Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Pharmaceutical Intermediates
Introduction to Next-Generation Indole Synthesis
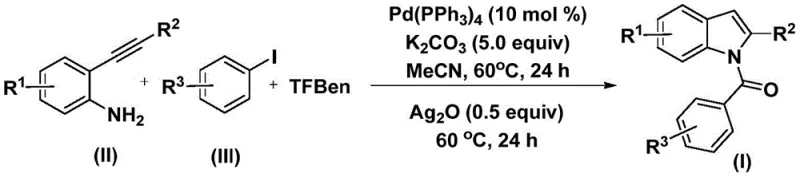
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules including anti-inflammatory, anti-tumor, and anti-viral agents. The efficient construction of N-acyl indole derivatives remains a critical challenge for process chemists aiming to streamline the supply chain for high-purity pharmaceutical intermediates. A groundbreaking approach detailed in Chinese Patent CN112898192B introduces a robust, palladium-catalyzed methodology that circumvents traditional limitations associated with carbonylation reactions. This innovation leverages a solid carbon monoxide surrogate to achieve a one-pot synthesis, significantly enhancing operational safety and reaction efficiency. By integrating oxidative cyclization directly into the catalytic cycle, this process offers a streamlined pathway for generating complex heterocyclic architectures essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for N-acyl indoles often rely on the direct use of carbon monoxide gas, which presents severe safety hazards due to its high toxicity and flammability. Furthermore, gas-liquid reactions typically require specialized high-pressure autoclaves and rigorous containment protocols, creating substantial barriers to entry for standard laboratory setups and complicating scale-up efforts in commercial manufacturing environments. Alternative methods involving pre-functionalized acyl chlorides can generate stoichiometric amounts of corrosive waste and often suffer from poor atom economy. Additionally, multi-step sequences required to install the acyl group post-cyclization frequently lead to reduced overall yields and increased production costs, making these legacy processes less attractive for the cost-sensitive production of fine chemicals and active pharmaceutical ingredients.
The Novel Approach
The methodology disclosed in the patent revolutionizes this landscape by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe, solid carbon monoxide source, effectively eliminating the need for hazardous gas handling infrastructure. This innovative strategy utilizes a dual-catalytic system where palladium facilitates the initial carbonylative coupling, followed by a silver-mediated oxidative cyclization, all within a single reaction vessel. The process operates under remarkably mild conditions, specifically at a constant temperature of 60°C in acetonitrile, ensuring high energy efficiency and minimizing thermal degradation of sensitive substrates. This one-pot protocol not only simplifies the workflow by removing intermediate isolation steps but also demonstrates exceptional functional group tolerance, allowing for the rapid diversification of the indole core with various electronic and steric properties.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The reaction mechanism initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This electrophilic acyl complex then undergoes nucleophilic attack by the amino group of the 2-alkynylaniline, followed by reductive elimination to yield an amide intermediate while regenerating the active palladium catalyst. This sequence effectively constructs the exocyclic amide bond with high precision, setting the stage for the subsequent ring-closing event that defines the indole architecture.
In the second phase of the reaction, the introduction of silver oxide serves a dual purpose as both an oxidant and a cyclization promoter. The silver species likely activates the alkyne moiety of the amide intermediate towards intramolecular nucleophilic attack by the adjacent nitrogen or facilitates an oxidative C-H activation process. This step drives the thermodynamic equilibrium towards the formation of the fused five-membered pyrrole ring, completing the indole skeleton. The use of silver oxide under these mild thermal conditions ensures that the cyclization proceeds smoothly without requiring harsh acidic or basic conditions that could compromise the integrity of acid-sensitive functional groups, thereby preserving the structural diversity of the final N-acyl indole products.
How to Synthesize N-Acyl Indole Efficiently
To implement this sophisticated synthesis in a practical setting, precise control over reagent stoichiometry and reaction timing is paramount to achieving optimal yields. The protocol dictates a sequential addition strategy where the initial carbonylation phase is allowed to reach completion before the oxidative cyclization agent is introduced. This temporal separation prevents potential interference between the palladium catalytic cycle and the silver oxidant, ensuring that each transformation proceeds with maximum efficiency. Operators should adhere strictly to the specified molar ratios, particularly the 10 mol % loading of the palladium catalyst and the 5.0 equivalents of potassium carbonate base, to maintain the necessary reaction kinetics. For a comprehensive, step-by-step guide on executing this synthesis with standardized parameters, please refer to the detailed instructions below.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, carbon monoxide substitute (TFBen), 2-alkynylaniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers profound advantages by fundamentally altering the cost structure of N-acyl indole production. The substitution of gaseous carbon monoxide with a stable solid surrogate drastically reduces the capital expenditure associated with safety infrastructure, such as gas detection systems and high-pressure reactors, leading to significant overhead savings. Moreover, the reliance on commercially available starting materials like aryl iodides and 2-alkynylanilines ensures a robust and resilient supply chain, mitigating the risks associated with sourcing exotic or unstable reagents. The simplified one-pot nature of the process also translates to reduced labor costs and shorter batch cycle times, as the need for intermediate workups and purifications is eliminated, thereby enhancing overall throughput capacity.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the use of readily available solid reagents significantly lower the barrier to entry for production, resulting in substantial operational cost savings. By avoiding the complexities of gas-liquid mass transfer and the need for specialized containment, manufacturers can utilize standard glass-lined or stainless steel reactors, optimizing asset utilization. Furthermore, the high atom economy of the carbonylation step minimizes raw material waste, contributing to a more sustainable and economically viable production model that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solids like TFBen and common aryl halides ensures consistent quality and availability, reducing the volatility often seen with gaseous reagents. This stability allows for better inventory management and long-term planning, as the key carbon source does not degrade over time or require special storage conditions. Consequently, production schedules become more predictable, and the risk of supply disruptions due to logistics issues with hazardous gases is effectively neutralized, securing a steady flow of critical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions of 60°C and the absence of toxic gas emissions make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The simplified workup procedure, involving basic filtration and chromatography, reduces the volume of solvent waste and hazardous byproducts generated per unit of product. This alignment with environmental regulations reduces the burden on waste treatment facilities and lowers compliance costs, making the technology highly attractive for large-scale commercial adoption in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing a reliable foundation for process decision-making.
Q: Why is 1,3,5-tricarboxylic acid phenol ester (TFBen) used instead of carbon monoxide gas?
A: TFBen serves as a solid carbon monoxide surrogate, eliminating the safety hazards and specialized high-pressure equipment required for handling toxic CO gas, thereby simplifying the operational protocol and enhancing workplace safety.
Q: What is the specific role of silver oxide in this reaction mechanism?
A: Silver oxide acts as a crucial oxidant and cyclization promoter in the second stage of the reaction, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure under mild thermal conditions.
Q: Does this method support a wide range of functional groups on the substrate?
A: Yes, the patented method demonstrates excellent substrate compatibility, tolerating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on the aromatic rings without significant loss in yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can be tailored to your specific project needs. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, one-pot methodology. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring your supply chain is optimized for both performance and profitability.
