Our technical team has thoroughly analyzed the potential of the metal complex route described in CN115724739A and is well-positioned to implement this methodology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for cannabinoid intermediates. We understand that consistency is key, and our process engineering experts are dedicated to optimizing the crystallization and filtration steps to maximize yield and quality.
We invite procurement leaders and R&D directors to collaborate with us to leverage this technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific metal complex route can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to provide not just a chemical product, but a comprehensive solution that enhances your operational efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
