Advanced Palladium-Catalyzed Carbonylation for Efficient N-Acyl Indole Production and Commercial Scale-Up
Advanced Palladium-Catalyzed Carbonylation for Efficient N-Acyl Indole Production and Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as privileged structures in medicinal chemistry. Patent CN112898192B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for N-acyl indole compounds. This technology leverages a palladium-catalyzed carbonylation cyclization strategy that utilizes 2-alkynyl aniline and aryl iodides as primary building blocks. Unlike traditional approaches that often rely on hazardous gaseous carbon monoxide, this novel process employs a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), thereby enhancing operational safety and scalability. The method operates under relatively mild conditions, typically at 60°C, and demonstrates remarkable versatility across a wide range of substrates, making it an attractive candidate for the industrial production of high-value pharmaceutical intermediates.
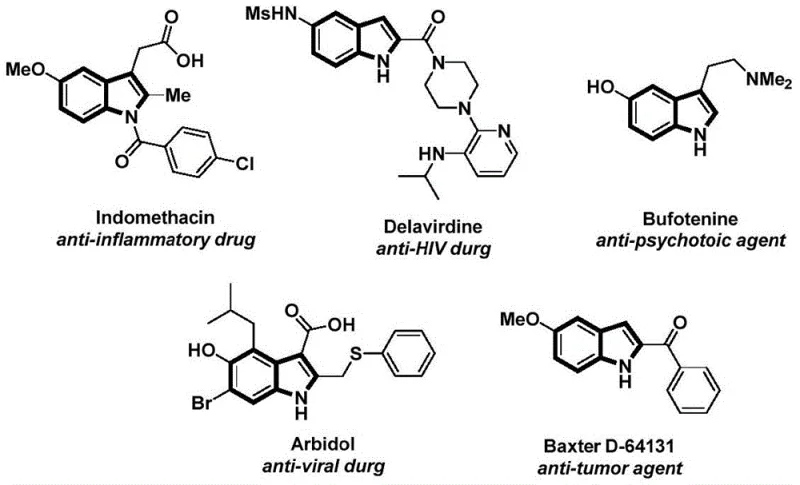
Indole motifs are ubiquitous in nature and are critical components of numerous FDA-approved drugs exhibiting antitumor, anti-inflammatory, antiviral, and antidepressant activities. As illustrated in the structural diversity of known agents, the ability to functionalize the nitrogen atom of the indole ring with acyl groups is a frequent requirement in drug discovery and process development. The methodology described in CN112898192B addresses the growing demand for reliable pharmaceutical intermediate supplier capabilities by offering a direct route to these N-acylated systems. By integrating the carbonylation and cyclization steps into a streamlined sequence, this invention not only simplifies the synthetic workflow but also potentially reduces the environmental footprint associated with multi-step syntheses, aligning with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has been fraught with challenges related to reagent toxicity, harsh reaction conditions, and limited substrate scope. Conventional carbonylation reactions frequently necessitate the use of pressurized carbon monoxide gas, which poses severe safety risks including toxicity and explosion hazards, requiring specialized high-pressure reactors and rigorous safety protocols that increase capital expenditure. Furthermore, traditional methods often suffer from poor regioselectivity or require pre-functionalized starting materials that are expensive and difficult to source on a large scale. Many existing protocols also rely on stoichiometric amounts of toxic heavy metals or strong bases that generate significant waste streams, complicating downstream purification and waste treatment. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing.
The Novel Approach
The innovative strategy outlined in the patent overcomes these hurdles by utilizing a solid CO surrogate, TFBen, which releases carbon monoxide in situ under thermal conditions, effectively bypassing the need for gas cylinders and high-pressure equipment. This approach allows the reaction to proceed in standard glassware or stainless steel reactors at atmospheric pressure, significantly lowering the barrier to entry for commercial scale-up of complex heterocycles. The process employs a dual-stage heating protocol where the initial coupling occurs at 60°C for 24 hours, followed by the addition of silver oxide and a subsequent 24-hour heating period to drive the cyclization to completion. This one-pot tandem reaction design minimizes solvent usage and intermediate isolation steps, leading to improved overall atom economy. The use of readily available palladium catalysts and inexpensive inorganic bases like potassium carbonate further enhances the economic viability of this route for industrial applications.
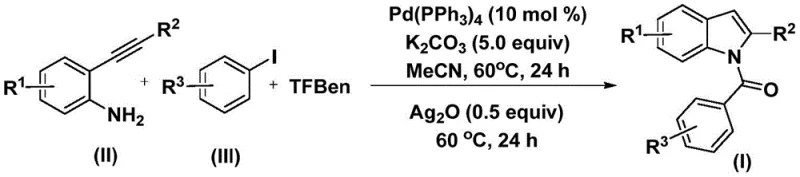
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction mechanism proceeds through a sophisticated catalytic cycle initiated by the oxidative addition of the aryl iodide to the zero-valent palladium species, generating an aryl-palladium(II) intermediate. Concurrently, the thermal decomposition of TFBen releases carbon monoxide, which subsequently inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, resulting in the formation of an amide linkage and regenerating the active palladium catalyst after reductive elimination. This initial phase constructs the N-aryl amide backbone with high fidelity, setting the stage for the subsequent ring closure. The careful selection of ligands and the specific oxidation state of the palladium precursor are critical to maintaining catalytic turnover and preventing catalyst deactivation during this extended heating period.
In the second phase of the transformation, the introduction of silver oxide plays a pivotal role in promoting the intramolecular cyclization. The silver species likely acts as a soft Lewis acid or an oxidant that activates the alkyne moiety towards nucleophilic attack by the adjacent aromatic ring or facilitates a C-H activation process, depending on the specific electronic nature of the substituents. This step effectively closes the five-membered pyrrole ring of the indole system, yielding the final N-acyl indole product. The tolerance of the catalytic system to various functional groups, including electron-withdrawing halogens and electron-donating methoxy groups, suggests a robust mechanism that is not easily perturbed by steric or electronic variations in the substrate. This mechanistic resilience ensures consistent product quality and high-purity N-acyl indole outputs even when processing diverse raw material batches.
How to Synthesize N-Acyl Indole Compounds Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls as defined in the patent examples. The process begins with the thorough mixing of the palladium catalyst, base, CO source, and substrates in an anhydrous organic solvent such as acetonitrile. Maintaining an inert atmosphere is recommended to protect the sensitive palladium species from oxidation prior to the reaction start. The following section details the standardized operational parameters required to achieve optimal yields and purity profiles suitable for pharmaceutical grade materials.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to complete cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this technology offers tangible benefits regarding cost stability and logistical simplicity. By replacing hazardous gaseous reagents with stable solids, the process eliminates the need for specialized gas handling infrastructure and reduces regulatory compliance burdens associated with toxic gas storage. This shift translates directly into lower overhead costs and reduced insurance premiums for manufacturing facilities. Furthermore, the use of commodity chemicals like potassium carbonate and commercially available aryl iodides ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source vendor lock-ins. The ability to source raw materials from multiple global suppliers enhances negotiating power and secures long-term supply continuity for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactor requirements and the use of a solid CO source significantly reduce capital investment and operational complexity. Additionally, the one-pot nature of the reaction minimizes solvent consumption and labor hours associated with intermediate workups, leading to substantial cost savings in the overall production budget. The high atom efficiency of the carbonylation step ensures that expensive starting materials are converted into product with minimal waste, optimizing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on shelf-stable reagents like TFBen and common inorganic salts mitigates the risk of supply interruptions caused by the transportation restrictions often placed on hazardous gases. This stability allows for larger inventory buffers to be maintained safely on-site, ensuring uninterrupted production schedules even during global logistics slowdowns. The broad substrate scope means that alternative starting materials can be quickly validated if a specific supplier faces issues, providing flexibility and redundancy in the sourcing strategy.
- Scalability and Environmental Compliance: The mild reaction conditions of 60°C are energy-efficient compared to high-temperature processes, reducing the carbon footprint of the manufacturing operation. The simplified post-treatment involving filtration and column chromatography is amenable to scaling, and the absence of toxic gas emissions simplifies environmental permitting and waste gas treatment. This alignment with green chemistry standards facilitates faster regulatory approvals and supports corporate sustainability goals without compromising on production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these nuances is essential for R&D teams planning to transfer this technology from the laboratory to pilot or commercial scales. The answers provided are derived directly from the experimental data and specifications contained within the patent documentation.
Q: What is the advantage of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid, safe, and easy-to-handle carbon monoxide surrogate, eliminating the need for high-pressure CO gas equipment and significantly improving laboratory and plant safety profiles.
Q: What is the role of Silver Oxide (Ag2O) in this reaction?
A: Silver oxide acts as a crucial additive in the second stage of the reaction, facilitating the oxidative cyclization step that converts the intermediate amide into the final N-acyl indole structure.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN112898192B for the rapid development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market supply is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of N-acyl indole intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for high-purity pharmaceutical intermediates.
