Advanced 3-Step Synthesis of Mirabegron: Scalable Manufacturing for Global Pharma Supply Chains
The pharmaceutical landscape for overactive bladder treatments has been significantly shaped by the introduction of Mirabegron, a potent beta-3 adrenergic receptor agonist. As global demand for this active pharmaceutical ingredient (API) continues to rise, the efficiency of its supply chain becomes a critical focal point for procurement and R&D leaders alike. Patent CN103896872A introduces a transformative synthetic methodology that addresses the longstanding bottlenecks associated with traditional Mirabegron production. This technical insight report analyzes the patented three-step synthesis route, which strategically bypasses the cumbersome protection-deprotection sequences typical of earlier generations of synthesis. By leveraging a direct ring-opening strategy followed by catalytic hydrogenation and condensation, this approach offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks seeking to optimize their manufacturing portfolios.
The core innovation lies in the direct utilization of p-nitrophenylethylamine and (R)-styrene oxide as starting materials. Unlike previous iterations that required extensive functional group masking, this method capitalizes on the specific reactivity profiles of these precursors to construct the chiral amino-alcohol backbone in fewer operational units. For decision-makers evaluating cost reduction in pharmaceutical intermediates manufacturing, the elimination of auxiliary reagents such as di-tert-butyl dicarbonate (Boc2O) represents a tangible decrease in raw material expenditure. Furthermore, the process is designed with commercial scale-up of complex pharmaceutical intermediates in mind, prioritizing unit operations like recrystallization over labor-intensive chromatographic separations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mirabegron has been plagued by inefficiencies that hinder large-scale production capabilities. The conventional route, often referenced in early literature such as the Maruyama method, relies heavily on the use of tert-butoxycarbonyl (Boc) protecting groups to manage the nucleophilicity of the amine functionality during the synthesis. This necessitates an additional protection step prior to the formation of the core structure and a subsequent deprotection step to reveal the active amine for final coupling. These extra steps not only increase the overall reaction time but also introduce significant purification challenges.
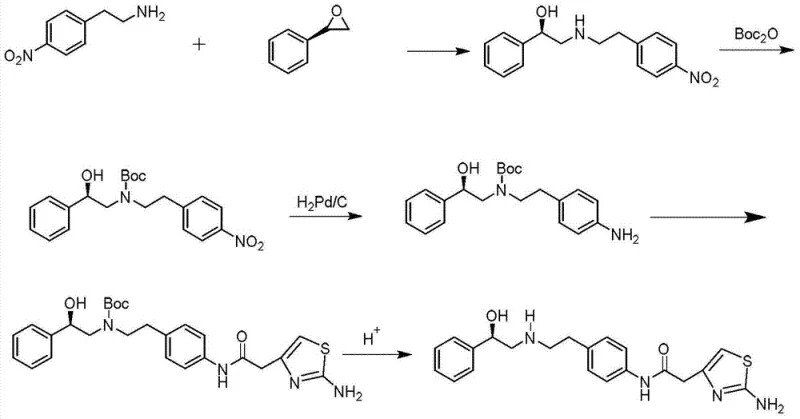
Specifically, the reliance on column chromatography for the purification of Boc-protected intermediates is a major bottleneck for industrial application. Chromatography is inherently difficult to scale, consumes vast quantities of organic solvents, and results in lower throughput compared to crystallization-based workflows. Additionally, the use of expensive reagents like Boc anhydride drives up the cost of goods sold (COGS). The cumulative effect of these factors is a process with a lower overall yield, higher environmental footprint due to solvent waste, and a supply chain that is vulnerable to delays caused by complex post-treatment requirements. These limitations make the conventional route less attractive for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
The Novel Approach
In stark contrast, the methodology disclosed in CN103896872A streamlines the synthetic trajectory into a concise three-step sequence that effectively circumvents the need for amine protection. The process initiates with a regioselective ring-opening reaction between p-nitrophenylethylamine and (R)-styrene oxide. This step constructs the critical chiral amino-alcohol scaffold directly, establishing the stereochemistry early in the synthesis without the need for masking groups. The subsequent reduction of the nitro group to an amine is achieved via catalytic hydrogenation, a robust and scalable transformation.
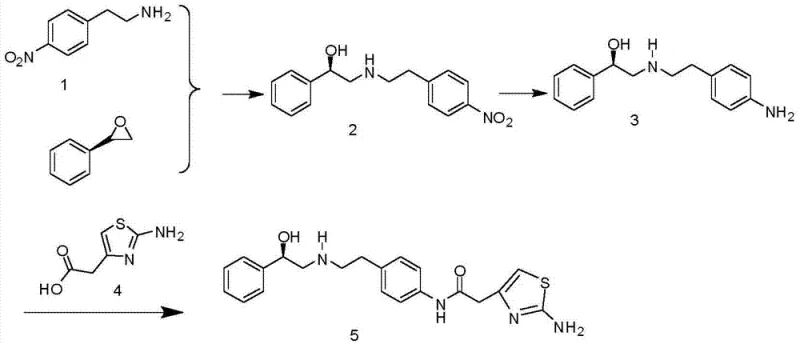
The final step involves the condensation of the resulting diamine intermediate with 2-(2-aminothiazol-4-yl)acetic acid to form the target amide bond. A key differentiator of this novel approach is the purification strategy; instead of column chromatography, the intermediates and the final product are purified via recrystallization. This shift from chromatographic to crystalline purification is a game-changer for manufacturing scalability. It allows for the processing of larger batch sizes with significantly reduced solvent consumption and operational complexity. For supply chain heads, this translates to a more resilient production schedule and a product profile that meets stringent quality standards with greater consistency and efficiency.
Mechanistic Insights into Regioselective Ring-Opening and Catalytic Reduction
The success of this synthetic route hinges on the precise control of the initial ring-opening reaction. When p-nitrophenylethylamine reacts with (R)-styrene oxide, the nucleophilic attack occurs preferentially at the less hindered benzylic position of the epoxide, although electronic factors also play a role. The reaction is typically conducted in solvents such as acetonitrile or isopropanol at temperatures ranging from 60°C to 70°C. Under these conditions, the amine attacks the epoxide ring to open it, forming the (R)-2-((4-nitrophenethyl)amino)-1-phenylethanol intermediate. The stereochemical integrity of the (R)-styrene oxide is largely preserved during this transformation, ensuring that the final product maintains the required optical purity essential for biological activity.
Following the ring-opening, the nitro group on the aromatic ring must be reduced to an aniline derivative to enable the final amide coupling. The patent specifies the use of catalytic hydrogenation, typically employing 5% Pd/C as the catalyst under a hydrogen atmosphere at room temperature. This reduction is highly chemoselective; it reduces the nitro group to the primary amine without affecting the secondary benzylic alcohol or the chiral center. The use of Pd/C is advantageous not only for its selectivity but also for its ease of removal via filtration, allowing the catalyst to be recovered and potentially recycled. This mechanistic elegance ensures that the intermediate obtained is of high purity, ready for the final coupling step without the need for extensive purification, thereby maintaining the overall efficiency of the process.
How to Synthesize Mirabegron Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the nitro-alcohol intermediate, followed by its reduction to the diamine, and concludes with the amide coupling. Each step has been optimized to minimize side reactions and facilitate easy isolation of the product. The detailed operational procedures, including specific molar ratios, solvent choices, and temperature controls, are critical for reproducing the high yields reported in the patent data. For R&D teams looking to adopt this technology, understanding the nuances of the recrystallization solvents and the stoichiometry of the coupling agents is paramount.
- Perform a regioselective ring-opening reaction between p-nitrophenylethylamine and (R)-styrene oxide at 60-70°C to form the nitro-alcohol intermediate.
- Execute a catalytic hydrogenation reduction using Pd/C in an alcohol solvent to convert the nitro group to an amine without affecting the chiral center.
- Conduct a condensation reaction with 2-(2-aminothiazol-4-yl)acetic acid using EDCI/HOBt coupling agents, followed by recrystallization for high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial benefits that extend beyond mere technical feasibility. For procurement managers and supply chain directors, the primary value drivers are cost efficiency, supply reliability, and environmental compliance. The elimination of the Boc protection strategy removes a significant cost center associated with expensive protecting group reagents and the solvents required for their removal. Furthermore, the shift away from column chromatography to recrystallization drastically reduces the volume of organic waste generated, aligning the manufacturing process with modern green chemistry principles and reducing disposal costs.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the simplification of the synthetic sequence. By removing two distinct steps (protection and deprotection), the overall processing time is shortened, leading to lower labor and utility costs. The avoidance of column chromatography is particularly significant, as chromatographic purification is one of the most expensive unit operations in fine chemical manufacturing due to high solvent usage and low throughput. Additionally, the starting materials, p-nitrophenylethylamine and (R)-styrene oxide, are commodity chemicals that are readily available from multiple global suppliers, ensuring competitive pricing and reducing the risk of raw material shortages.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The use of standard catalytic hydrogenation and ambient temperature coupling reactions minimizes the need for specialized high-pressure or cryogenic equipment, making the process adaptable to a wider range of manufacturing facilities. The high yields reported for each step contribute to a predictable output, allowing for more accurate demand forecasting and inventory management. Moreover, the ability to purify intermediates via recrystallization ensures that the supply of high-quality material can be maintained consistently, reducing the likelihood of batch failures that could disrupt the downstream API production schedule.
- Scalability and Environmental Compliance: Scalability is inherently built into this process design. Recrystallization is a unit operation that scales linearly and predictably, unlike chromatography which faces significant engineering challenges at multi-kilogram or ton scales. This makes the technology ideal for meeting the growing global demand for Mirabegron. From an environmental standpoint, the reduction in solvent consumption and the elimination of hazardous reagents associated with protection chemistry lower the facility's environmental footprint. This alignment with sustainability goals is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility targets and regulatory requirements regarding waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their supply chains.
Q: How does this new synthesis route improve upon conventional Mirabegron manufacturing methods?
A: The novel route eliminates the need for Boc protection and deprotection steps, thereby removing the requirement for column chromatography and significantly simplifying post-treatment procedures.
Q: What are the key advantages regarding scalability and environmental impact?
A: By utilizing recrystallization instead of chromatography and avoiding expensive protecting group reagents, the process reduces solvent waste and is highly suitable for industrial scale-up production.
Q: What level of optical and chemical purity can be achieved with this method?
A: The method achieves high chemical purity (HPLC >99.9%) and maintains excellent optical purity (ee >97%) through controlled reaction conditions and effective recrystallization protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirabegron Supplier
The technological advancements detailed in patent CN103896872A represent a significant leap forward in the manufacturing of Mirabegron intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory processes into robust commercial realities. As a premier CDMO partner, we possess the technical expertise and infrastructure necessary to execute this streamlined synthesis with precision. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite pharmaceutical companies and procurement specialists to explore the potential of this optimized supply chain solution. By partnering with us, you gain access to a reliable source of high-quality Mirabegron intermediates produced via a cost-effective and environmentally friendly route. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain security and drive down your overall production costs.
