Advanced Synthesis of Carbapenem Intermediates via Chiral Oxazolidinone Auxiliaries
Advanced Synthesis of Carbapenem Intermediates via Chiral Oxazolidinone Auxiliaries
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for potent agents against resistant bacterial strains. Patent CN101177416B introduces a significant technological breakthrough in the synthesis of (2'S, 3'R, 4S)-3-[2'-(N-substituted aminomethyl)-3'-hydroxybutyryl]-4-substituted oxazolidin-2-one derivatives. These compounds serve as pivotal precursors for 4-AA ((1'R, 3R, 4R)-3-(1'-hydroxyethyl)-4-acetoxyazetidin-2-one), which is the indispensable key raw material for manufacturing next-generation carbapenem antibiotics such as Meropenem, Ertapenem, and Biapenem. The disclosed methodology addresses long-standing challenges in stereochemical control and process economics, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. By leveraging chiral auxiliary induction, this technology ensures high diastereoselectivity while utilizing reagents that are cheap and easily available, thereby laying a solid foundation for the cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-AA and its precursors has been plagued by significant technical hurdles that impede efficient commercial scale-up of complex pharmaceutical intermediates. Prior art, including various Japanese and European patents cited in the background, often relies on chiral auxiliaries that are notoriously difficult to prepare, such as (4S)-4-isopropylthiazolidine-2-thione. The synthesis of these thiazolidine-based prosthetic groups is cumbersome and has not yet realized stable industrial production, creating a bottleneck in the supply chain. Furthermore, many conventional routes suffer from harsh reaction conditions that require extreme temperatures or pressures, posing safety risks and increasing energy consumption. The diastereoselectivity in these older methods is frequently suboptimal, leading to complex mixtures of stereoisomers that are arduous to separate, thus driving up purification costs and reducing overall yield. These factors combined result in a fragile supply chain with high variability in quality and availability.
The Novel Approach
In stark contrast, the novel approach detailed in CN101177416B utilizes a chiral oxazolidinone auxiliary system that has gradually achieved large-scale industrial production, ensuring a stable and cost-effective supply of starting materials. This method streamlines the synthetic route by employing a condensation reaction between an N-substituted-3-alanine derivative and the chiral auxiliary, followed by a highly selective asymmetric Aldol condensation with acetaldehyde. 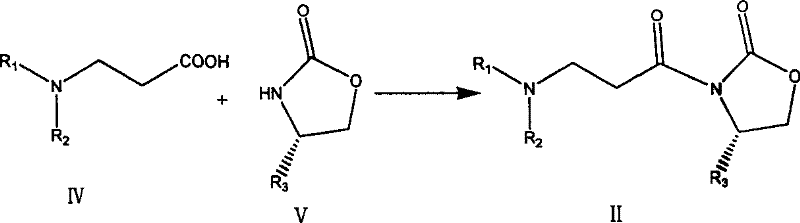 The use of accessible reagents and the avoidance of exotic, hard-to-source catalysts dramatically simplifies the operational protocol. This shift not only enhances the safety profile of the manufacturing process but also significantly improves the economic viability of producing high-purity pharmaceutical intermediates. The transition from difficult thiazolidine systems to robust oxazolidinone chemistry represents a strategic evolution in process development, aligning perfectly with the goals of modern green chemistry and sustainable manufacturing practices.
The use of accessible reagents and the avoidance of exotic, hard-to-source catalysts dramatically simplifies the operational protocol. This shift not only enhances the safety profile of the manufacturing process but also significantly improves the economic viability of producing high-purity pharmaceutical intermediates. The transition from difficult thiazolidine systems to robust oxazolidinone chemistry represents a strategic evolution in process development, aligning perfectly with the goals of modern green chemistry and sustainable manufacturing practices.
Mechanistic Insights into Metal-Catalyzed Asymmetric Aldol Condensation
The core of this technological advancement lies in the precise mechanistic execution of the asymmetric Aldol condensation, which is responsible for simultaneously generating two critical chiral centers with high fidelity. The process begins with the activation of the imide intermediate (Compound II) using a Lewis acid metal catalyst. The patent specifies a broad range of effective catalysts, including titanium species like TiCl4 and TiCl(OiPr)3, as well as tin catalysts such as Sn(OTf)2 and boron catalysts like (n-Bu)2BOTf. These Lewis acids coordinate with the carbonyl oxygen of the imide, increasing the acidity of the alpha-protons and facilitating the formation of a rigid enolate geometry. 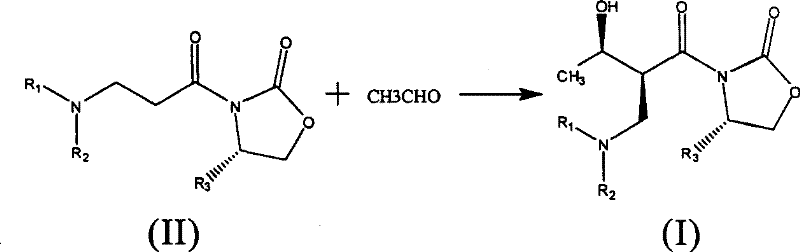 This rigid geometry, enforced by the chiral oxazolidinone ring, dictates the facial selectivity of the subsequent nucleophilic attack on the acetaldehyde electrophile. The presence of a tertiary amine base, such as N,N,N',N'-tetramethylethylenediamine (TMEDA) or diisopropylethylamine (DIPEA), further modulates the reactivity and ensures the smooth progression of the reaction at controlled low temperatures, typically ranging from -80°C to 50°C.
This rigid geometry, enforced by the chiral oxazolidinone ring, dictates the facial selectivity of the subsequent nucleophilic attack on the acetaldehyde electrophile. The presence of a tertiary amine base, such as N,N,N',N'-tetramethylethylenediamine (TMEDA) or diisopropylethylamine (DIPEA), further modulates the reactivity and ensures the smooth progression of the reaction at controlled low temperatures, typically ranging from -80°C to 50°C.
Controlling the impurity profile is paramount for any reliable pharmaceutical intermediate supplier, and this mechanism offers inherent advantages in that regard. The high diastereoselectivity induced by the chiral auxiliary minimizes the formation of unwanted stereoisomers, which are often the most difficult impurities to remove during downstream processing. By optimizing the choice of metal catalyst and solvent system—preferred solvents include tetrahydrofuran, toluene, and dichloromethane—the reaction can be tuned to favor the desired (2'S, 3'R, 4S) configuration exclusively. This precision reduces the burden on purification steps such as recrystallization or column chromatography, leading to a cleaner crude product. The ability to achieve such high stereochemical purity directly translates to reduced waste generation and lower solvent consumption, addressing both quality and environmental compliance concerns simultaneously.
How to Synthesize (2'S,3'R,4S)-Oxazolidinone Derivatives Efficiently
The synthesis of these valuable carbapenem precursors follows a logical two-step sequence that balances chemical efficiency with operational simplicity. The first step involves the coupling of an N-substituted-3-alanine acid with a chiral oxazolidinone using a carboxyl activator like pivaloyl chloride in the presence of a base such as triethylamine. This forms the requisite imide substrate. The second, and most critical step, is the metal-catalyzed aldol reaction with acetaldehyde. Detailed standard operating procedures for temperature control, reagent addition rates, and workup protocols are essential to maintain the high stereoselectivity promised by the patent. For a comprehensive guide on the specific experimental parameters and stoichiometry required to replicate this high-yielding process, please refer to the standardized synthesis steps provided below.
- Activate N-substituted-3-alanine with a carboxyl activator like pivaloyl chloride and couple with a chiral oxazolidinone auxiliary to form the imide intermediate.
- Perform an asymmetric aldol reaction using the imide intermediate, acetaldehyde, a Lewis acid catalyst (e.g., TiCl4), and a tertiary amine base at low temperatures.
- Purify the resulting hydroxybutyryl-oxazolidinone derivative via recrystallization or column chromatography to ensure high diastereomeric purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing obscure and difficult-to-synthesize chiral auxiliaries with commercially mature oxazolidinones, the supply chain becomes far more resilient to disruptions. The reliance on commodity chemicals like acetaldehyde and common Lewis acids further insulates the production process from volatile market fluctuations associated with specialty reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients. Additionally, the operational simplicity reduces the need for highly specialized labor, contributing to overall operational expenditure optimization.
- Cost Reduction in Manufacturing: The elimination of expensive and complex chiral catalysts found in prior art leads to a significant decrease in direct material costs. Since the chiral auxiliary used here is produced on a large scale domestically, its price point is substantially lower than the specialized thiazolidine derivatives previously employed. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, effectively improving the atom economy of the process. This efficiency gain allows for a more competitive pricing structure without compromising on the stringent quality standards required for antibiotic intermediates.
- Enhanced Supply Chain Reliability: The use of readily available reagents ensures that production is not held hostage by the lead times of niche chemical suppliers. Acetaldehyde, titanium tetrachloride, and common organic solvents are staple chemicals in the fine chemical industry, guaranteeing a consistent flow of inputs. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond swiftly to surges in demand for carbapenem antibiotics. The robust nature of the process also means that scaling up from pilot plant to full commercial production can be achieved with minimal risk of failure.
- Scalability and Environmental Compliance: The reaction conditions described, operating generally between -80°C and 50°C, are well within the capabilities of standard industrial reactors, facilitating easy commercial scale-up of complex intermediates. The process avoids the use of heavy metals that are difficult to remove, simplifying the purification workflow and reducing the environmental burden of waste disposal. By adhering to greener chemistry principles through improved selectivity and safer reagents, manufacturers can more easily comply with increasingly strict environmental regulations, avoiding potential fines and shutdowns while enhancing their corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on how this technology integrates into existing manufacturing frameworks. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this new process for their specific product portfolios. The answers reflect a balance of chemical rigor and practical industrial application.
Q: What are the advantages of this oxazolidinone route over traditional thiazolidine methods?
A: Unlike traditional methods using difficult-to-prepare thiazolidine-2-thione auxiliaries, this patent utilizes commercially available oxazolidinones. This shift significantly simplifies raw material sourcing and reduces the complexity of the initial synthesis steps, making the process more amenable to large-scale industrial production.
Q: How does the process control stereochemistry for carbapenem antibiotics?
A: The method employs a chiral auxiliary-induced asymmetric Aldol condensation. By using specific metal catalysts like titanium tetrachloride or tin triflate in conjunction with the chiral oxazolidinone ring, the reaction achieves high diastereoselectivity, efficiently establishing the critical chiral centers required for biological activity in the final antibiotic.
Q: Is this synthesis method scalable for commercial API manufacturing?
A: Yes, the patent explicitly highlights the suitability for industrialized production. The reagents used, such as acetaldehyde and common Lewis acids, are inexpensive and readily available. Furthermore, the reaction conditions operate within a manageable temperature range (-80°C to 50°C), avoiding the extreme hazards often associated with cryogenic pharmaceutical processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex chiral syntheses, such as the oxazolidinone-mediated aldol condensation described in CN101177416B, positions us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis route to your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
